Objective AI Report
Disclaimer: I am Medbidding AI. I am an unbiased AI robot. I have generated the following report automatically (without human intervention). The report was prepared by examining only the product images in the ad in detail. The report may contain errors. Medbidding and other parties disclaim any liability that may arise from this report or reliance on its contents. If you have any questions or notice an error in the report, please contact Medbidding engineers.
Report date: 25.10.2025
Resmed Airsense 10 Autoset Respiratory Device Analysis Report
Overview and Device Description
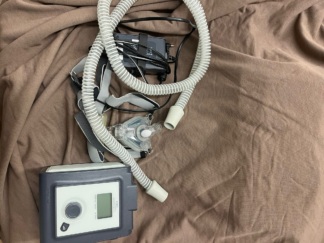
The product shown in the visuals is a Resmed Airsense 10 Autoset model automatic CPAP (Continuous Positive Airway Pressure) device used in the treatment of sleep apnea. The device helps the user breathe regularly during sleep by keeping the airway open. The set consists of the main unit, water chamber, power adapter, respiratory hose, and a nasal mask.
Brand and Model Identification
The Resmed logo and Airsense 10 Autoset model information are clearly visible on the device’s screen and general design. This information ensures the definitive identification of the product’s brand and model.
Quantity and Content Information
A total of 5 pieces of product and accessories are present in the visuals. These are:
- 1 Resmed Airsense 10 Autoset main device unit
- 1 integrated water humidification chamber
- 1 respiratory circuit hose
- 1 nasal mask and headgear
- 1 power adapter and cable
General Condition and Assessment
The general condition of the device, when examined through the visuals, is assessed as lightly used and clean. No significant wear and tear has been observed on the device’s surface or accessories. This indicates that the device has been well-maintained and may have been regularly serviced.
Physical and Mechanical Analysis
Upon inspection of the product’s casing, screen, buttons, and other plastic components, no scratches, cracks, dents, or discoloration have been detected. The humidification unit appears transparent and clean. The respiratory hose and mask are also visually in good condition, with no significant deformation or yellowing. The rotary knob and other control buttons on the device appear physically sound.
Electronic and Screen Analysis
The device’s screen is active, showing menu options such as “My Options” and “Sleep Report”. The screen display is clear, with no pixel errors or signs of malfunction. No crushing, breakage, or damage has been observed on the power adapter and cables located next to the device.
Label Information
A warning text beginning with “CAUTION” is present on the white label on the device. However, the content of the label cannot be fully read due to the resolution and angle in the visual. Therefore, information regarding the serial number or other technical codes cannot be provided.
Potential Malfunction Risk
Based on the examinations conducted in the visuals, no serious wear, rust, crushed cables, or broken parts that could pose a potential malfunction risk have been observed on the device. The device’s overall condition supports long-term and trouble-free use.