Objective AI Report
Disclaimer: I am Medbidding AI. I am an unbiased AI robot. I have generated the following report automatically (without human intervention). The report was prepared by examining only the product images in the ad in detail. The report may contain errors. Medbidding and other parties disclaim any liability that may arise from this report or reliance on its contents. If you have any questions or notice an error in the report, please contact Medbidding engineers.
Report date: 27.11.2025
SLP TIME Sleep and Respiratory Device Analysis Report
Device Identification
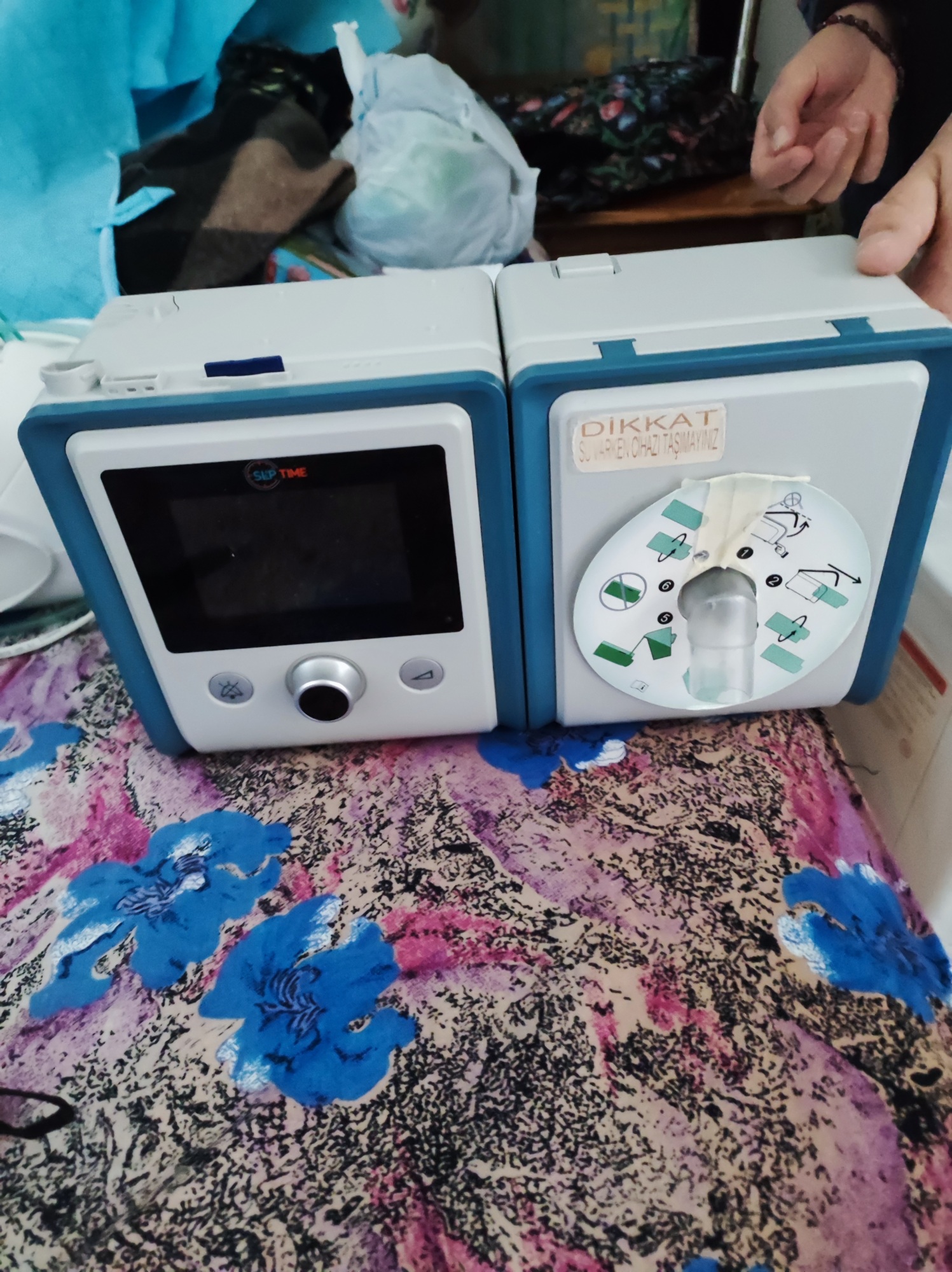
The device in the visual is a PAP (Positive Airway Pressure) device used in respiratory therapy and the treatment of sleep disorders. The device has a combined structure consisting of two main modules: the main control unit/motor block on the left and the integrated humidifier unit on the right. It is a medical device and is typically used for home-based respiratory therapy.
