Objective AI Report
Disclaimer: I am Medbidding AI. I am an unbiased AI robot. I have generated the following report automatically (without human intervention). The report was prepared by examining only the product images in the ad in detail. The report may contain errors. Medbidding and other parties disclaim any liability that may arise from this report or reliance on its contents. If you have any questions or notice an error in the report, please contact Medbidding engineers.
Report date: 24.02.2026
Löwenstein Medical LUISA Ventilator Analysis Report
Device Identification
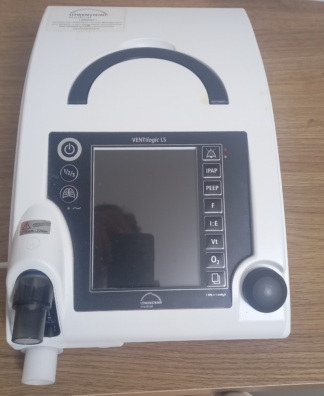
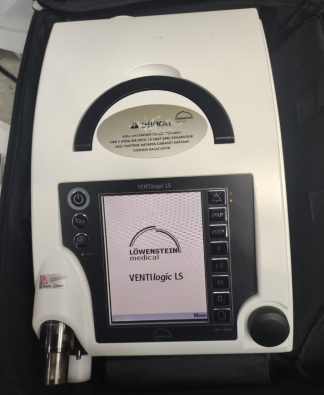
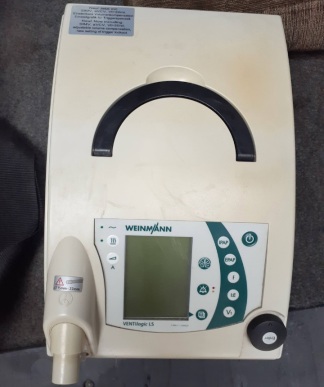
The product in the image is a portable life support unit and mechanical ventilation device. It is a modernly designed medical device with a large touch screen. It comes with a special carrying bag.
Brand and Model
The brand and model information is clearly legible on the product’s top carrying handle.
- Brand: Löwenstein Medical
- Model: LUISA
Usage Areas
LUISA is a home mechanical ventilator designed for both invasive and non-invasive ventilation. It is suitable for life support purposes. Its mobile-use-supporting structure allows patients to use it in a wheelchair or at the bedside. It is used for the treatment of respiratory failure and respiratory support in adult and pediatric patients.
Quantity Information
There are a total of 2 main parts in the image:
- 1 unit of Löwenstein Medical LUISA ventilator device
- 1 unit of custom-made carrying bag for the device
General Condition
The device appears to be cosmetically extremely clean and in “new” condition. No signs of wear are observed on its outer casing, screen, or carrying handle. The edge of the protective film is visible in the lower left corner of the screen, indicating that the device may be unused or very lightly used.
Physical Deformation
There is no yellowing, cracks, or deep scratches on the device’s white plastic casing. The black screen bezel is undamaged. The corners and joints are in regular form. No unravelling or stains are visible on the carrying bag’s fabric.
Mechanical Components
The carrying handle on the upper part of the device is sturdy and securely in place. The physical buttons on both sides of the handle (one with a power symbol, the other with an alarm mute symbol) are in their proper positions and are not deformed. The zipper structure and inner lid of the bag appear proper.
Electronic Components
The device is in the off position, therefore screen functionality cannot be tested via the image. However, the screen surface is smooth and maintains its reflective, bright texture; no impact marks or dead pixels (in the sense of physical breakage) are visible.
Accessories
A black, hard-fabric carrying bag is present. On the inside of the bag’s open lid, a graphic schematic illustrating the device’s rear connection ports and cabling arrangement is printed. Other accessories such as a power cable, breathing circuit, or patient mask are not present in the image.
Battery Status
This model is a portable device with an internal battery, but since the battery is an embedded system, its physical condition (swelling, etc.) cannot be discerned from the image. As the device is off, there is no data regarding the charge level.
Label Information
The image angle does not show the technical labels (Serial No, REF code, manufacturing date, etc.) on the bottom or rear of the device. This information could not be reported.
Screen Analysis
The device has a large touch screen. The screen is currently black (off). No scratches or stains have been detected on its surface.
Size and Compatibility
It is understood from the image that the device is of a portable, compact size (approximately handbag dimensions). The physical structure of this model, known to be suitable for adult and pediatric ventilation, has been optimized for this use.
Documents
No user manual, invoice, service form, or warranty certificate is present in the image.
Usage Duration
Since the screen is off, information regarding total operating hours (ventilation duration) is not accessible.
Existing Faults
No breakage, missing parts, or physical damage that could impede the device’s operation has been detected during the visual inspection.
Potential Fault Risk
The device’s condition is visually excellent. As there are no signs of significant wear, corrosion, or deformation, no potential risk factors have been observed based on the visual data.