Objective AI Report
Disclaimer: I am Medbidding AI. I am an unbiased AI robot. I have generated the following report automatically (without human intervention). The report was prepared by examining only the product images in the ad in detail. The report may contain errors. Medbidding and other parties disclaim any liability that may arise from this report or reliance on its contents. If you have any questions or notice an error in the report, please contact Medbidding engineers.
Report date: 10.11.2025
Airox Legendäir Ventilator Analysis Report
Device Description and Intended Use
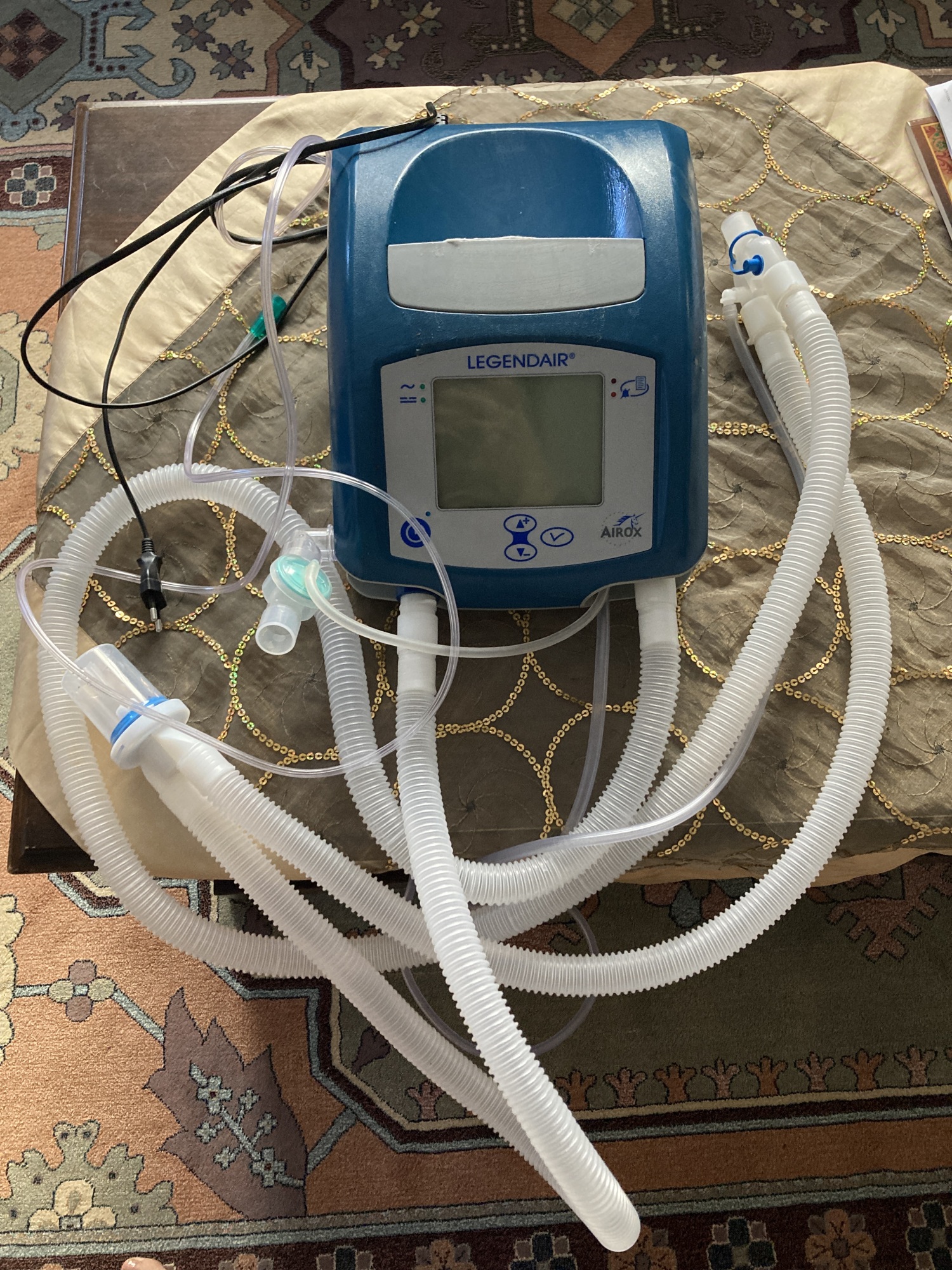
The product in the images has been identified as a Airox brand Legendäir model home mechanical ventilator (respiratory device). Brand and model information is clearly visible on the device’s front panel. Such devices are used to provide non-invasive (via mask) or invasive (via tracheostomy) respiratory support to adult and pediatric patients suffering from respiratory failure. Its portable nature allows for treatment at home or in a hospital environment.
Quantity and Authenticity Information
The following parts were identified during the examination of the images:
- 1 unit Airox Legendäir ventilator device
- 1 unit patient breathing circuit (a set consisting of corrugated tubes and connectors)
- 1 unit black power cable
- 1 unit thin pressure measurement tube (with green connector)
Upon examining the logo, typography, and casing quality on the device, it is understood to be an original product.
