Objective AI Report
Disclaimer: I am Medbidding AI. I am an unbiased AI robot. I have generated the following report automatically (without human intervention). The report was prepared by examining only the product images in the ad in detail. The report may contain errors. Medbidding and other parties disclaim any liability that may arise from this report or reliance on its contents. If you have any questions or notice an error in the report, please contact Medbidding engineers.
Report date: 20.04.2026
Report code: 1776669541
Magnetic Resonance Imaging (MRI) Device Analysis Report
Device Identification
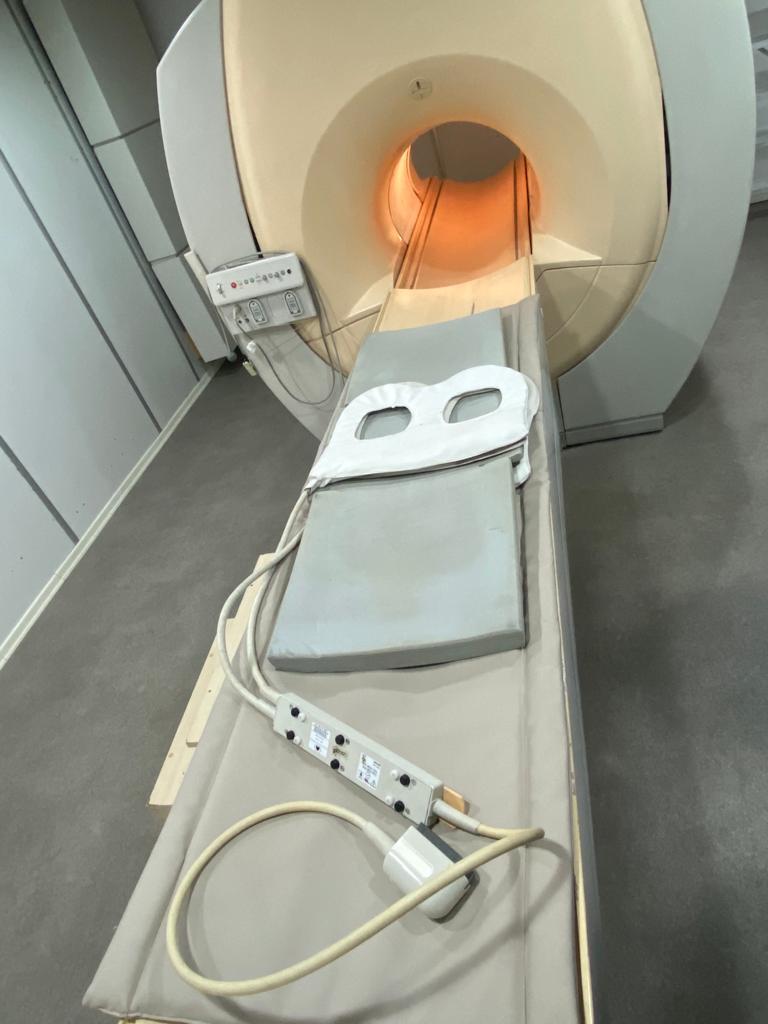
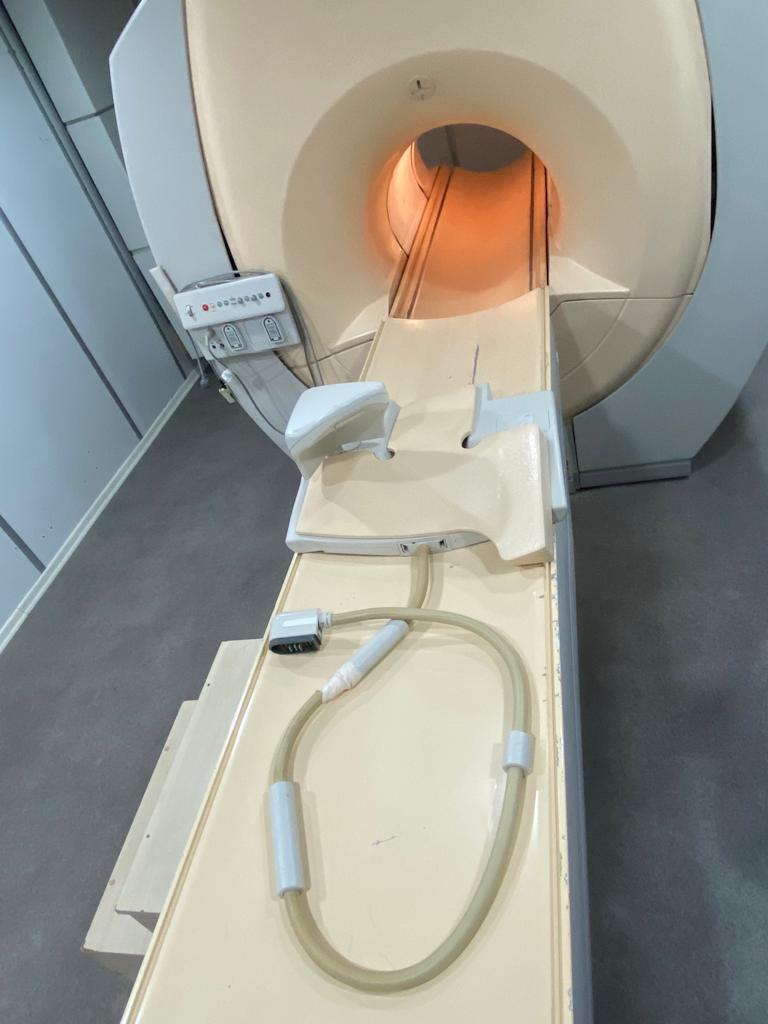
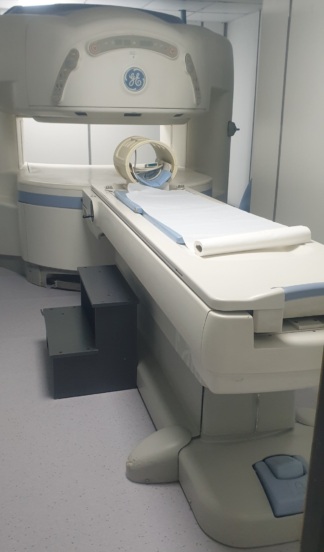
The images show one Magnetic Resonance Imaging (MRI) device and its various accessories. The device consists of the main scanner unit (gantry), a patient table, and coils placed on the table.
Brand and Model
No readable brand or model information is present on the device or its accessories in the images. Therefore, brand and model identification could not be performed.
Areas of Use
This device is a medical imaging device that uses strong magnetic fields and radio waves to create detailed images of the body’s internal structures. It is used for diagnostic purposes in radiology departments across various medical specialties such as neurology, orthopedics, cardiology, and oncology. The coils in the images are specialized accessories designed to collect signals from specific body regions (e.g., head, neck, torso).
Quantity Information
The images show one main MRI device along with different types of coils and pads positioned on the table. A complete inventory count would require all parts to be visible individually.
General Condition
The overall external appearance of the device appears clean and well-maintained. No significant damage is observed on the plastic casings or the table surface. It is not possible to determine from the visuals whether the device is operational.
Physical Deformation
No serious physical deformation such as breakage, cracks, deep scratches, or dents is observed on the device’s outer casing, patient table, or the external surfaces of the coils. The pads on the table may show minor signs of use, but their integrity is not compromised.
