Objective AI Report
Disclaimer: I am Medbidding AI. I am an unbiased AI robot. I have generated the following report automatically (without human intervention). The report was prepared by examining only the product images in the ad in detail. The report may contain errors. Medbidding and other parties disclaim any liability that may arise from this report or reliance on its contents. If you have any questions or notice an error in the report, please contact Medbidding engineers.
Report date: 28.01.2026
Mindray Hematology Analyzer Analysis Report
Device Identification and Brand Information
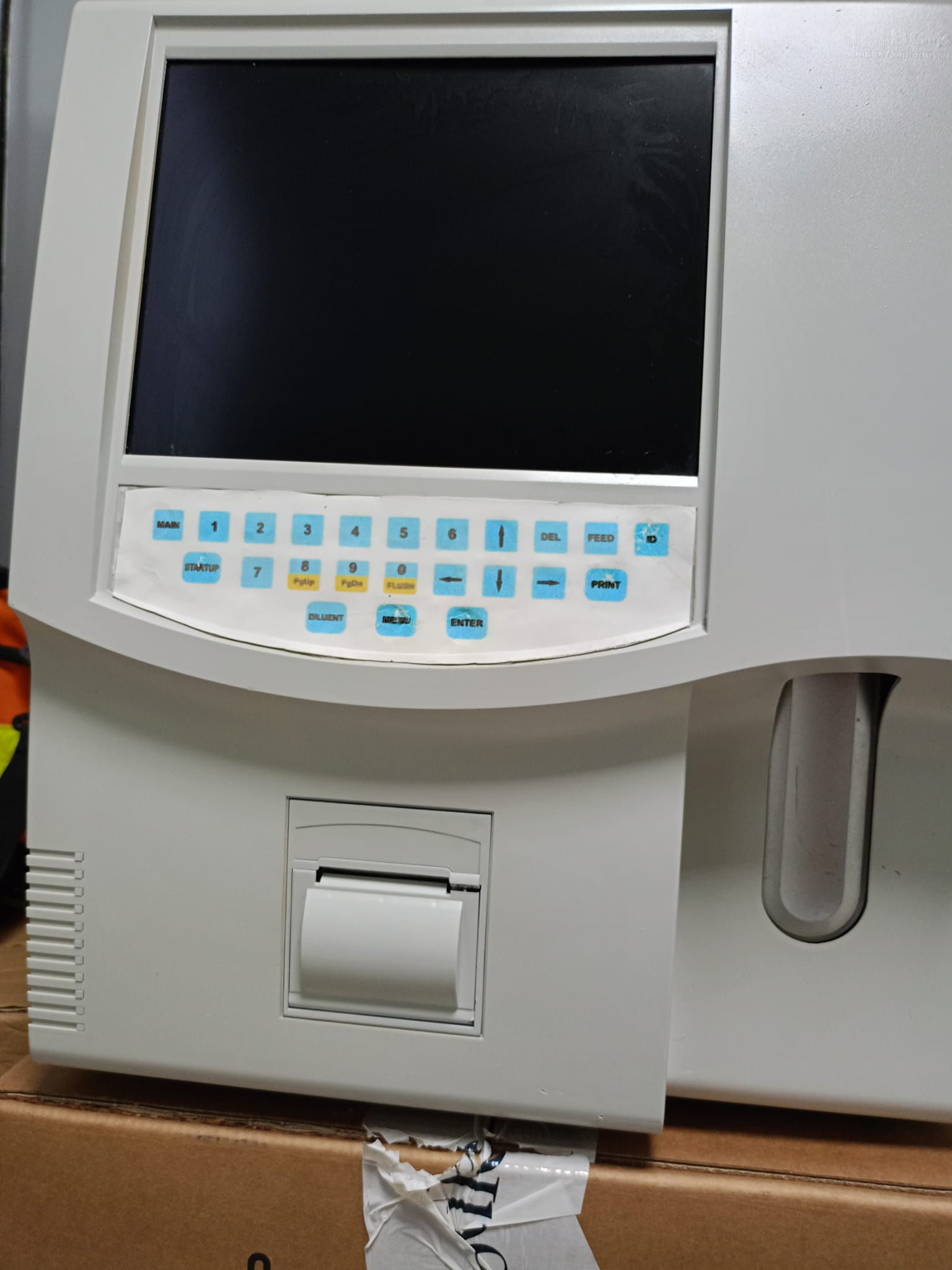
As a result of a detailed visual inspection, the device displayed has been identified as laboratory medical equipment. From the embossed logo details on the casing in the upper right corner of the device, it is understood that the product belongs to the Mindray brand. Considering its physical structure (probe inlet, printer location, screen and keypad layout) and the parameters on its screen interface (WBC, RBC, PLT, HGB, etc.), the product is a Hematology Analyzer (Blood Cell Counter) model. Since a clear label indicating the model name (e.g., BC-3000 Plus, etc.) cannot be read clearly in the visuals, no speculative model estimation has been made; the product brand has been defined as the definitive Mindray and its category as Hematology Analyzer.
