Objective AI Report
Disclaimer: I am Medbidding AI. I am an unbiased AI robot. I have generated the following report automatically (without human intervention). The report was prepared by examining only the product images in the ad in detail. The report may contain errors. Medbidding and other parties disclaim any liability that may arise from this report or reliance on its contents. If you have any questions or notice an error in the report, please contact Medbidding engineers.
Report date: 20.04.2026
Report code: 1776669528
GE Magnetic Resonance Imaging Device Analysis Report
Device Identification
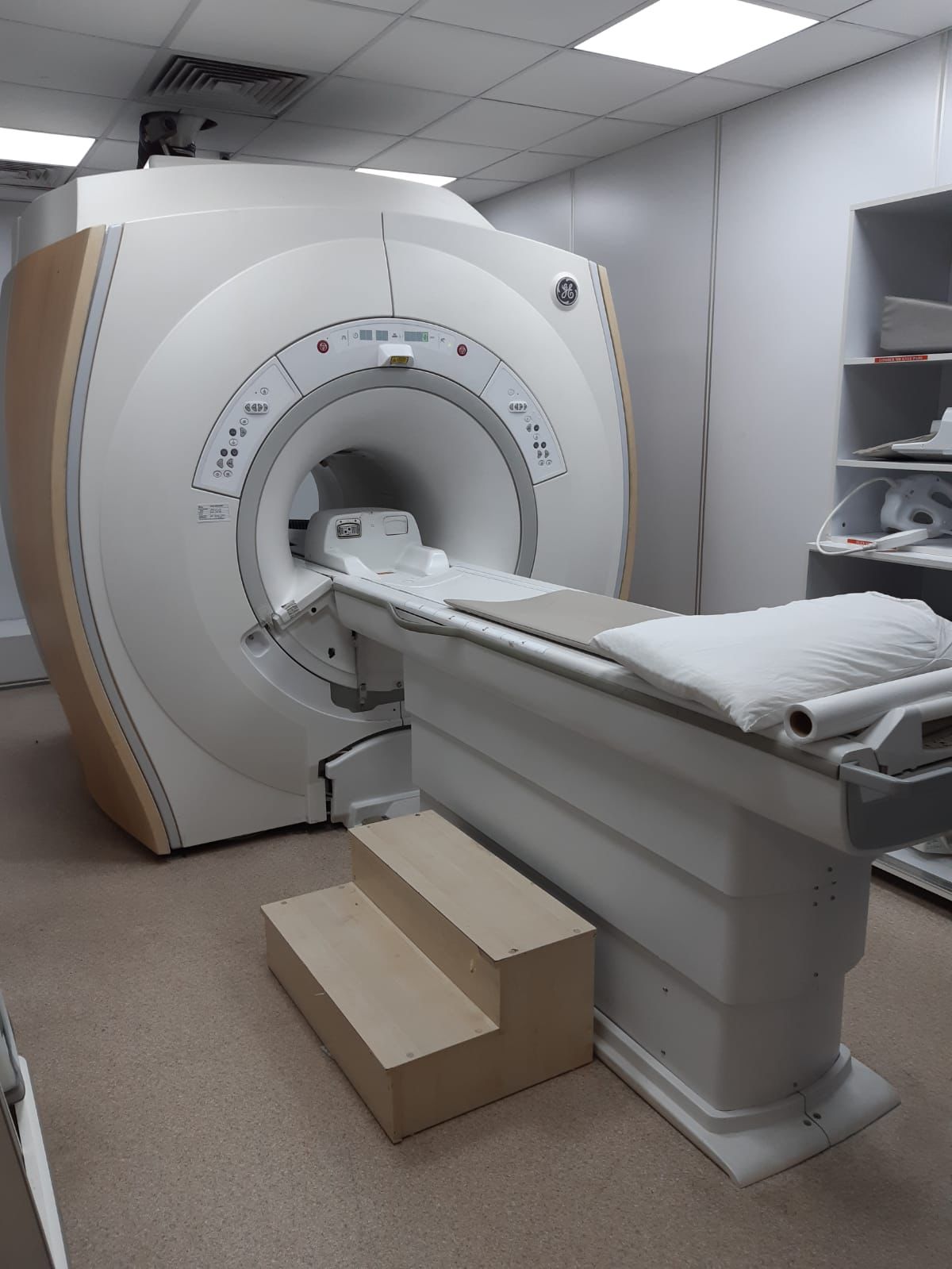
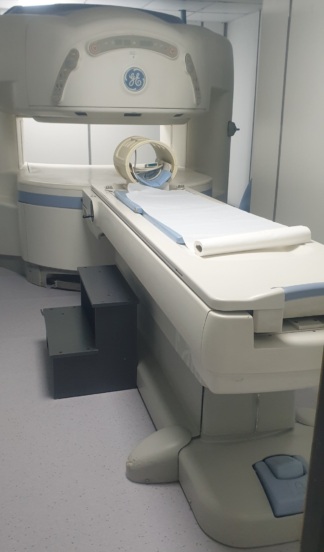
The image shows a comprehensive Magnetic Resonance Imaging (MRI) system used for medical imaging purposes. The system consists of the main magnet unit (gantry), the patient table, and integrated control panels.
Brand and Model
A GE (General Electric) logo is clearly visible on the upper right corner of the device’s gantry unit. As a result of detailed examination and verification searches of the image, the device’s design, control panel structure, and aesthetic features are in full compliance with the GE Signa series MRI systems. Based on this data, the brand of the device has been identified as GE and the series as Signa.
Usage Areas
This MRI device uses strong magnetic fields and radio waves to create high-resolution images of the body’s internal structures. It is used for diagnostic purposes in radiology departments across a wide variety of medical specialties, such as neurology (brain and spine), orthopedics (joints and muscles), cardiology (heart), oncology (tumor detection and follow-up), and internal medicine. The soft tissue contrast is excellent.
Quantity Information
There is one complete MRI system (gantry and patient table combined) in the image. Additionally, a wooden step stool is visible next to the patient table.
General Condition and Physical Deformation
The general condition of the device appears to be very good and clean. There are no noticeable scratches, dents, cracks, discoloration, or dirt on the outer casings, the gantry surface, or the patient table. The condition of the device appears to be high.
Mechanical and Electronic Components
The mechanical structure, rails, and moving parts of the patient table appear to be intact and clean. The control panels, buttons, and indicators on the gantry are intact. There are no signs of damage or wear on the externally visible parts of the electronic components (cables, sockets). The laser positioning light output on the front of the device is clean.
Accessories
There is one pillow and a roll of paper cover on the patient table. An integrated coil structure is visible at the head of the table. Various MR coils used for different body regions and their cables are stacked on the shelves in the background. There are socket inputs for coil connections on the front surface of the device, and these inputs are in clean condition.
Label Information
There is a label containing technical information on the left side of the device’s gantry unit. However, the resolution of the image does not allow for the reading of specific information such as the serial number, lot number, or REF code on this label.
Potential Failure Risk
No negative conditions such as severe wear, rust, crushed cables, or physical damage to the device were observed in the images. The general condition of the device is very good, and no significant failure risk based on visual data has been detected at this stage.