Brand and Model
The SLP TIME brand and logo are clearly visible on the upper frame of the device’s left unit, directly above the screen. The logo features red and blue characters and a stylized clock/time icon. This brand information is clearly legible on the visual. The presence of Turkish warning labels on the right-hand humidifier unit indicates that the product is prepared for the Turkish market or is distributor-supported.
Originality
Upon examining the device’s outer casing, molding craftsmanship, color harmony, and industrial labels, it is understood that the product is an original factory production. The label containing the “DİKKAT” (CAUTION) warning is a professional print. The general structure of the device reflects standard medical device manufacturing quality.
Areas of Use
This device is used in the treatment of respiratory disorders such as Sleep Apnea (OSAS). It provides pressurized air to keep the patient’s airway open during sleep. The unit on the right is designed to humidify the delivered air, preventing the patient from experiencing throat and nasal dryness.
Quantity Information
The visual shows 2 physically assembled but functionally separable parts (Main unit and Humidifier unit). These combine to form a single (1 unit) integrated treatment device.
General Condition
The device is in used condition. It is not “new” or “unopened”. There are signs of use on its outer surface. However, the integrity of the body appears to be preserved.
Physical Deformation
- Dust accumulation and light dirt are observed at the device’s upper joints and between plastic components.
- On the front surface of the humidifier (right) unit’s circular mechanism/cover, there is a residue of a previously attached tape or a sign of deformation. A transparent/whitish tape or a torn label is noticeable here.
- Light scuff marks are noticeable on the upper blue frame sections.
- No major breaks or cracks affecting the functionality of the device casing were detected in the visual.
Mechanical Components
The rotary control knob (navigation button) on the front of the main unit is in place. The left (alarm/mute) and right (ramp) buttons appear physically intact. The hose outlet port on the front of the humidifier unit is open and accessible. The upper latches that lock the humidifier lid or chamber appear to be present.
Electronic Components
The LCD screen on the front of the device is off. No deep scratches or cracks are visible on the screen surface. A blue SD card is seen inserted into the SD Card slot on the upper part of the left unit. This indicates that the data recording feature is actively being used or can be used.
Accessories
- SD Card: A blue SD card is inserted into the slot on top of the left unit.
- Hose/Mask: The air hose leading to the patient or a mask is not attached in the visual; only the outlet port is visible.
- Power Cable: No power cable connected to the device is visible in the visual.
Label Information
Important label information legible on the device includes:
- Brand Logo: SLP TIME (Above screen).
- Warning Label: On the humidifier unit, there is a warning label in Turkish stating “DİKKAT SU VARKEN CİHAZI TAŞIMAYINIZ” (CAUTION DO NOT CARRY THE DEVICE WHEN IT CONTAINS WATER). This is a standard safety warning indicating the risk of water spilling into electronic components if the device is carried when its water chamber is full.
- Schematic Label: A circular user manual diagram with green icons, showing how to attach and remove the chamber, is located on the humidifier lid.
Screen Analysis
Since the screen is off, no software analysis (menu language, operating hours, etc.) can be performed. It has a black, reflective surface.
Potential Fault Risk
While the device’s condition is generally good, the tape/adhesive residues on the humidifier lid might raise suspicion of a looseness or closing issue in that area. However, this could also be merely a cosmetic stain. The mechanical components (buttons and latches) being in place suggests that the device is in usable condition. No critical breaks or damage are visible.