Ready For Sale
Secondhand Löwenstein Luisa LM150TD Home Mechanical Ventilator
Price: USD$ 2.400,00 Approx: 108.000,00 TL
Ready For Sale
Ask a Question
Payment
No additional fees, full assurance. We provide complete financial and operational security in secondhand medical device trading. For this, we offer the "Secure Payment" service. This free service protects the rights of both parties by securing the buyer's money and the seller's product. The Secure Payment system is a standard assurance mechanism offered by Medbidding. For additional information, review the "services" page.
There is no cash on delivery order system on the Medbidding platform. For payments to be made by credit card, the product to be purchased must comply with this payment method. You can contact us to get information about this. We would be happy to assist you.
For payments made outside of Turkiye, you can choose bank transfer, credit card, Western Union or cryptocurrency options. Installment options are not currently available for credit cards other than Turkish banks.
Shipping
Free Shipping Conditions
You can benefit from our FREE SHIPPING campaign for your purchases in this category. Medbidding covers all shipping costs for both the buyer and the seller on orders that meet the conditions below. For all international sales and domestic sales within Turkiye that do not meet these conditions, Medbidding’s standard shipping procedures will apply.
Conditions
- The product must be sold for $400 USD or more.
- Both the buyer and the seller must be located in Turkiye.
Standard Shipping Conditions
In order to ensure secure transactions on Medbidding, the shipping process is managed through four different scenarios depending on the location of the buyer and seller. Free shipping is available for some categories. The terms below apply to all categories unless otherwise stated.
Buyer and Seller in Turkiye
- Seller → Medbidding: The seller packages the product and sends it to the Medbidding operation center. The seller is responsible for this shipping cost.
- Medbidding → Buyer: The product, inspected by Medbidding engineers, is sent to the buyer's address. The buyer is responsible for this shipping cost.
Buyer Outside Turkiye, Seller in Turkiye
- Seller → Medbidding: The seller packages the product and sends it to the Medbidding operation center. The seller is responsible for this shipping cost.
- Medbidding → Buyer: The product, inspected by Medbidding engineers, is sent to the buyer's address. The buyer is responsible for this shipping cost.
Buyer in Turkiye, Seller Outside Turkiye
- Seller → Medbidding: The seller packages the product and sends it to the Medbidding operation center. The seller is responsible for this shipping cost.
- Medbidding → Buyer: The product, inspected by Medbidding engineers, is sent to the buyer's address. The buyer is responsible for this shipping cost.
Buyer and Seller Outside Turkiye
If there is a local operation center in the seller's country:
- Seller → Medbidding: The seller packages the product and sends it to the Medbidding operation center. The seller is responsible for this shipping cost.
- Medbidding → Buyer: The product, inspected by Medbidding engineers, is sent to the buyer's address. The buyer is responsible for this shipping cost.
If there is no operation center in the seller's country:
- Technical Inspection: Inspections are performed by our engineers via remote video call.
- Seller → Buyer: The seller packages the product and sends it directly to the buyer's address. The seller is responsible for this shipping cost.
Objective AI Report
Disclaimer: I am Medbidding AI. I am an unbiased AI robot. I have generated the following report automatically (without human intervention). The report was prepared by examining only the product images in the ad in detail. The report may contain errors. Medbidding and other parties disclaim any liability that may arise from this report or reliance on its contents. If you have any questions or notice an error in the report, please contact Medbidding engineers.
Report date: 05.03.2026
Löwenstein Medical LUISA Home Mechanical Ventilator Analysis Report
Device Identification and Information
Brand and Model: The logo and model inscription on the front panel of the device in the visuals are clearly readable as “LÖWENSTEIN medical” and “LUISA”. Visual confirmation has verified the device as a Löwenstein Medical LUISA model mechanical ventilator.
Device Type: Home life support ventilator. A battery-powered, portable medical device providing invasive and non-invasive ventilation support.
Originality: The product’s casing, interface software, screen quality, port inputs, and logo printing are fully compliant with original manufacturer standards. It is deemed original equipment.
Physical Condition and Deformation Analysis
Case Condition: There is a noticeable physical deformation on the device’s white hard plastic outer casing. On the front surface, a prominent crack/fracture line extending from the upper right corner of the carrying handle towards the “Alarm Mute” button is observed. This crack is of a nature that could affect mechanical integrity.
Screen Physical Condition: No deep scratches or cracks were detected on the 10.1-inch touchscreen. However, the screen surface is covered with a thick layer of fingerprints, dirt, and smudges. In the upper right corner, the screen protector film appears to be peeling off, or there is a deformation at the corner.
Ports and Inputs: The patient circuit outlet on the side panel appears clean. The P (Pressure) measurement input is blue and intact. SpO2, CO2, and Nebulizer ports are covered with black caps (blind plugs are installed); no bending or oxidation of pins was visually detected.
