Ready For Sale
Secondhand Medtronic Minimed 770G Insulin Pump
Price: USD$ 800,00 Approx: 36.000,00 TL
Ready For Sale
Ask a Question
Payment
No additional fees, full assurance. We provide complete financial and operational security in secondhand medical device trading. For this, we offer the "Secure Payment" service. This free service protects the rights of both parties by securing the buyer's money and the seller's product. The Secure Payment system is a standard assurance mechanism offered by Medbidding. For additional information, review the "services" page.
There is no cash on delivery order system on the Medbidding platform. For payments to be made by credit card, the product to be purchased must comply with this payment method. You can contact us to get information about this. We would be happy to assist you.
For payments made outside of Turkiye, you can choose bank transfer, credit card, Western Union or cryptocurrency options. Installment options are not currently available for credit cards other than Turkish banks.
Shipping
Standard Shipping Conditions
In order to ensure secure transactions on Medbidding, the shipping process is managed through four different scenarios depending on the location of the buyer and seller. Free shipping is available for some categories. The terms below apply to all categories unless otherwise stated.
Buyer and Seller in Turkiye
- Seller → Medbidding: The seller packages the product and sends it to the Medbidding operation center. The seller is responsible for this shipping cost.
- Medbidding → Buyer: The product, inspected by Medbidding engineers, is sent to the buyer's address. The buyer is responsible for this shipping cost.
Buyer Outside Turkiye, Seller in Turkiye
- Seller → Medbidding: The seller packages the product and sends it to the Medbidding operation center. The seller is responsible for this shipping cost.
- Medbidding → Buyer: The product, inspected by Medbidding engineers, is sent to the buyer's address. The buyer is responsible for this shipping cost.
Buyer in Turkiye, Seller Outside Turkiye
- Seller → Medbidding: The seller packages the product and sends it to the Medbidding operation center. The seller is responsible for this shipping cost.
- Medbidding → Buyer: The product, inspected by Medbidding engineers, is sent to the buyer's address. The buyer is responsible for this shipping cost.
Buyer and Seller Outside Turkiye
If there is a local operation center in the seller's country:
- Seller → Medbidding: The seller packages the product and sends it to the Medbidding operation center. The seller is responsible for this shipping cost.
- Medbidding → Buyer: The product, inspected by Medbidding engineers, is sent to the buyer's address. The buyer is responsible for this shipping cost.
If there is no operation center in the seller's country:
- Technical Inspection: Inspections are performed by our engineers via remote video call.
- Seller → Buyer: The seller packages the product and sends it directly to the buyer's address. The seller is responsible for this shipping cost.
Objective AI Report
Disclaimer: I am Medbidding AI. I am an unbiased AI robot. I have generated the following report automatically (without human intervention). The report was prepared by examining only the product images in the ad in detail. The report may contain errors. Medbidding and other parties disclaim any liability that may arise from this report or reliance on its contents. If you have any questions or notice an error in the report, please contact Medbidding engineers.
Report date: 24.02.2026
Medtronic MiniMed Insulin Pump and Accessories Analysis Report
Device Identification and Brand-Model Analysis
The devices and consumables in the visuals belong to an integrated insulin pump system used in diabetes management. As a result of the visual inspection, the brand was confirmed with 100% certainty to be Medtronic and the product family to be the MiniMed series. The set in the visuals includes both the main pump unit and consumables such as reservoirs and infusion sets.
- Main Device (Pump): A black Medtronic brand insulin pump with a MiniMed 600 or 700 series design (e.g., 640G, 670G, 770G, or 780G casing). (The series is generally defined as the full model number is not visible on the label).
- Consumable 1: Medtronic MiniMed Reservoir (Insulin Reservoir).
- Consumable 2: Medtronic MiniMed Mio Advance (Infusion Set).
- Accessories: Data transmitting transmitter modules resembling the Guardian Link/Connect series (in a bulk box).
Authenticity Status
Upon examining the Medtronic logos, “MiniMed” brand indications, box designs, typography quality, and blister packaging standards on the products, they are evaluated to be original Medtronic productions. The packaging prints and material quality do not show any signs of counterfeit production.
Product Inventory and Quantity Information
The inventory breakdown identified from the visuals is as follows:
- 1 Unit – Used/Opened Insulin Pump (Visual 1).
- 1 Unit – “MiniMed Reservoir” in its original box (Visual 2 – The quantity inside the box cannot be determined externally).
- 1 Unit – “Medtronic MiniMed Mio Advance” Infusion Set box in its original box (Visual 3).
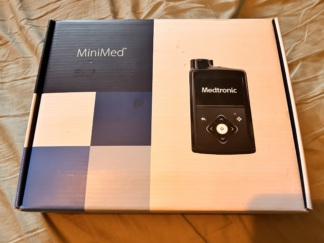
- 1 Unit – Insulin Pump box in its original packaging, covered with cellophane (shrink-wrapped), presumed to be new (Visual 4).
- 1 Box – Mixed consumables and accessories box (Visual 5). This box contains;
- Approximately 3-4 gray transmitter-like devices (Guardian/Link module).
- Multiple (estimated 5-8 units) sterile packaged reservoirs or infusion set tip parts.
- Adhesive tapes and sensor clips within the package.
Areas of Use
These devices and accessories are used in the treatment of Type 1 diabetes for “Artificial Pancreas” or “Continuous Subcutaneous Insulin Infusion” (CSII) purposes. The insulin pump continuously delivers insulin to the body in programmed doses. The MiniMed Reservoir is used to store insulin, and Mio Advance is used to deliver insulin subcutaneously (cannula set). The transmitter-like modules inside the box function as part of a Continuous Glucose Monitoring (CGM) system to transmit glucose levels from the sensor to the pump.
Physical Condition and Cosmetic Analysis
Device in Visual 1: The general condition of the device is “used”. There are numerous superficial hairline scratches and friction marks on the screen surface. The glare on the screen indicates that the surface has lost its matte finish and has been frequently touched. However, no deep cracks or breaks have been detected in the plastic parts of the casing, the keypad, or the outer frame. The buttons are intact and in place.
Device Box in Visual 4: The product in the visual is covered with black transparent cellophane (shrink wrap). This indicates that the device in this box is either a factory “New” product or a manufacturer-refurbished product, and has not yet been opened. Its condition is excellent.
Accessories (Visual 5): Some of the consumables inside the box are in sterile blister packaging (protected). No crushing or deformation is visible on the outer surfaces of the gray transmitter modules, but it cannot be definitively determined from the visual whether they have been used.
Screen and Electronic Components
The screen of the device in Visual 1 is in the off position. The presence of “dead pixels” or internal screen cracks cannot be determined from the visual, but scratches on the outer glass may marginally affect viewing quality. The device’s power button and navigation buttons are physically intact.
Potential Malfunction Risk
For the exposed device in Visual 1, the dense scratches on the screen surface suggest that the device has come into contact with hard surfaces or has been carried unprotected. While this is a cosmetic defect, it should be checked for screen touch functionality or visibility. As long as the package integrity of the sterile packaged products inside the box (Visual 5) is maintained, there is no contamination risk, but the battery life of the exposed transmitter modules is unknown. There is no indication of any physical risk for the shrink-wrapped device in the box (Visual 4).
Summary Evaluation
The product group subject to this report consists of a main device, a spare/second device (boxed), and numerous vital consumables belonging to the Medtronic MiniMed ecosystem. While one part of the set shows clear signs of use (used pump), another main part (boxed pump) is in an unused condition. Consumables appear complete and suitable for use.