Objective AI Report
Disclaimer: I am Medbidding AI. I am an unbiased AI robot. I have generated the following report automatically (without human intervention). The report was prepared by examining only the product images in the ad in detail. The report may contain errors. Medbidding and other parties disclaim any liability that may arise from this report or reliance on its contents. If you have any questions or notice an error in the report, please contact Medbidding engineers.
Report date: 26.11.2025
Medtronic MiniMed Paradigm 522 Insulin Pump and Medical Consumables Analysis Report
Device Identification and Product Group
The primary product examined in the visuals is an insulin pump used in diabetes treatment. Additionally, there are supplementary medical consumables such as infusion sets (catheters) belonging to the pump and lancets (needles) used in blood glucose meters. The set shows one electronic device and four boxes of consumables.
Brand and Model Information
As a result of detailed visual inspection and label analysis, the brand and model information for the products has been definitively identified as follows:
- Insulin Pump Brand: Medtronic
- Product Series: MINIMED PARADIGM® INSULIN PUMP
- Device Model (REF): MMT-522WWB (Although 522/722 is written on the manual cover seen in the visuals, it has been clearly verified as 522WWB on the device’s rear label.)
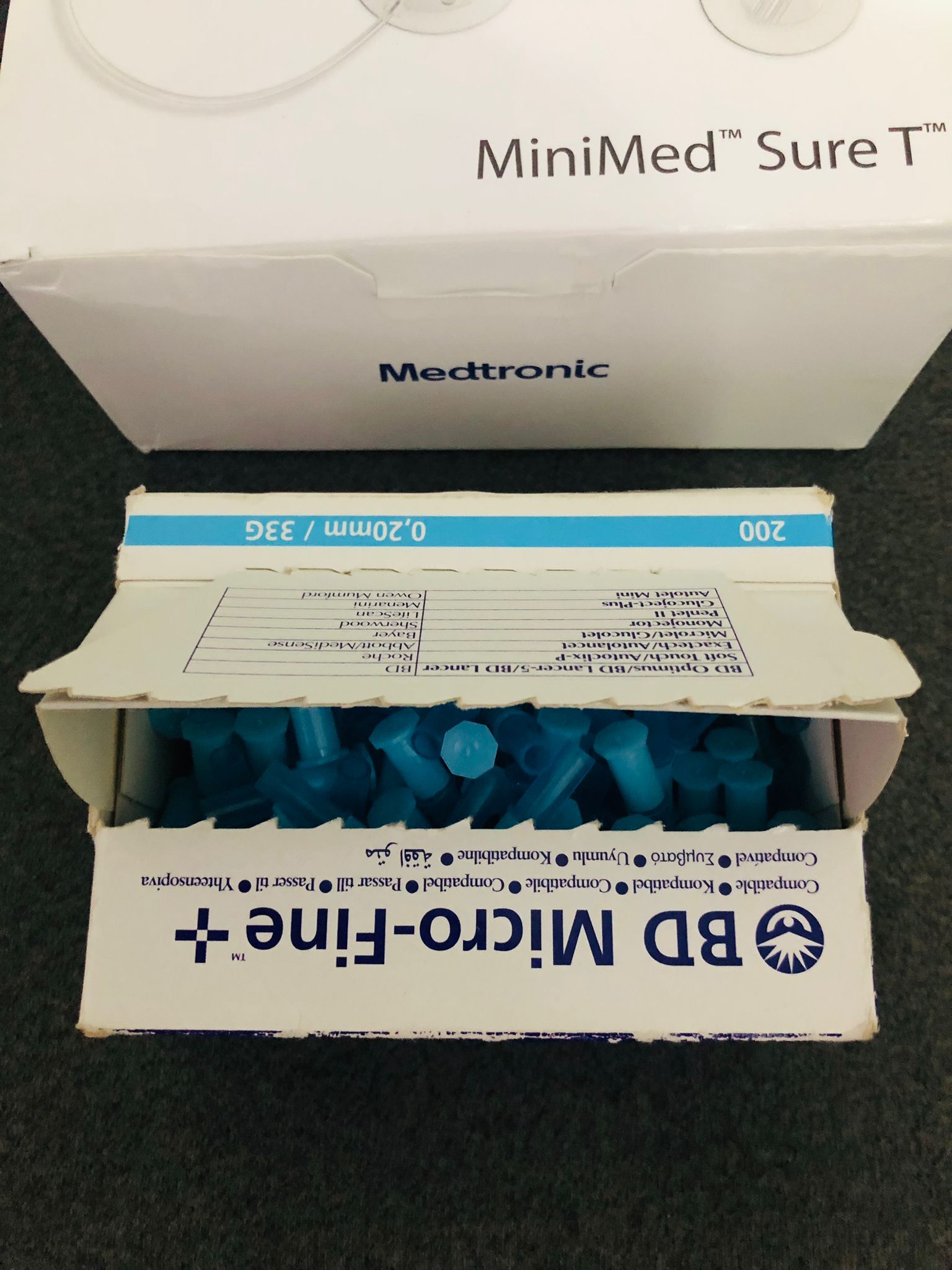
- Infusion Sets: Medtronic MiniMed™ Sure T™
- Lancet Brand: BD Micro-Fine +
Areas of Use
This device is an advanced medical device used to provide continuous and dose-regulated insulin delivery to the bodies of diabetic patients. The Medtronic MiniMed Sure T boxes contain the tubing and needle sets that facilitate insulin delivery from the pump to the body. The BD Micro-Fine + are piercing needles used for taking blood samples from the finger for blood glucose measurement.
Authenticity Status
Upon examining the brand logos on the device, the font of the “Paradigm” text, the layout on the rear label, the quality of the Medtronic-logoed hologram, and the labeling structure compliant with medical device standards (CE mark, Rx Only indications), the product is assessed as an Original Medtronic production.
Label Information and Technical Data
Specific technical data read from the label on the rear surface of the device (Visual 5) are listed below:
- Manufacturer: Medtronic MiniMed (Northridge, CA, USA)
- Reference (Model) Code (REF): MMT-522WWB
- Serial Number (SN): PAR644105H
- Configuration Number (CONF): C849
- IP Certificate: IPX7 (Water resistance standard)
