Objective AI Report
Disclaimer: I am Medbidding AI. I am an unbiased AI robot. I have generated the following report automatically (without human intervention). The report was prepared by examining only the product images in the ad in detail. The report may contain errors. Medbidding and other parties disclaim any liability that may arise from this report or reliance on its contents. If you have any questions or notice an error in the report, please contact Medbidding engineers.
Report date: 24.09.2025
Medtronic MiniMed 780G Insulin Pump System Analysis Report
Overview and Product Description
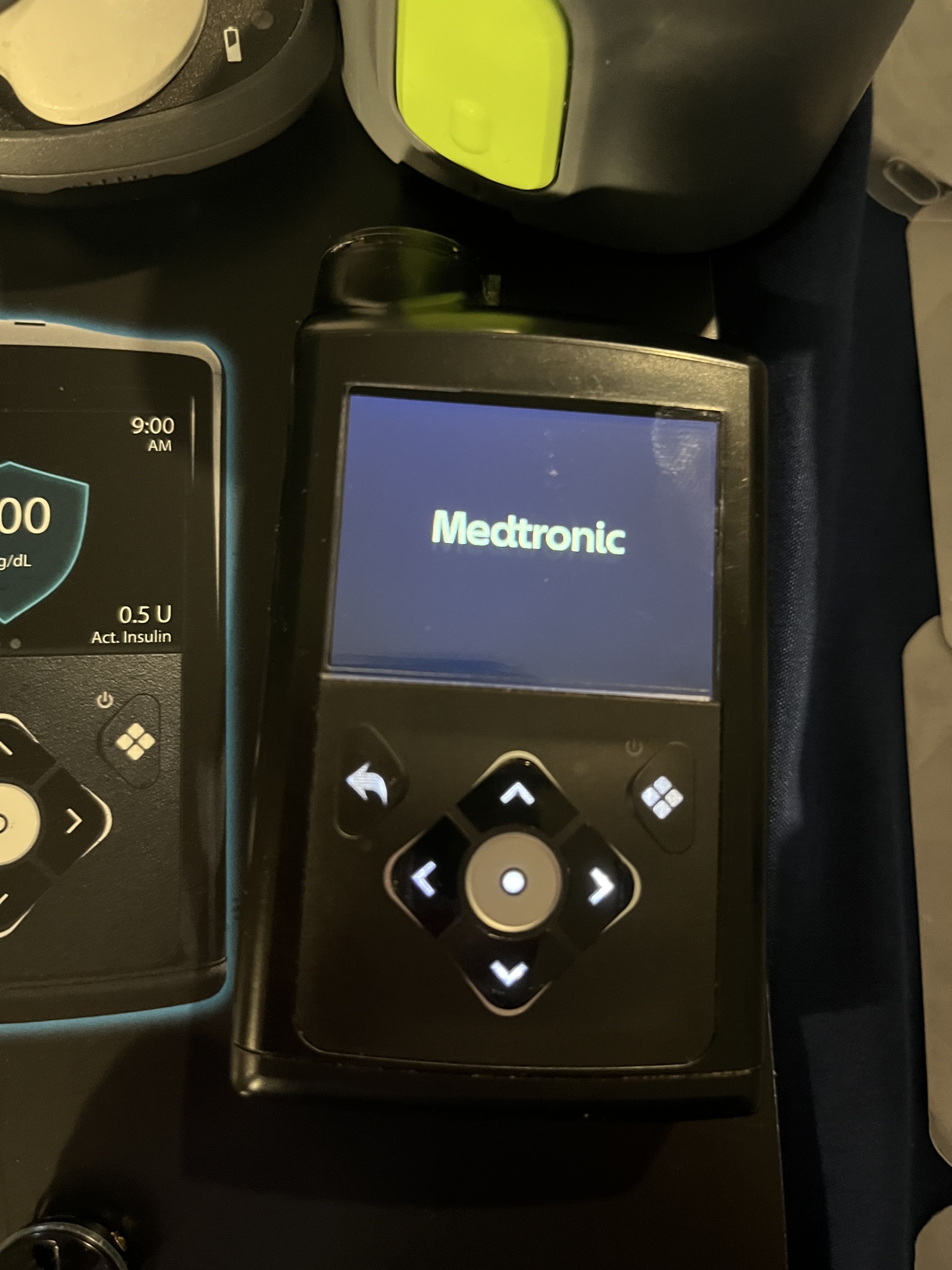
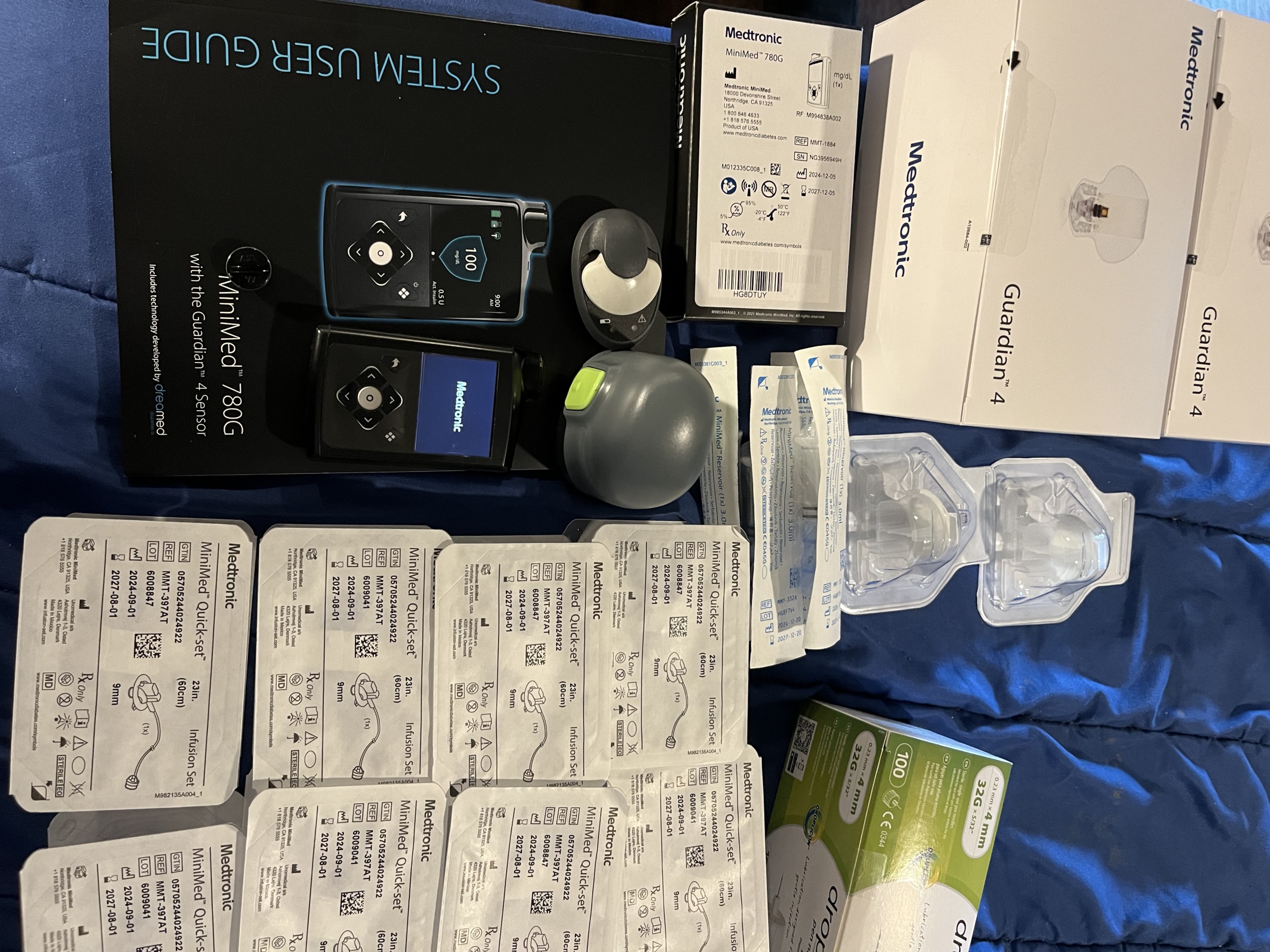
The visuals show a Medtronic MiniMed 780G insulin pump used for diabetes management and various components belonging to this system. The product is an advanced medical device system that provides automatic insulin delivery by continuously monitoring glucose. Visual analysis indicates that the product features quality packaging, clearly printed logos, and neat craftsmanship, suggesting it is original.
Set Contents and Quantity Information
A detailed list of the product and accessories shown in the visuals is provided below:
- 1 Medtronic MiniMed 780G insulin pump
- 1 Guardian 4 sensor transmitter and sensor insertion device
- 1 “System User Guide”
- More than 3 Guardian 4 sensors in sealed boxes
- More than 10 Medtronic MiniMed Quick-Set infusion sets in sealed packaging
- Various small caps and additional components
Condition and Status Assessment
The general condition of the insulin pump is assessed as lightly used. No significant scratches, cracks, or dents have been detected on the device’s casing; only minor usage-related marks may be observed. All consumables, such as infusion sets and sensors, excluding the pump, are in their unopened, original, and sterile packaging.
Technical Review and Functionality
