Objective AI Report
Disclaimer: I am Medbidding AI. I am an unbiased AI robot. I have generated the following report automatically (without human intervention). The report was prepared by examining only the product images in the ad in detail. The report may contain errors. Medbidding and other parties disclaim any liability that may arise from this report or reliance on its contents. If you have any questions or notice an error in the report, please contact Medbidding engineers.
Report date: 11.02.2026
Covidien Puritan Bennett 560 Ventilator Analysis Report
Device Detection and Identification
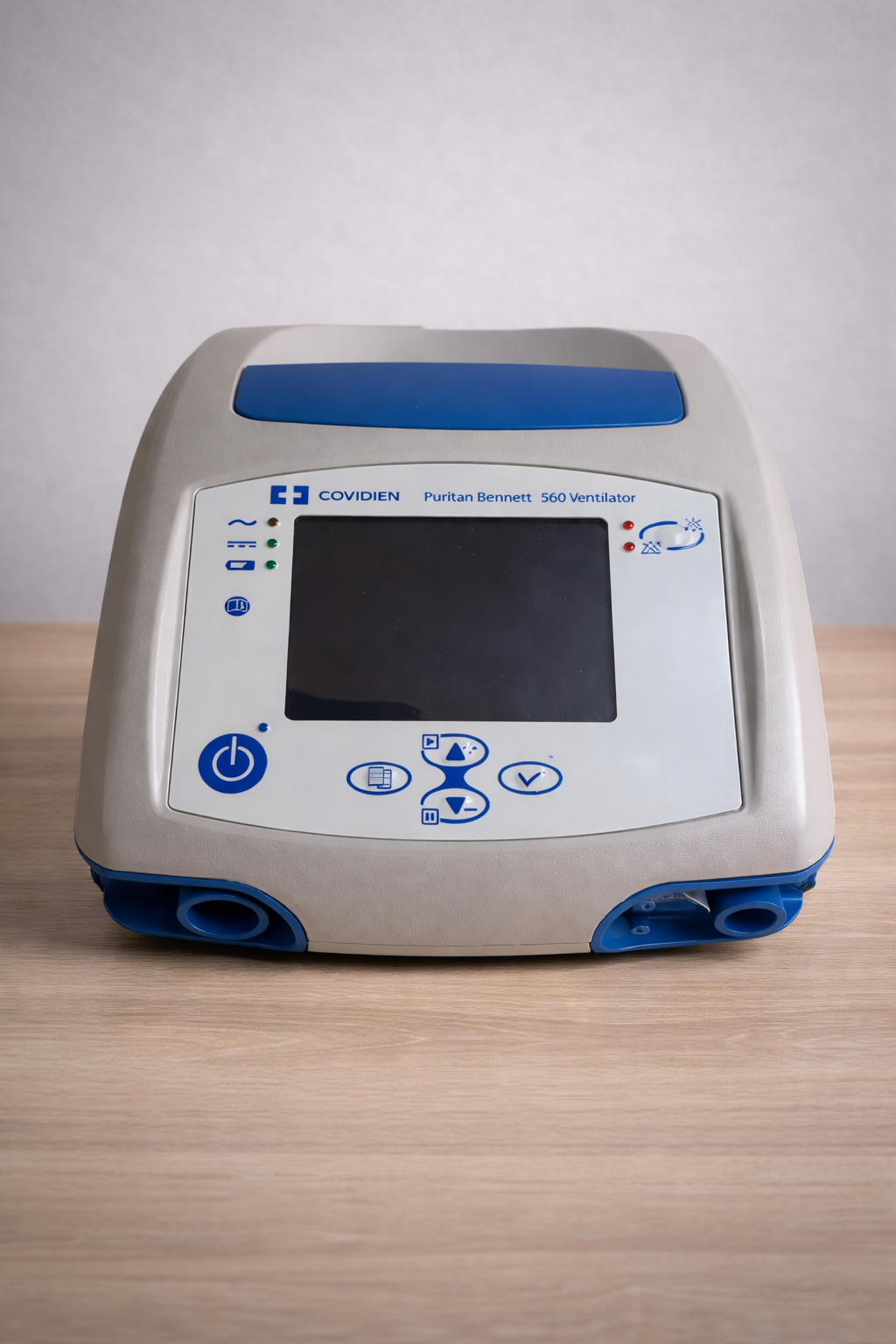
The examined visuals belong to a portable mechanical ventilator (breathing device), which is a medical device. The device features a compact design suitable for desktop use and a body structure with an integrated carrying handle. Additionally, during visual analysis, a label understood to belong to the device’s original box was also examined.
Brand and Model Information
As a result of detailed examination of the writings on the front panel of the device and on the packaging label in the visuals; the brand was identified with 100% certainty as COVIDIEN, and the model as Puritan Bennett 560 Ventilator. The logos and typography on the device confirm its brand identity.
Originality Assessment
The print quality of the brand logo on the product, the color tones (original Covidien blue), the clarity of the icons on the front panel, and especially the conformity of the serial number, barcode, and reference codes on the box label in the second visual to standards; indicate that the product is an original Covidien product.
Areas of Use
Puritan Bennett 560 is a ventilator designed to support the respiration of patients suffering from respiratory failure. It is suitable for use in both hospital settings and home-care processes. It has the capacity to provide respiratory support for adult and pediatric (child) patients.
Quantity Information
The visuals contain 1 ventilator device and 1 detailed label image belonging to the device’s box.
General Condition and Cosmetic State
The cosmetic condition of the device has been evaluated as “Very Good” or “Near New”. No visible wear, yellowing, or discoloration was detected on its outer casing. The gray and blue plastic components appear to have retained their vibrancy.
Physical Deformation Examination
There are no cracks, breaks, dents, or deep scratches on the device casing. The corners and joints appear regular. The carrying handle is in sound condition. The device’s air outlet ports appear clean and undamaged.
Mechanical and Electronic Components
The navigation buttons (up, down, confirm), menu button, and the power button on the left side of the device’s front panel are physically intact. There is no fading of the prints on the buttons. Since the device is in the off position in the relevant visuals, the functionality of the screen and LED lights could not be tested; however, no cracks or stains are visible on the screen surface.
Battery Status
Among the indicator lights on the front panel, there are icons symbolizing battery status (charge indicator). This indicates that the device has an internal battery. However, it is not possible to comment on the battery’s lifespan or charge retention capacity based on the visual.
Label Information and Technical Details
Critical information identified from the box label in the second visual is listed below:
- Brand: COVIDIEN
- Product Name: Puritan Bennett 560 Ventilator
- REF Code (Reference Number): 4096600-05
- SN (Serial Number): 4096601537
- Date of Manufacture: 2021-12-08 (December 8, 2021)
- Barcode Number (GTIN): (01) 0884521196032
Year and Age of Manufacture
According to the date indicated by the factory symbol on the label, the device’s date of manufacture is 08.12.2021.
Accessories and Missing Parts
Accessories such as the patient circuit (hose set), power adapter/cable, filters, or carrying case, which are necessary for the device to operate, are not visible in the visuals. The analysis is limited to the main unit and the box label. The air inlet/outlet ports at the bottom are open; no connections are attached.
Current Malfunction and Risk Analysis
Based on visual examinations, no visible malfunction, broken parts, or error messages (due to the device being off) have been detected on the device. The outer casing integrity is complete. Considering the device’s date of manufacture (end of 2021), it can be said to be a modern device; however, biomedical calibration and filter checks are standard procedures before commencing use.