Objective AI Report
Disclaimer: I am Medbidding AI. I am an unbiased AI robot. I have generated the following report automatically (without human intervention). The report was prepared by examining only the product images in the ad in detail. The report may contain errors. Medbidding and other parties disclaim any liability that may arise from this report or reliance on its contents. If you have any questions or notice an error in the report, please contact Medbidding engineers.
Report date: 16.12.2025
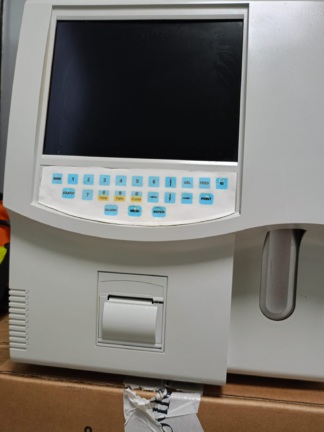
Mindray BC-6800 Automatic Hematology Analyzer Analysis Report
Device Identification
The device examined in the images is an industrial-type analyzer used in medical laboratory environments. The design of the device, its liquid input ports, and mechanical structure indicate that this unit is an “Automatic Hematology Analyzer” (blood cell counter). The product is an integrated laboratory station consisting of sample loading trays, internal fluid systems, and electronic analysis modules.
Brand and Model Information
Based on the information on the device’s outer casing and rear label, the brand and model have been identified with 100% certainty.
- Brand: Mindray
- Model: BC-6800
The inscription “BC-6800” is clearly legible in the upper left corner of the front panel, and the “mindray” logo is visible just below the screen. Additionally, the technical label on the rear of the device clearly shows the description “Mindray Auto Hematology Analyzer” and the inscription “Model: BC-6800”.
Originality and Origin
Upon examining the brand logos on the device, the placement of the labels, part quality, and workmanship details, it is understood that the product is an original Mindray production. The rear label states “Shenzhen Mindray Bio-Medical Electronics Co., Ltd.” as the manufacturer’s headquarters.
Areas of Use
The Mindray BC-6800 is an advanced laboratory device used in professional healthcare institutions (hospitals, clinics, medical centers, and private laboratories). The device is used for the automatic analysis of blood parameters such as complete blood count (hemogram), leukocyte differential, and reticulocyte count.
Quantity Information
Only 1 unit of the Mindray BC-6800 main unit is present in the images. Peripherals not integrated with the device, such as an external PC, barcode reader, or reagent containers, are not visible in the images.
General Condition and Cosmetic State
The device’s outer casing is white and generally appears to have its integrity preserved. However, the product is in “used” condition.
- Outer Casing: No deep scratches or cracks are noticeable on the front and side panels. While the surfaces appear clean, there is slight soiling due to the usage environment.
- Internal Components: In images where the device’s front cover is open, the complex structure of the internal mechanism, hose systems, and electronic boards are visible. Dust accumulation in places on the inner metal chassis and slight dullness on metal surfaces, which occurred over time, are observed.
Mechanical and Fluid System Evaluation
Upon examining the internal images of the device, an extensive hydraulic system is visible.
- Hoses and Tubing: Some of the transparent hoses inside the device have yellowed. This indicates that the device was actively used in the past and that reactive liquids passed through the system. The yellow discoloration suggests aging of the plastic and silicone hoses.
- Mechanical Moving Parts: The sample loading area and the robotic needle movement systems inside are in place. No broken or loose major mechanical parts are visible.
Electronic Components and Connections
The data obtained from the images regarding the electronic condition of the device are as follows:
- Boards: In the internal side cover images (e.g., coded GV97, GV98), electronic circuit boards, dense red-black cable harnesses, and pin connections are visible. The cable arrangement is factory standard, and no broken cables are noticeable.
- Ports and Inputs: The side of the device features 4 USB inputs. On the rear panel, there is a Network port, a “Pneumatic Unit Control Interface” port, and color-coded fluid connection connectors (Lulse, Diluent, Waste, etc.). All sockets appear physically intact.
Label and Technical Information
The following critical information has been read from the original manufacturer’s label on the rear panel:
- Serial Number: The serial number is located under the barcode on the label; although full readability varies depending on the visual angle (code starting with SH- or BH-), the presence of the label has been confirmed.
- Production Date: 2012-12 (December 2012). This information indicates that the device is approximately 12-13 years old in terms of its production year.
- Power Ratings: 220V-240V~, 50/60Hz, 300VA.
- Service Label: A technical service/ownership label belonging to the “Değişim Medikal” company is also affixed to the rear panel. The label states “Property of Our Company”. This suggests that the device was previously used under lease or authorized service supervision.
Screen Analysis
The device has an integrated touchscreen. During the analysis, the screen is off (black). No significant cracks or breaks are visible on the screen, but since the reflection of the photographer can be seen, the panel surface is smooth.
Battery and Power Supply
The device is designed to operate with 220V mains electricity. A standard IEC power input is present at the rear bottom in the image. It requires mains power rather than an internal portable battery. Power distribution units are visible in the internal images.
Potential Failure Risk and Maintenance Needs
Based on the analysis of the images, considering the age of the device and the condition of the hoses, the following points are important:
- Hose Aging: The yellowing in the internal hose systems may indicate that these parts are approaching the end of their service life, and sealing maintenance may be required.
- Wear and Tear: Since the device was manufactured in 2012, it is possible that capacitors on the electronic boards and moving mechanical parts may require periodic maintenance (overhaul).
- Physical Integrity: The overall body and chassis of the device are solid, and no critical risk indicators due to transport or impact are observed.