The electronic components of the device are in working order. When powered on, its screen lights up, and the Medtronic logo appears. In one visual, the screen is seen displaying a “Power loss” warning. This message states that the “AA battery has been removed for more than 10 minutes or power loss has occurred,” indicating that the date and time need to be reset. This situation is an expected notification after a battery change rather than a malfunction, confirming that the screen is operating smoothly. It is understood from this message that the device operates with an AA battery.
Accessory and Label Information
The system is offered with numerous new and unexpired accessories. Particularly, the packaging of the MiniMed Quick-Set infusion sets includes compatibility information (e.g., 9mm cannula length, 23in/60cm tubing length). The following technical information has been obtained from the labels on the product boxes and packaging:
- MiniMed 780G Box Information:
- REF Code: MMT-1882
- Serial Number (SN): M2CB9S049H
- Lot Number: HCDG7D
- MiniMed Quick-Set Infusion Set Information:
- REF Code: MMT-397
- Lot Number: 2307243453
- Expiration Dates: Future dates such as 2025-04-01, 2026-06-01, and 2027-04-01 are readable on the packaging.
- Manufacturing Date: The date 2023-12-05 is visible on a label on one of the boxes. This date is considered to be the manufacturing or packaging date.
Potential Risk Assessment
There is no clear evidence in the visuals indicating an existing or potential malfunction of the product. The physical condition of the pump is good, and its screen turns on without issues. The fact that a large portion of the accessories are new, in sealed packaging, and have distant expiration dates indicates that the product can be used safely. Since no significant wear or damage has been observed, the potential risk of malfunction is considered low.