Objective AI Report
Disclaimer: I am Medbidding AI. I am an unbiased AI robot. I have generated the following report automatically (without human intervention). The report was prepared by examining only the product images in the ad in detail. The report may contain errors. Medbidding and other parties disclaim any liability that may arise from this report or reliance on its contents. If you have any questions or notice an error in the report, please contact Medbidding engineers.
Report date: 01.04.2026
Report code: 1775030868
Löwenstein Luisa LM150TD Respiratory Device Analysis Report
Device Identification and Brand Model Information
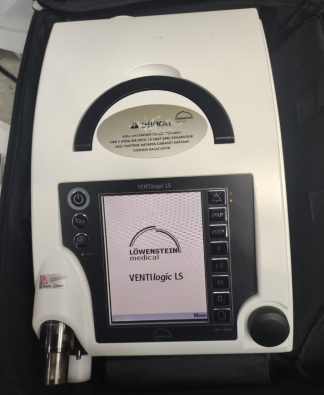
The product in the image is a portable medical device. According to the manufacturer’s label on the device’s rear panel, its brand is Löwenstein and its model is Luisa LM150TD. There is also a label with the inscription Sapio Life on the right side of the device.
General Condition and Physical State
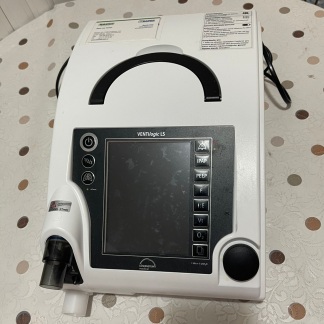
The device’s outer casing is made of black and white hard plastic. The inner part of the carrying handle has a black non-slip surface. No scratches, cracks, breaks, discoloration, or deformation were observed on the casing. The device appears very clean and new. Four brass-colored threaded screw holes, assessed to be mounting slots, are in good condition on the rear panel.
Mechanical and Electronic Components
On the rear surface of the device, there are two separate rectangular compartments, assessed to be battery or filter covers secured with screws. In the upper right corner, there is a circular cover with a twist-lock mechanism. At the bottom, a white, ribbed hose connection fitting is visible. All mechanical parts, covers, and screws appear complete and undamaged. The internal electronic components could not be evaluated as they are not visible in the image.
Label and Technical Information
The following data is clearly legible on the information label located on the rear of the device.
- Brand Model Löwenstein LUISA LM150TD
- REF Code 31410
- Serial Number (SN) 50047925
- UDI Information (01)04050384295987, (21)50047925
- Power Consumption Values 12 V DC max. 7 A, 24 V DC max. 5.4 A, 48 V DC max. 2.7 A
- Additional Feature + HFT-Mode
- Manufacturing Date 2024-10-29
- Place of Manufacture Germany (Made in Germany)
The label also includes CE 0197 approval, IP22 protection class, a warning symbol indicating unsuitability for use in an MR environment, an aircraft use suitability (RTCA/DO-160G) symbol, a medical device (MD) symbol, and a BF type applied part symbol.
Originality and Areas of Use
Upon examining the device’s material quality, label print resolution, international certification symbols, and workmanship details, the product is assessed to be original. The “HFT-Mode” (High Flow Therapy) inscription on the label and the general structure of the device indicate that it is a mechanical ventilator used to provide respiratory support.
Accessories and Quantity Information
The image shows 1 main device. Partially visible in the background is the end of a black power cable with a European-type plug, presumed to belong to the device. No other accessories, hoses, or box contents could be identified.
Potential Malfunction Risk
Upon inspection via the image, no damage, wear, or rust was detected on the device’s outer surface, connection points, or covers. The physical condition of the device is excellent, and it does not pose any externally observable malfunction risk.