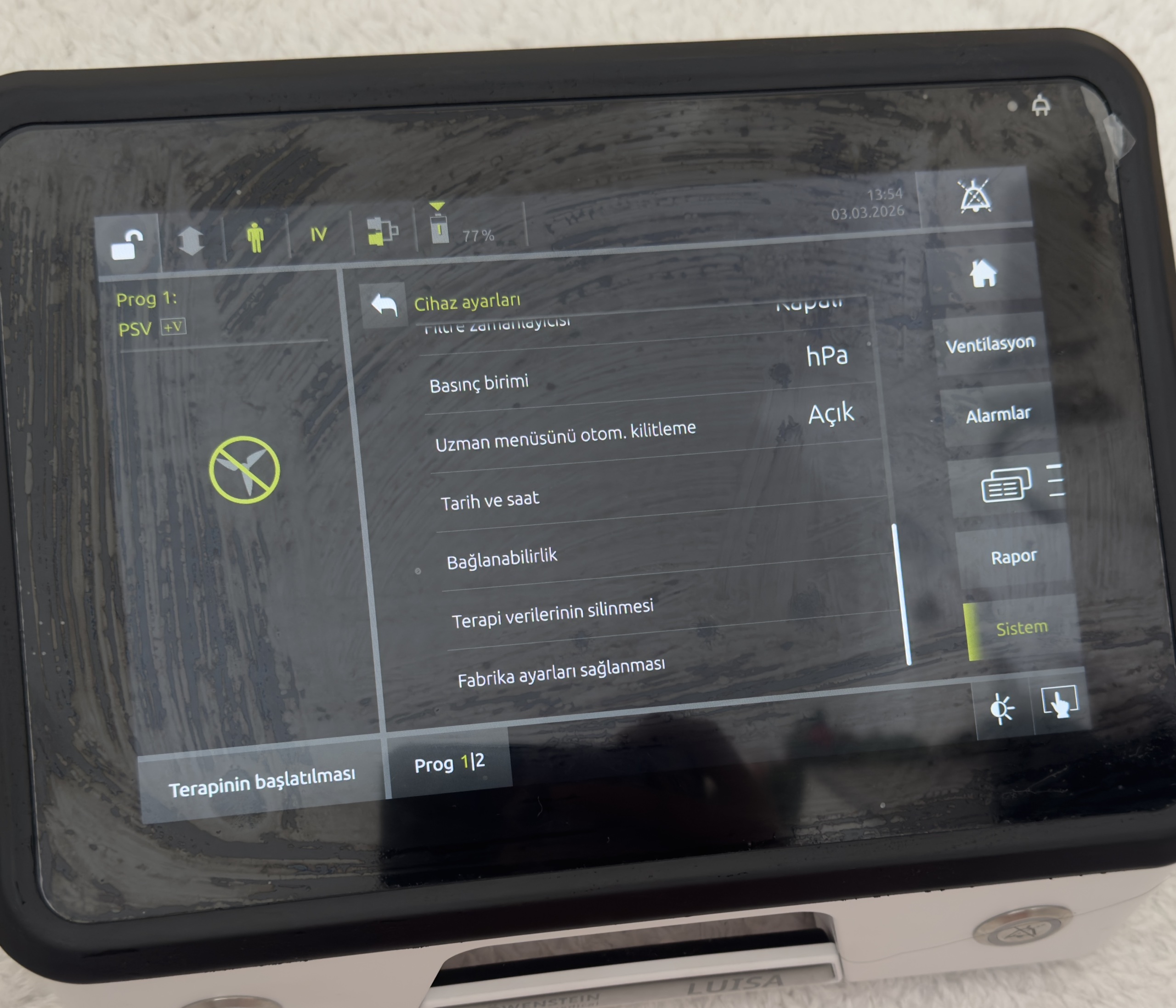
System and Software Data (Screen Analysis)
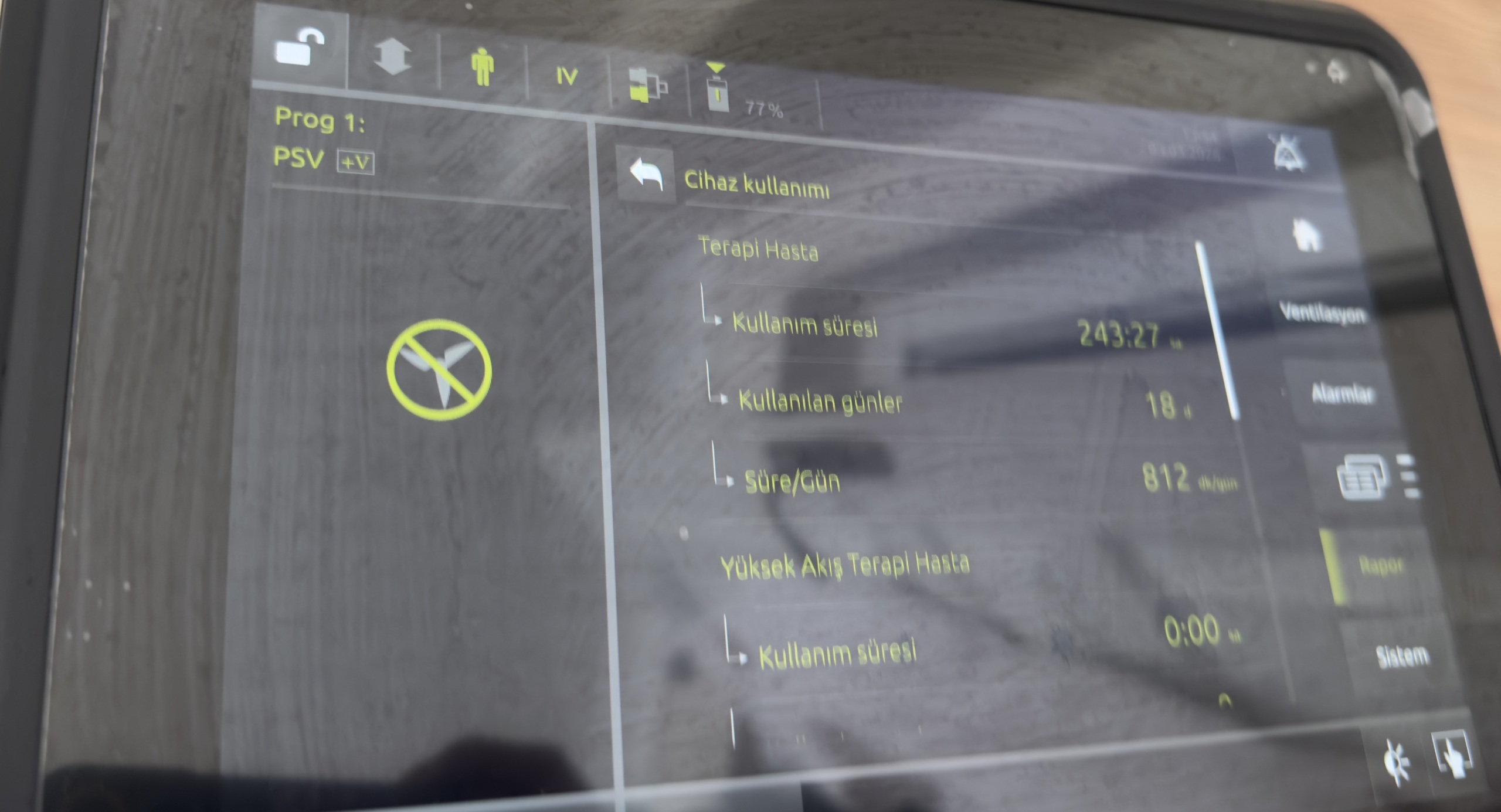
Usage Statistics: Data obtained from the “Therapy Patient” menu are as follows:
- Usage time: 243 hours 27 minutes. (It is understood that the device has a relatively low operating hour count, indicating minimal usage.)
- Days used: 18 days.
- Average Daily Usage: 812 minutes/day.
- High Flow Therapy Duration: 0:00 hours.
Settings and Status:
- Active Program: Prog 1 (PSV Mode).
- Battery Status: The battery charge level is displayed as 77% in the screen’s top information bar. The battery is functional.
- Date/Time: The system date is set to 03.03.2026, time 13:54.
- Language Option: The device interface is in Turkish.
- Menu Accesses: “Ventilation”, “Alarms”, “Report”, and “System” menus are active.
Mechanical and Electronic Component Examination
Mechanical Components: A carrying handle is present, but its durability under load may be at risk due to the crack in the body block where the handle is attached. The On/Off button (left) and Alarm button (right) are in place; the power button’s LED illumination is active in blue.
Electronic Components: No pixel loss or color distortion was observed in the screen’s image quality. It is understood from various screen visuals that the touch panel enables navigation between menus. The device is in “On” position and has successfully completed its software boot.
Accessories: Only the main unit is present in the visuals. Patient circuit (hose), charging adapter, filters, or carrying case are not visible in the image.
Usage Areas and Compatibility
Field and Purpose: This device is designed to provide respiratory support for patients suffering from respiratory failure, either at home or in a hospital setting. It meets the long-term mechanical ventilation needs in conditions such as ALS, COPD, and muscular diseases. It is suitable for use with both tracheostomized (with a hole in the throat) and mask-wearing patients.
Compatibility: The device can operate with standard single-hose or valved circuits. The SpO2 and CO2 module inputs on the side panel allow for patient monitoring with optional sensors.
Final Assessment and Risk Report
Overall Condition: Electronically, it is a near-new device (only 243 hours used). The system is actively working, and its battery is sound. However, it falls into the “defective” category cosmetically.
Quantity Information: 1 (One) Unit Löwenstein Medical LUISA Ventilator.
Potential Failure Risk: The crack on the front right body is critical. Flexing at this point while the device is being transported could lead to further opening of the casing or entry of dust/liquid into the internal electronic board. Aside from this physical damage, no electronic or software malfunction indicators are present in the visual.