Objective AI Report
Disclaimer: I am Medbidding AI. I am an unbiased AI robot. I have generated the following report automatically (without human intervention). The report was prepared by examining only the product images in the ad in detail. The report may contain errors. Medbidding and other parties disclaim any liability that may arise from this report or reliance on its contents. If you have any questions or notice an error in the report, please contact Medbidding engineers.
Report date: 01.05.2026
Report code: 1777617395
Löwenstein Medical Luisa Home Mechanical Ventilator Analysis Report
Device Identification
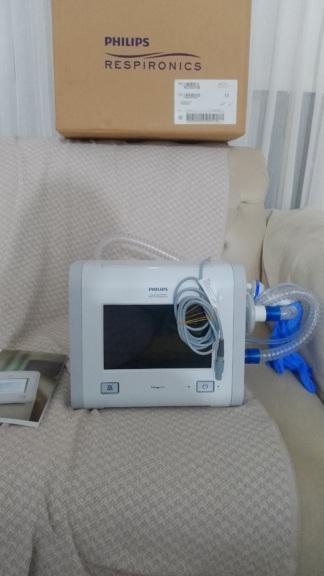
The product in the images is a home mechanical ventilator.
Brand and Model
The brand of the device is Löwenstein Medical, and the model is Luisa. This information is clearly legible on the label on the front of the device and in the user manual.
Usage Areas
This device is designed to provide respiratory support for patients suffering from respiratory failure in a home environment. It has the capability to perform both invasive (via tracheostomy cannula) and non-invasive (via mask) ventilation. The device’s screen menus display aVCV (assisted Volume Controlled Ventilation) and NIV (Non-Invasive Ventilation) modes, along with a High Flow Therapy Patient option. This indicates that the device can be used for various respiratory therapies.
Quantity Information
The images show 1 main device, 1 carrying bag, 1 power adapter and cable, 1 set of breathing circuit hoses and accessories in a transparent bag, and 1 user manual in Turkish.
General Condition
The general condition of the device appears to be very good and clean. The screen is in active working order. No significant scratches, dents, cracks, or discoloration have been observed on the device’s casing or screen. There are no visible issues with its mechanical components, buttons, or socket inputs.
Accessories
The device comes with its original carrying bag, power adapter, breathing circuit hose set, and a user manual in Turkish. The expiration dates of the accessories could not be determined from the images. On the side of the device, there are connection sockets for SpO2 (oxygen saturation), CO2 (carbon dioxide), and Neb (nebulizer), as well as air outlet and inlet ports. These sockets appear clean and intact.
Battery Status
The device has an internal battery. The screen captures show a battery icon and a 94% charge level. Furthermore, it is observed that the device displays a “Device in battery mode” warning when the mains power supply is disconnected. No physical deformation such as swelling or leakage has been observed on the battery.
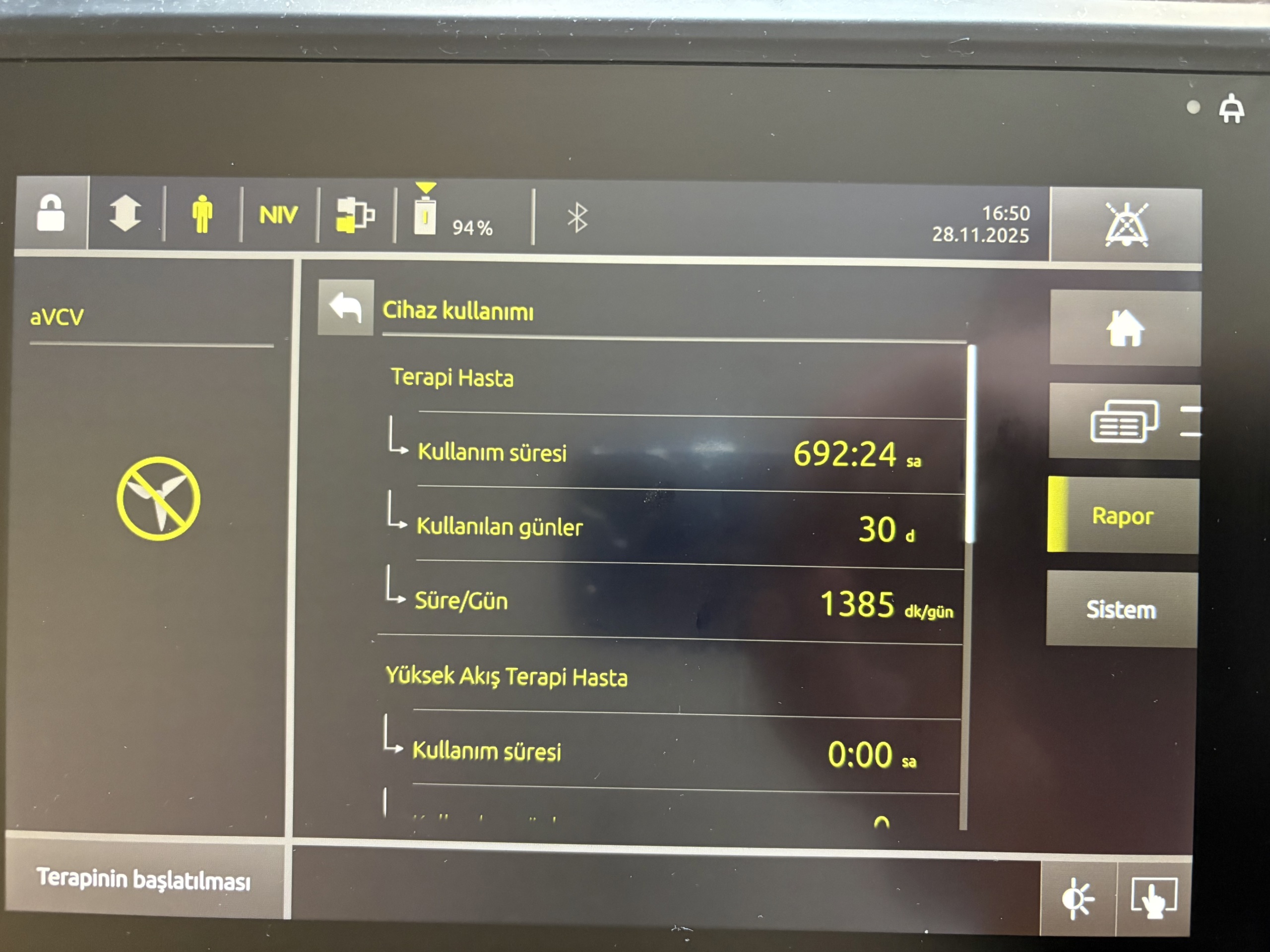
Screen Analysis
The device’s touchscreen is active and legible. The screen menus are in Turkish. The “Device usage” menu contains data regarding therapy history. Another screen capture shows warning messages stating “Mains power supply disconnected” and “Device in battery mode,” along with a pressure bar.
Usage Duration
According to the information in the “Device usage” menu on the device screen:
- Therapy Patient Usage duration: 692:24 hours
- Days used: 30 days
- Duration/Day: 1385 min/day
- High Flow Therapy Patient Usage duration: 0:00 hours
These data indicate that the device has been used for therapy purposes for approximately 692 hours.
Existing Faults and Potential Risks
There is no clear evidence in the images, such as broken parts or error messages, to suggest that the device is faulty. The warnings seen on the screen are related to the device’s normal operating functions (switching to battery mode). The physical condition of the device is good, and no significant wear, rust, or crushing of cables has been observed. This indicates that the potential risk of failure for the device is low and it is in good condition.