General State and Condition
The general condition of the device is assessed as used. Cosmetically, there are signs of use in various places. While small scratches and abrasions are observed on its blue casing, there is no serious damage that would suggest a functional problem in its overall structure. The overall condition of the device is normal for a second-hand medical device.
Physical Assessment
The physical condition of the device has been examined in detail:
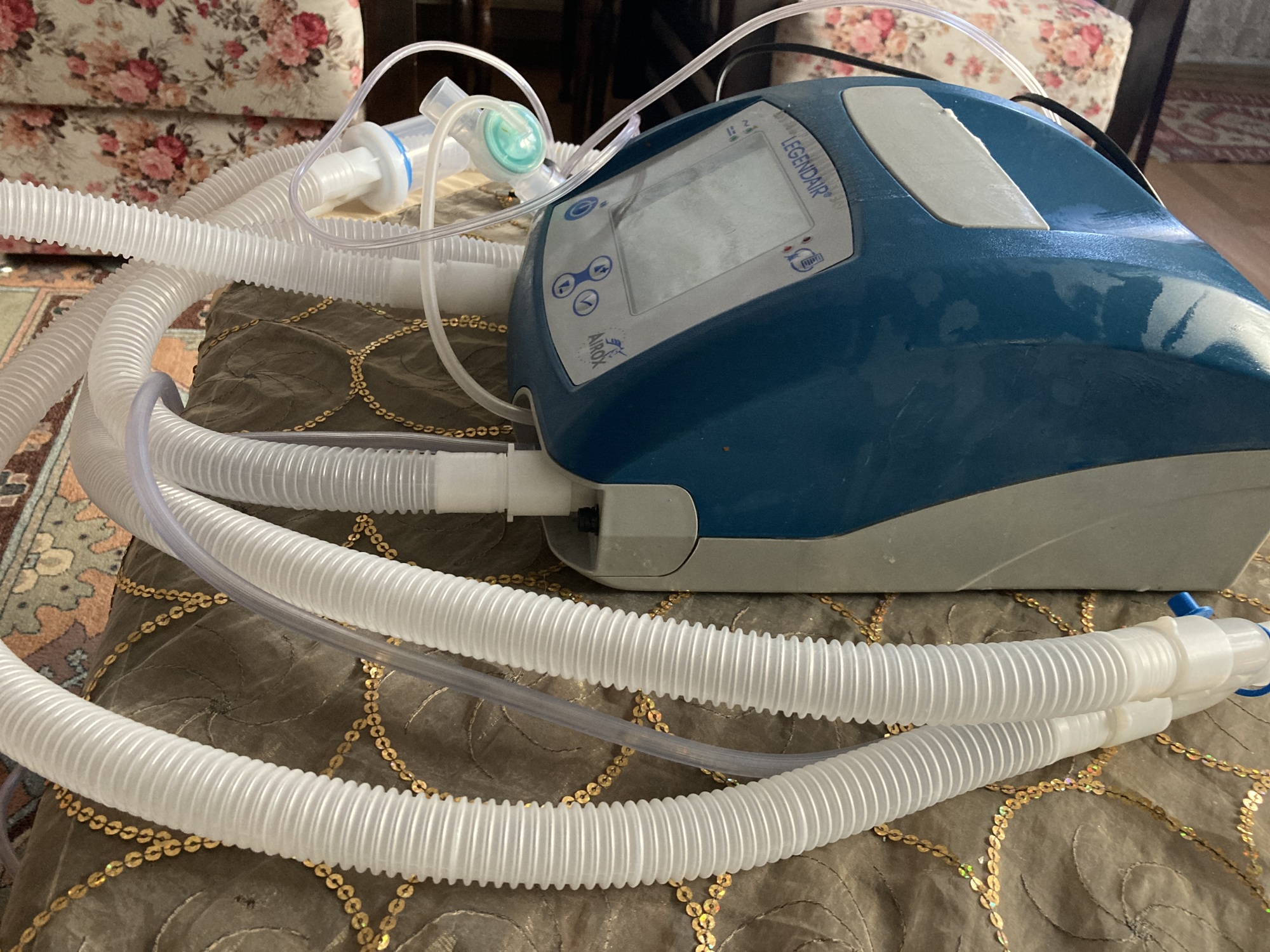
- Casing: There are no prominent cracks or breaks on the device’s blue and gray plastic casing. On the white area of the upper part, there is a mark that appears to be an adhesive residue or surface stain. The ventilation grilles and label area on the rear panel are physically intact.
