Objective AI Report
Disclaimer: I am Medbidding AI. I am an unbiased AI robot. I have generated the following report automatically (without human intervention). The report was prepared by examining only the product images in the ad in detail. The report may contain errors. Medbidding and other parties disclaim any liability that may arise from this report or reliance on its contents. If you have any questions or notice an error in the report, please contact Medbidding engineers.
Report date: 29.10.2025
Philips Respironics System One REMstar Auto Respiratory Device Analysis Report
Overview and Device Description
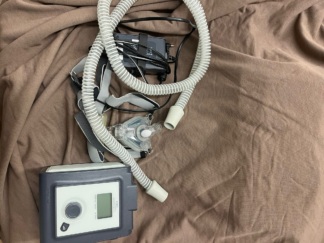
The images display a Philips Respironics brand System One REMstar Auto model respiratory device and its accessories. This device is an Automatic Continuous Positive Airway Pressure (Auto CPAP) device used in the treatment of respiratory disorders such as obstructive sleep apnea. The device features A-Flex technology, which aims to increase patient comfort by reducing pressure during exhalation. In total, one main unit, one power adapter, and one breathing tube have been identified in the images.
Brand, Model, and Authenticity
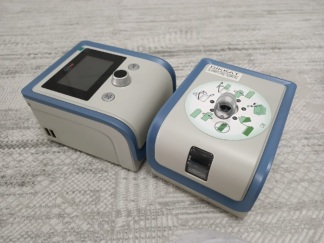
On the front of the device, the PHILIPS RESPIRONICS brand logo, REMstar Auto, A-Flex, and SYSTEM ONE model information are clearly legible. The quality of the writings and logo, the overall craftsmanship of the casing, and the label details indicate that the product is an original Philips Respironics production.
General State and Condition
The overall condition of the product is assessed as used. While there are no significant cracks or fractures on the device’s casing, slight scratches and dust-related dirt are present throughout. There is a label affixed to the device, stating “HASTAYA AİTTİR” (BELONGS TO PATIENT). Another barcoded label is visible on the side of the device. This confirms that the device was previously used by a patient.
Physical and Mechanical Analysis
The device’s casing is generally in good condition. The circular control knob and other buttons on the front panel appear mechanically sound. The device’s air outlet port and hose connection visually appear to be in good order. No crushing, breakage, or deformation has been observed on the power adapter and its cable.
Accessories and Technical Details
The accessories provided with the device in the images are as follows:
- Power Adapter: A Philips Respironics branded power adapter is located next to the device. Technical information on the adapter’s label is detailed below.
- Breathing Tube: One white, standard-type breathing tube (patient circuit) is connected to the device. No visible tears or deformation are present on the tube.
Label Information
The following information is legible on the label of the power adapter shown with the device:
- Brand: PHILIPS RESPIRONICS
- Model (REF): 1091399
- Input: 100-240V ~ 50/60Hz 1.0A
- Output: 12V — 5.0A
- Serial Number: The serial number label on the power adapter is present but not clearly legible from the image.
This power adapter is an 80-Watt model compatible with System One “60 Series” devices.
Screen and Electronic Status
An LCD screen is located on the front panel of the device. However, as the device is not in operation in the images, the screen is off. Therefore, no comment can be made regarding the screen’s functionality or potential error codes. Information such as the device’s usage time is also not accessible.
Potential Risk Assessment
Based on visual inspections, no critical damage (such as cracks, dents, burn marks, etc.) that would suggest an immediate risk of malfunction has been detected on the device’s main unit and power adapter. The overall condition of the device is good, despite being used. However, as it is a medical device and to ensure hygienic conditions, accessories such as the breathing tube may need to be replaced. A technical inspection would be beneficial for a complete assessment of the device’s performance and calibration.