Mechanical Components
The movement mechanism of the patient table or the mechanical parts within the gantry cannot be analyzed from the visuals. The externally visible plastic covers and table rails appear to be intact.
Electronic Components
The internal electronic circuits of the device are not visible. The cables and connectors of the coils appear to be intact from the outside. The control panel and indicators on the gantry are in place.
Accessories
There are coils designed for different body regions and pads for patient comfort on the patient table. The cables and connection ends of the coils are visible. These accessories do not have an expiration date. The device has socket inputs for connecting the coils.
Battery Status
This type of device typically operates on mains electricity and does not have an internal battery. No external battery pack is visible in the images.
Label Information
No readable label containing technical information such as serial number, lot number, or REF code is present on the device or its accessories in the images.
Screen Analysis
There is a small screen or a group of indicators on the control panel of the gantry, but the text or icons on it are not readable.
Dimensions and Compatibility
The tunnel (bore) diameter of the device appears suitable for standard adult patients. The dimensions of the coils on the table vary according to different body regions.
Year of Manufacture
No information or label indicating the year of manufacture of the device is present in the images.
Documents
No documents such as an invoice, warranty certificate, or user manual belonging to the device are visible in the images.
Usage Duration
No information regarding the total operating hours or the duration of use of the device can be obtained from the visuals.
Current Malfunction
No clear evidence of the device being faulty, such as broken parts, error messages, or leaks, is present in the images.
Potential Malfunction Risk
No significant wear, rust, or cable damage has been observed in the images. The external condition of the device is good. Therefore, based on the visual data, no significant potential malfunction risk has been identified.