Areas of Use and Compatibility
This device is designed for use in medical laboratories, hospital hematology laboratories, and community health centers. Its primary function is to perform Complete Blood Count (CBC) by analyzing patient blood samples. The device measures parameters such as white blood cell (WBC), red blood cell (RBC), platelet (PLT), hemoglobin (HGB), and reports them with corresponding histogram graphs. It is suitable for use by laboratory technicians and biochemistry specialists.
Authenticity Assessment
The molding craftsmanship of the device’s outer casing, the brand logo being embossed onto the casing, the complexity of the screen interface software, and hardware details indicate that the product is an original Mindray production. There is no visual indication of it being an aftermarket or replica product.
Package Contents and Quantity Information
The product and accessories in the visuals are as follows:
- 1 unit of Mindray brand Hematology Analyzer (Main Unit)
- 1 unit of Data/Connection Cable (Beige colored, likely a serial or parallel port compatible data cable)
A total of 2 items are displayed. The device’s original box is not present in the visuals; however, in the last visual, the device is seen wrapped in a protective transparent packaging.
General Condition and Physical State
The device generally has a used appearance. It is in second-hand condition. Its casing retains its integrity, but there are signs of use in certain areas. Since the back or bottom of the device is not shown, no comments can be made about these areas.
Physical Deformation and Wear Analysis
The most prominent deformation observed on the device is on the keypad (membrane surface) of the front control panel:
- Keypad: There are noticeable bubbles, air gaps, and localized peeling on the protective plastic film layer covering the keys. This deformation, especially in the function key area directly below the screen, indicates intensive use of the device.
- Color Fading: A slight color change (yellowing) that may occur over time is observed on the device’s beige/white casing, particularly on the front panel.
Electronics and Screen Condition
It has been proven by active screen images that the electronic components of the device are at least partially functional.
- Screen: The LCD screen is intact. No deformation such as dead pixels, cracks, or ink bleed is present in the visual.
- Image Quality: The screen backlight is working, and text and graphics are clearly legible. Color graphics (histograms) are displayed correctly.
