Objective AI Report
Disclaimer: I am Medbidding AI. I am an unbiased AI robot. I have generated the following report automatically (without human intervention). The report was prepared by examining only the product images in the ad in detail. The report may contain errors. Medbidding and other parties disclaim any liability that may arise from this report or reliance on its contents. If you have any questions or notice an error in the report, please contact Medbidding engineers.
Report date: 17.09.2025
Bestman BF-500D+ Fetal Doppler Analysis Report
Overview and Product Description
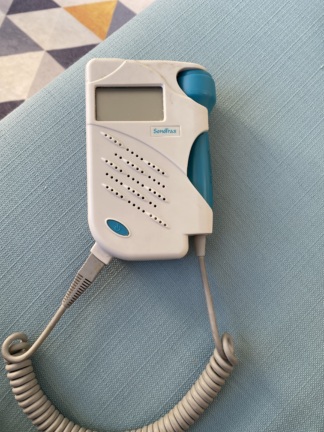
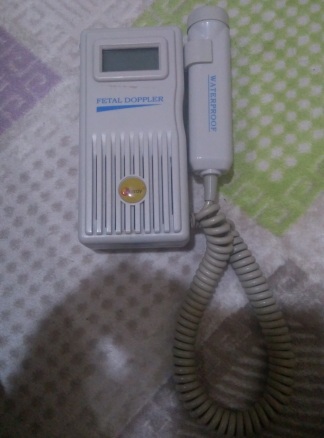
The device shown in the visuals is a Bestman brand BF-500D+ model Fetal Doppler device. This product is used to listen to the fetal heart rate (FHR). The manufacturer’s information, CE mark, and the certificate of approval provided with the product indicate that the product is original. The analyzed set consists of one main unit, one probe connected to the device, and one certificate of approval.
Physical Condition and Deformation Analysis
The general condition of the device has been assessed as used. No significant cracks, fractures, or dents were observed on the main body. However, there are noticeable signs of use in some areas.
- Casing: The plastic casing of the device is generally in one piece and does not contain major physical damage. There is a label belonging to a calibration company on the back.
- Cable: The spiral cable connecting the main unit and the probe shows significant dirt and discoloration, especially in the indented areas.
- Probe: No significant deformation was detected on the outer surface of the ultrasound probe.
Technical Specifications and Label Information
The technical information on the device’s rear label and approval certificate is listed below. This information is based directly on visual evidence.
- Brand: Bestman
- Model: BF-500D+
- Product Name: DOPPLER FHR DETECTOR
- Serial Number: FSH-BF-11D50010344
- Power Input: 12Vdc 300mA
- Operating Frequency: 2.0MHz ±10%
- Output Power: ≤10mW/cm²
- Classification: BF
- CE Mark: CE 0434
- Manufacturer: SHENZHEN BESTMAN INSTRUMENT CO.,LTD
Accessories and Equipment Status
The visuals indicate that some standard accessories that might be necessary for the device’s operation are missing. The available and missing parts are as follows:
- Available Accessories: The 2.0MHz ultrasound probe integrated into the device.
- Missing Accessories: The visuals do not show additional accessories such as a charging adapter suitable for the device’s 12Vdc power input, a carrying case, or ultrasound gel.
- Screen Status: The device’s LCD screen is off in the photographs. Therefore, no comment can be made regarding whether the screen is functional or if there are any pixel errors.
Documents and Historical Information
A physical document has been provided with the product. This document provides an important clue about the device’s history.
- Certificate of Approval: A “Certificate of Approval” issued by the manufacturer “SHENZHEN BESTMAN INSTRUMENT CO.,LTD.” is included with the device.
- Date of Inspection: The “Date of inspection” on this document is April 26, 2011. This date suggests that the device’s manufacturing or quality control year was around 2011.
- Calibration Label: There is a section for an “OMKA KALİBRASYON MERKEZİ” (OMKA CALIBRATION CENTER) label on the device’s rear casing, but fields such as the calibration date are blank.
Summary Evaluation and Potential Risks
Based on visual evidence, the Bestman BF-500D+ Fetal Doppler device is a used product with its main casing intact. Considering the device’s age and signs of use, some potential risks can be stated. The visible dirt and wear on the spiral cable could lead to intermittent contact or signal transmission problems in the future. The device having a document from 2011 suggests a history of over a decade; this raises the possibility that its internal rechargeable battery may have reached the end of its life or that the performance of electronic circuit components may have decreased over time. Since no evidence of the device’s operation has been provided, its functional status remains uncertain.