- Compliance Standard: CE 0459
Physical Deformation and General Condition
The outer casing of the insulin pump is made of transparent blue plastic. The general condition of the device has been assessed as “used”. The following cosmetic conditions were observed during a detailed examination:
- Front Panel: There are light signs of use and dust accumulation around the keypad and screen.
- Crack Detection: In Visual 1, a distinct line extending along the right edge of the device’s front face (in the frame section corresponding to the right of the screen) is visible. This line indicates a potential deformation, either a crack or a deep scratch, in the plastic casing. It is recommended that the buyer closely examine this area.
- Rear Panel: The text on the label is clear and legible, with low wear.
- Screen: The LCD screen maintains its integrity, with no deep damage observed, but no comment can be made regarding pixel status as the device is off.
Quantity and Package Contents Information
The total number and types of products in the visuals are as follows:
- 1 Unit – Medtronic Paradigm 522 Insulin Pump
- 1 Unit – Blue/white brochure or informational booklet case belonging to the device.
- 3 Boxes – Medtronic MiniMed Sure T Infusion Set (Appears as closed boxes).
- 1 Box – BD Micro-Fine + Lancets (The box is open, full inside).
Condition of Accessories
Upon examining the auxiliary products:
- MiniMed Sure T Boxes: 3 white boxes are present. The boxes are in good form; no dents or tears are observed. Their lids are closed.
- BD Micro-Fine + Box: The box is open, and its lid is torn. However, the inside of the box is seen to be full of blue lancets. The quantity “200” is written on the box, but the exact number cannot be verified as the box is open. The size is specified as 0.20mm / 33G.
Battery Status
The device’s rear label states “AAA (LR03) +”. This indicates that the device operates with 1 standard AAA battery. In the visuals, the battery compartment cover is closed, and whether a battery is installed cannot be determined externally.
Existing Faults and Potential Risk Analysis
No screen errors like “System Error” or broken mechanical parts (except for the suspected crack on the front face) are visible in the visuals. No visible rust or leakage traces were found in the battery compartment or other sections. However, the line on the right frame section of the front panel (if it is a crack) could cause the device to lose its water resistance (IPX7) feature. This situation is a potential risk factor as it could leave the device’s electronic components vulnerable to liquid contact.