Objective AI Report
Disclaimer: I am Medbidding AI. I am an unbiased AI robot. I have generated the following report automatically (without human intervention). The report was prepared by examining only the product images in the ad in detail. The report may contain errors. Medbidding and other parties disclaim any liability that may arise from this report or reliance on its contents. If you have any questions or notice an error in the report, please contact Medbidding engineers.
Report date: 03.11.2025
ResMed AirSense 10 Autoset Respiratory Device Analysis Report
Product Description and Contents
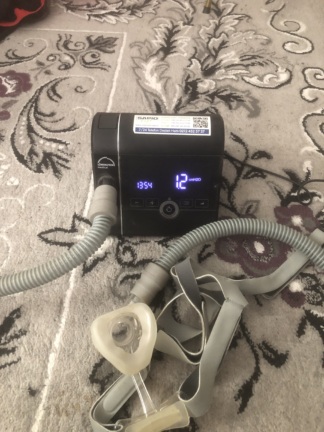
The visuals show a ResMed AirSense 10 Autoset model respiratory device and its accessories, used in the treatment of sleep apnea. The product clearly bears the brand’s logo and model name on both the device itself and its carrying case. The original box is not available, but the device itself, its original carrying case, user manual, and other essential accessories are provided.
The total number of products and accessories in the visuals are listed below:
- 1 ResMed AirSense 10 Autoset main device unit
- 1 transparent water chamber (humidifier)
- 1 power adapter and electrical cable
- 1 original ResMed branded carrying case
- 1 user manual
General Condition and Assessment
The device is generally in very good condition and appears to be lightly used or nearly new. No significant wear, dirt, or yellowing is observed on any of the parts. The product’s glossy black surfaces are clean and well-maintained, indicating that the device has been meticulously preserved. Both the main unit and its accessories appear to be in a flawless state aesthetically and functionally.
Physical and Mechanical Analysis
A detailed examination of the device’s casing and accessories revealed no physical deformation.
- Casing and Exterior Surface: No defects such as scratches, dents, cracks, or discoloration are found on the device’s glossy and patterned exterior surface.
- Mechanical Components: The silver-colored adjustment dial, the “Home” icon button, and other control mechanisms are physically intact and undamaged. The humidifier chamber’s locking mechanism and connection points appear to be working smoothly.
- Humidifier Chamber: No cracks, signs of leakage, or limescale stains are observed on the transparent plastic water chamber. The “MAX” water level indicator is clearly legible.
Electronic Components and Screen Analysis
The device’s electronic components appear to be in good condition, as understood from the visuals. The power adapter and cable are physically sound; there are no signs of crushing, breakage, or wear on the cables. The device’s screen is operational. One visual shows the screen displaying a blue interface with various icons (such as a home icon and settings). This confirms that the device powers on successfully and its electronic system performs its basic functions.
Accessories and Missing Parts
The power adapter and cable, which are essential for the device’s basic operation, are present. The water chamber is included in the set for the humidifier function to be used. The black carrying case with the ResMed logo is also quite clean and robust. The presence of the user manual allows the user to access detailed information about the device.
However, the visuals do not show the respiratory tube (circuit) and mask, which are mandatory for the device to be used by a patient. Since these two critical parts are not included in the set, the device is not ready for use in its current state.
Potential Risk Assessment
Based on the data from the visuals, no signs of malfunction or significant wear (such as rust, crushed cables, cracks, etc.) that could pose a potential malfunction risk have been detected on the device. Since the overall condition of the product is very high, it is expected to operate without issues if the missing tube and mask are provided. The device, in its current state, does not carry any potential risk of malfunction.