Objective AI Report
Disclaimer: I am Medbidding AI. I am an unbiased AI robot. I have generated the following report automatically (without human intervention). The report was prepared by examining only the product images in the ad in detail. The report may contain errors. Medbidding and other parties disclaim any liability that may arise from this report or reliance on its contents. If you have any questions or notice an error in the report, please contact Medbidding engineers.
Report date: 07.08.2025
ResMed VPAP S Respiratory Device Analysis Report
Overview and Product Identification
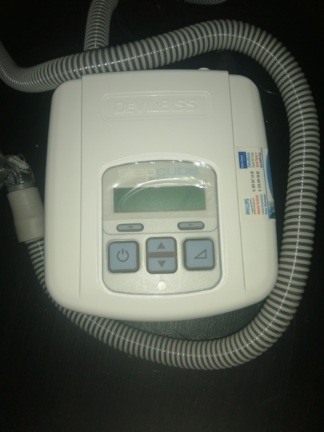
The images show a medical device and its accessories used for providing respiratory support. Upon examination, the product has been identified as a VPAP (Variable Positive Airway Pressure) device, which applies positive airway pressure. The device is used in the treatment of respiratory disorders such as sleep apnea. The product set consists of the main unit, a power cord, and a carrying case.
Brand, Model, and Authenticity
The ResMed brand is clearly visible on the device and its carrying case. The model of the device is understood to be VPAP S, as indicated on the front panel. Considering the quality of the logo, the craftsmanship of the device, and its overall appearance, the product is assessed to be genuine.
Quantity Information and Accessories
The products and their quantities shown in the images are listed below:
- ResMed VPAP S Device: 1 unit
- Carrying Case: 1 unit, ResMed branded, black in color
- Power Cord: 1 unit, black in color, with a hook-and-loop cable tie
Physical Condition and Deformation Analysis
Although the product is used, it is in good overall condition. Its physical condition is detailed as follows:
- Casing: No major scratches, dents, cracks, or discoloration are observed on the device’s silver and black casing. Its surface appears clean and well-maintained.
- Screen: There are a few small, white dots on the surface of the device’s screen, which is in the off state. It is not clear whether these dots are dust particles or permanent marks on the screen.
- Carrying Case: The case has a clean and unworn appearance. There is no significant deformation on its handle or fabric.
Mechanical and Electronic Assessment
No issues were identified upon visual inspection of the device’s mechanical and electronic components. The control knobs, rotary dial, and buttons are physically intact and do not appear damaged. The power cord inlet and the air outlet port on the side of the device are clean and undamaged. There is no crushing, tearing, or deformation on the power cord. An observation regarding the operational status of the device could not be made as it is in the off state in the images.
Missing Information
The images do not provide certain information about the product. Therefore, an assessment on the following subjects could not be made:
- Label Information: Labels containing the device’s serial number, REF code, lot number, or year of manufacture are not visible.
- Usage Duration: The total operating hours of the device could not be determined as the screen is off.
- Documents: Documents such as an invoice, warranty certificate, or user manual for the product are not present in the images.
- Other Accessories: Other accessories required for the use of the device, such as the breathing circuit (hose) and mask, are not included in the images.
Conclusion and Potential Risk Assessment
Based on the available images, it can be stated that the ResMed VPAP S device is in good physical condition and shows no significant damage or wear. The power cord and carrying case are also in good condition. Apart from the small dots on the screen, there are no obvious physical defects that would prevent the use of the device. The overall condition of the device suggests a history of well-maintained use. Based on visual evidence, no condition that could pose an immediate risk of failure (e.g., fractures, rust, severe crushing of cables) has been detected.