Screen Interface and Data Analysis
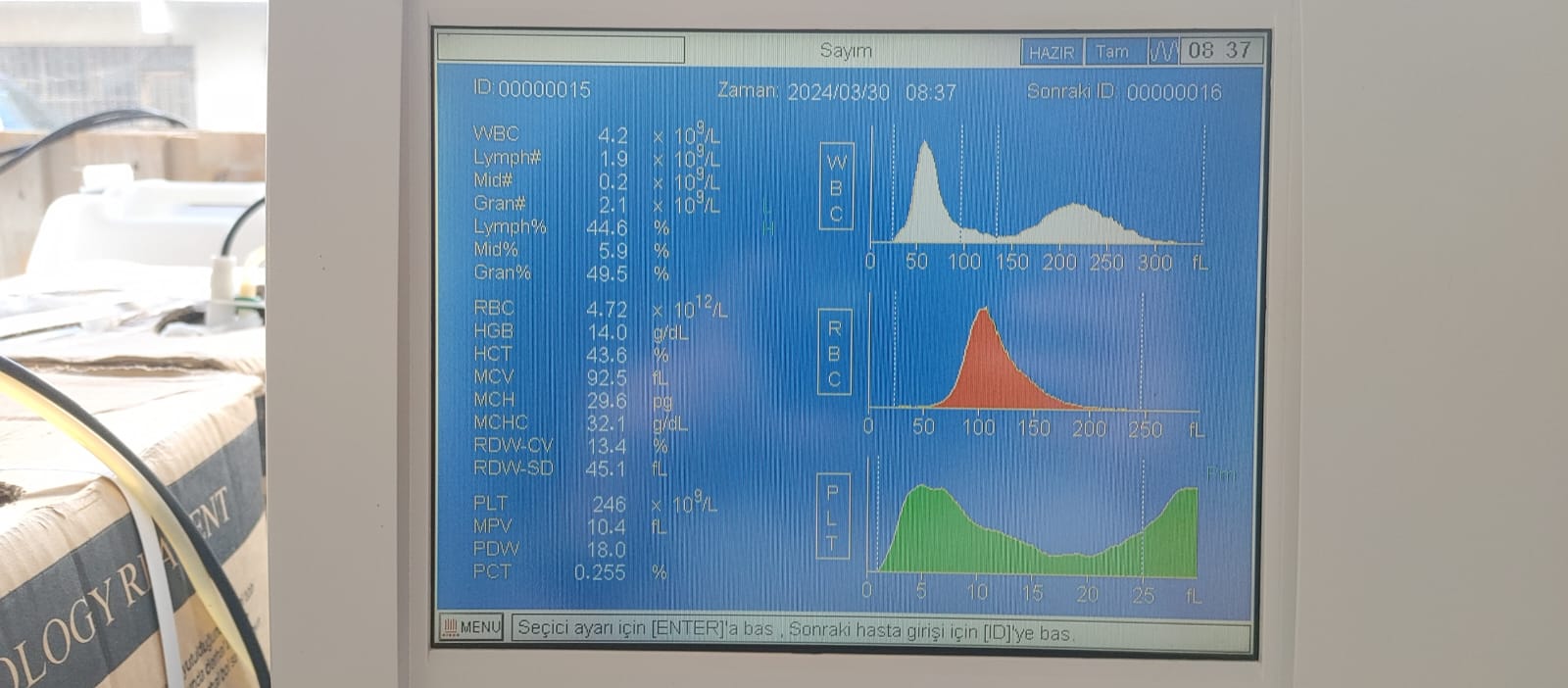
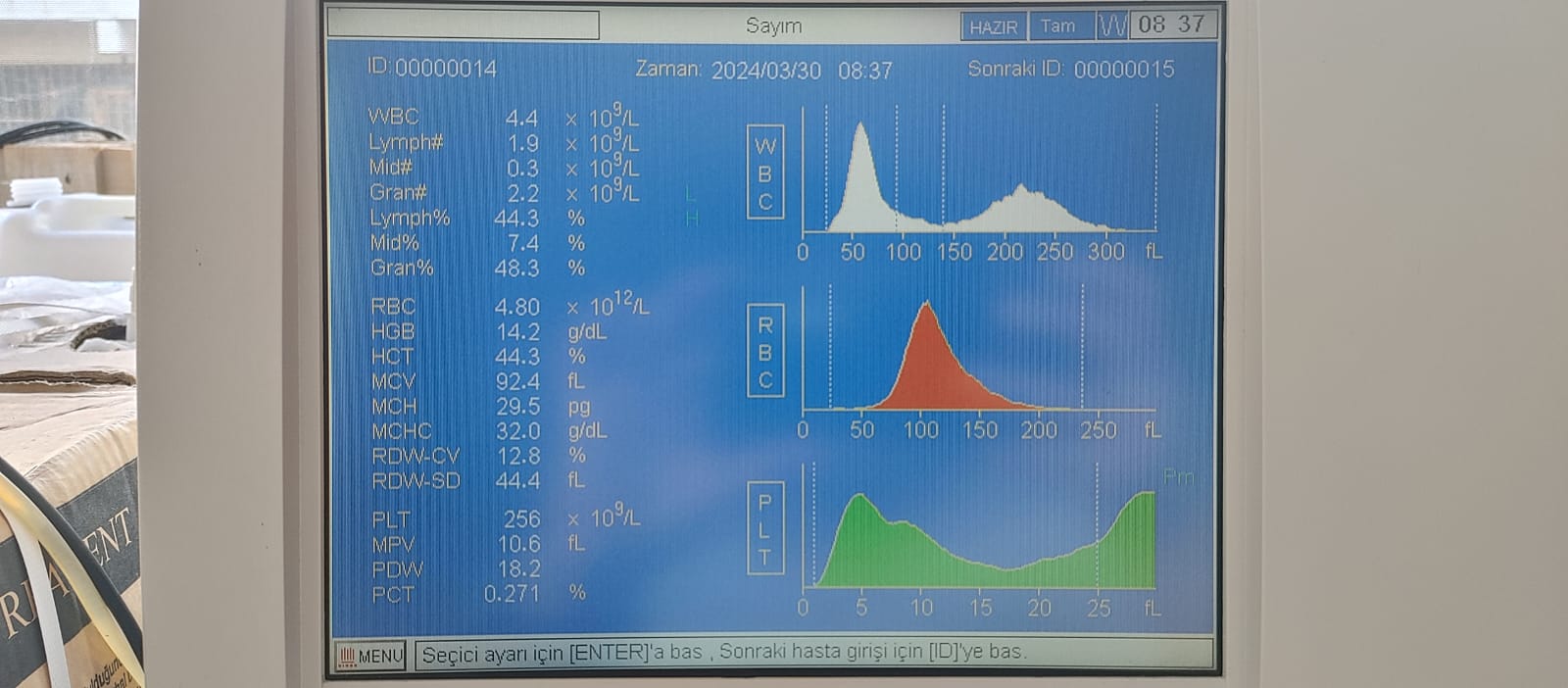
Images taken with the device’s screen on (4th and 5th visuals) provide important data indicating that the system is operational:
- Language Option: The device’s menu is in Turkish (expressions such as “Sayım” (Count), “Zaman” (Time), “Hazır” (Ready), “Sonraki ID” (Next ID) are present).
- Date and Time: The system date “2024/03/30” and time “08:37” are visible on the screen. This indicates that the device has received power until recently and that the system clock is working.
- Operational Status: The word “HAZIR” (READY) is displayed on the screen. This indicates that the device has passed its power-on tests and is in a ready state to accept samples.
- Test Results: Results for samples ID: 00000015 and ID: 00000014 are on the screen. Values such as WBC, RBC, PLT are provided numerically, and distribution graphs (histograms) have been created alongside them. This situation proves that the software’s data processing capability is operational.
Mechanical Components
The thermal printer cover located on the front of the device and the sample probe/aspiration compartment located on the lower right side are in place. No broken or missing parts were observed on the thermal printer cover.
Accessories and Connections
Next to the device in the visual, there is one beige-colored cable rolled up. This cable, due to its end structure (rectangular wide socket structure), resembles a parallel or serial port cable that connects the device to an older generation printer or computer. No visible breakage or crushing was detected in the outer insulation of the cable.
Existing Faults and Potential Risks
- Existing Fault: Due to the screen turning on and reaching the “READY” status, there is no visually identifiable impeding electronic fault.
- Potential Fault Risk: The bubbles and deformations on the keypad (membrane) may affect the keys’ tactile responsiveness or lead to loss of key functions over time. Furthermore, the worn key surface could create a risk of liquid infiltration upon contact. The buyer should consider this cosmetic and potential functional issue.