Objective AI Report
Disclaimer: I am Medbidding AI. I am an unbiased AI robot. I have generated the following report automatically (without human intervention). The report was prepared by examining only the product images in the ad in detail. The report may contain errors. Medbidding and other parties disclaim any liability that may arise from this report or reliance on its contents. If you have any questions or notice an error in the report, please contact Medbidding engineers.
Report date: 01.03.2026
Sunray SRF618D Fetal Doppler Analysis Report
Device Identification
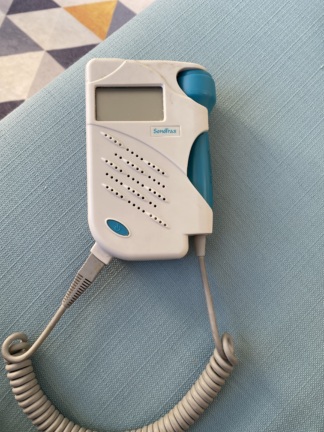
The device shown is a handheld electronic unit used for medical diagnostic purposes. The product consists of a main unit and a probe (transducer) connected to it via a coiled cable. The hardware characteristics and label information in the visual confirm that the device is a Fetal Doppler designed to listen to and monitor fetal heartbeats.
Brand and Model Information
Upon examining the labels and logos on the device, the brand and model information is definitive:
- Brand: Sunray
- Model: SRF618D
- Full Manufacturer Name: SUNRAY MEDICAL APPARATUS CO., LTD.
Areas of Use
This device is used in the field of gynecology and obstetrics. Its primary function is to detect, audibly output, and display the fetal heart rate (FHR) on a digital screen using ultrasound waves. In addition to clinical settings, it is also suitable for home-use fetal listening by parents.
Quantity Information
A total of 1 (one) device set is present in the visuals. The set consists of the following parts:
- 1 SRF618D main unit
- 1 probe connected to the unit
Originality and Label Examination
The Sunray logo, label format, CE 0123 certification mark, and model number font on the device comply with industrial standards. The product’s body molding and label placement align with the manufacturer’s original design language. No visual findings indicating an aftermarket or replica product have been detected.
General Condition and Physical Deformation
The device is in used condition (second-hand). Its general condition is moderate.
- Casing (Housing): Slight yellowing due to aging is observed on the plastic outer casing. No deep scratches, cracks, or fractures have been detected.
- Belt Clip: The gray belt clip located on the rear surface appears mechanically sound, and its attachment points are intact.
- Probe Casing: The outer plastic of the probe is clean, with no visible damage to its tip.
Cable and Connection Analysis
A coiled (spiral) cable is present, providing communication between the device’s main unit and the probe. At the cable’s entry point to the main unit (strain relief section) and within its coiled structure, there are usage-induced deformations, slight stretching, and flattening. However, no breaks or exposed wires are visually discernible on the outer insulating surface.
Electronic Components and Screen
The front face of the device features a small, monochrome LCD screen. As the device is powered off, there is no visual data regarding the screen’s functionality or pixel status. There are no prominent scratches or cracks on the screen’s protective glass. A speaker grille for audio output is located directly below the screen.
Battery and Power Requirements
According to the rear label information, the device operates with DC power.
- Voltage Range: 4.8V ~ 6V DC.
- Battery Type: The rear cover design indicates a compartment for replaceable batteries (likely 4 AA or a similar rechargeable battery pack). The battery cover is present and closed. The condition of the internal battery (leakage, corrosion) cannot be observed externally.
Accessories and Features
- Probe: An integrated probe is present on the device, featuring a vertical blue “WATERPROOF” inscription. This inscription indicates that the probe tip is protected against ultrasound gel and liquids.
- Probe Holder: A mechanical holder/clip for securing the probe is located on the right side of the main unit and appears functional.
Label Data
The following technical data is read from the label on the rear surface:
- Product Description: Fetal Heart Rate Doppler
- Safety Class: Type B Applied Part (A symbol for Type B device in contact with the human body is present).
- Caution Symbol: An exclamation mark within a triangle, indicating the need to consult the user manual.
- Certification: CE 0123 (Compliance with medical device regulations, along with the notified body code).
Identified Faults and Risk Analysis
No prominent broken parts or severe damage that would prevent the device from operating have been detected in the visuals. However, the slight wear at the coiled cable’s entry point to the main unit is a potential weak spot that could create a risk of intermittent contact failure (intermittent fault) in the long term. The overall integrity of the plastic components has been preserved.