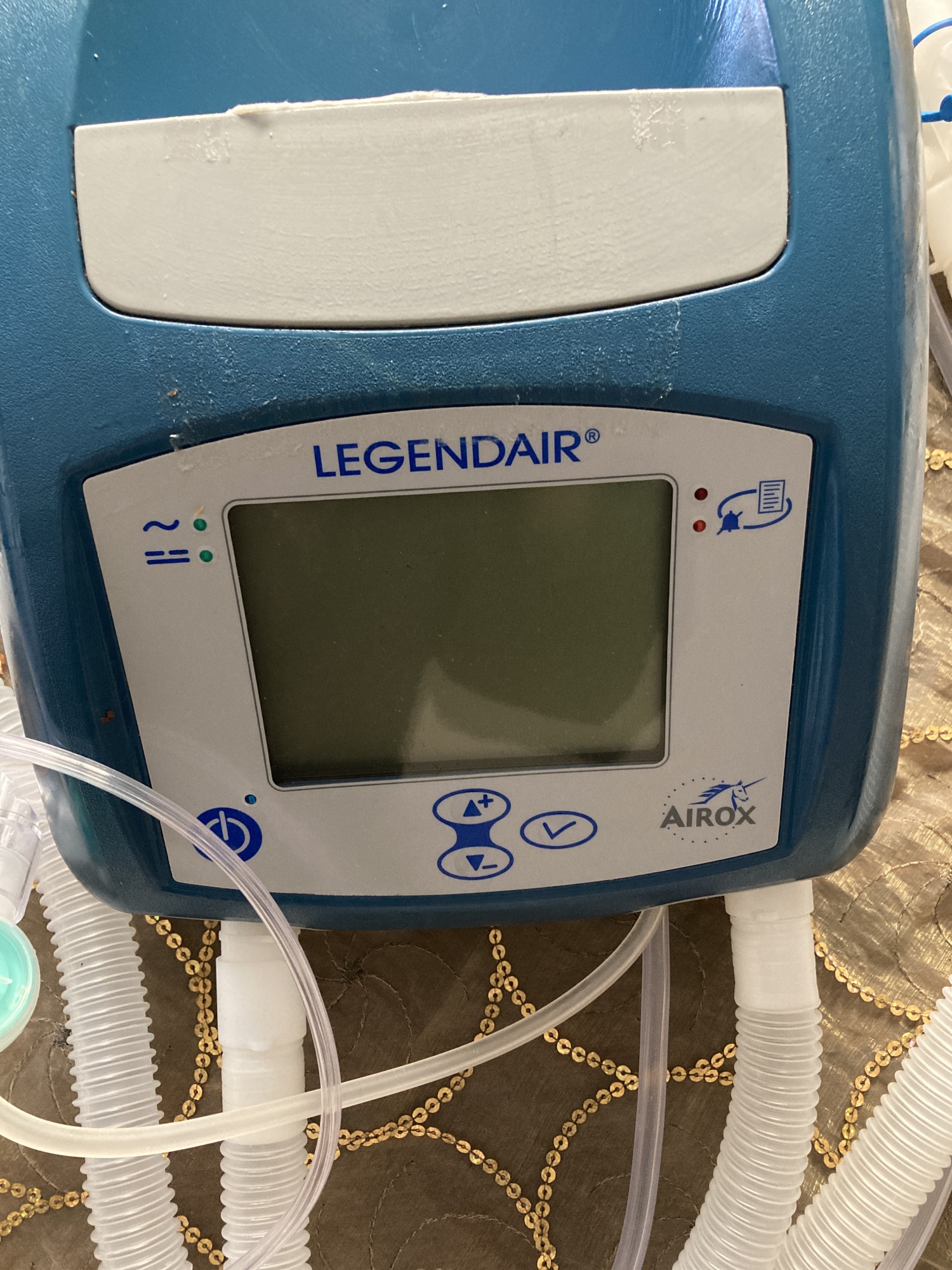
- Screen: The device’s screen is off. Therefore, no comment can be made regarding its operational performance or potential pixel errors. There are no deep scratches or cracks on the screen surface.
- Keypad: No tears or deformations have been observed on the membrane buttons (power, directional keys, confirmation button) on the front panel.
Mechanical and Electronic Components
According to the visual analysis of mechanical and electronic components:
- Connection Ports: The patient circuit outlets on the front panel and the power input (socket), oxygen input, and on/off switch on the rear panel appear physically undamaged. No bending or damage has been detected.
- Patient Circuit: No visible tears or holes are present in the white corrugated tubes and connectors connected to the device.
- Cables: There is no crushing, cuts, or peeling in the insulation of the power cable and the thin cables integrated into the patient circuit.
Since it cannot be determined from the images whether the device is in working condition, no assessment can be made regarding the functionality of the electronic components.
Label and Technical Information
The details of the labels on the device’s rear panel cannot be clearly read in the photos. Therefore, information such as serial number, reference code, or year of manufacture could not be obtained. Only a warning label indicating that the air inlet should not be obstructed (“ENTREE D’AIR NE PAS OBSTRUER / AIR INLET DO NOT OBSTRUCT”) is legible on the rear panel.
Existing and Potential Malfunction Risk
There is no direct evidence in the images (e.g., broken parts, error messages) that the device is faulty. The device is physically well-preserved. However, information regarding the condition of the internal battery, which is an important part of portable ventilators, cannot be obtained from the images. The battery’s performance or lifespan is uncertain. Patient breathing circuits are consumables that need to be replaced at regular intervals for hygiene and safety, and the existing circuit may also need to be checked or replaced before use.