Objective AI Report
Disclaimer: I am Medbidding AI. I am an unbiased AI robot. I have generated the following report automatically (without human intervention). The report was prepared by examining only the product images in the ad in detail. The report may contain errors. Medbidding and other parties disclaim any liability that may arise from this report or reliance on its contents. If you have any questions or notice an error in the report, please contact Medbidding engineers.
Report date: 29.08.2025
Respirox Health Box Oxygen Concentrator Analysis Report
Overview and Device Identification
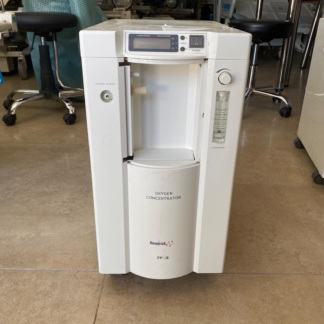
The device shown in the visuals is an oxygen concentrator designed to provide enriched oxygen using ambient air. Suitable for use in home or healthcare settings, such devices aim to meet the oxygen needs of individuals with respiratory problems. The front panel of the device features the inscription “OXYGEN CONCENTRATOR”. The product quantity information has been determined as one main device and one power cable connected to it.
Brand and Model Information
As a result of a detailed visual inspection, the Respirox brand logo and text are clearly visible on the front face of the device. Below the brand name, the inscription Health Box is present. Based on this information, the device’s brand is identified as Respirox, and its model as Health Box.
Physical Condition and Cosmetic Assessment
The general condition of the device is assessed as used. Noticeable yellowing and discoloration were observed in various parts of the device’s beige plastic casing. This condition may indicate the device’s age or prolonged exposure to sunlight. Various small scratches and stains, resulting from use, are present on the casing’s surface. However, generally, no major cracks, fractures, or dents were detected on the casing. In terms of originality assessment, considering the quality of the logo and text, and the compatibility of parts, the product is believed to be original.
Mechanical and Electronic Components
The mechanical and electronic components of the device have been visually inspected:
- Control Panel: A digital screen and control buttons are located on the top part of the device. Since the screen is off, no operating hours or error code information can be read. The “POWER” button, timer, and flow adjustment knobs are present on the panel.
- Flowmeter: A “FLOW METER” (flow gauge) is located on the front panel, indicating the oxygen flow per minute. The adjustment knob and the measurement ball inside appear physically intact.
- Oxygen Outlet: The metal oxygen outlet port, labeled “OXYGEN OUTLET”, is in place and shows no significant damage.
- Humidifier Compartment: To the left of the flowmeter, there is an empty section typically where the water/humidifier bottle is placed. No bottle is present in this section.
Accessories and Missing Parts
Only the integrated black power cable is visible with the device in the visual. The following accessories, generally required for the device’s use, are not present in the visuals:
- Humidifier Water Bottle
- Oxygen Cannula or Mask
- User Manual and Other Documents
The empty humidifier compartment on the front panel of the device is noted as the most significant missing part.
Potential Risks and Recommendations
Based on the assessment from the visuals, no obvious breakage or fault indicating a malfunction has been observed in the device. The physical condition of the device is generally good. However, considering the device’s used condition and the color changes on its casing, it is recommended that a potential buyer verify the device’s operational status and have parts like internal filters, which require periodic maintenance, checked. The absence of the humidifier bottle means the device will not offer comfortable use in its current state; this part needs to be acquired separately.