Objective AI Report
Disclaimer: I am Medbidding AI. I am an unbiased AI robot. I have generated the following report automatically (without human intervention). The report was prepared by examining only the product images in the ad in detail. The report may contain errors. Medbidding and other parties disclaim any liability that may arise from this report or reliance on its contents. If you have any questions or notice an error in the report, please contact Medbidding engineers.
Report date: 14.02.2026
Löwenstein Medical prisma25ST and prismaAQUA Device Analysis Report
Device Detection and Identification
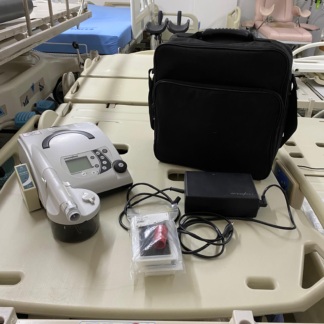
The device in the image is a respiratory therapy device with an integrated humidifier unit. It consists of two main modules: the main unit on the right controls treatment data and provides airflow, while a water reservoir module on the left, which heats and humidifies the air, is integrated. The device belongs to the PAP (Positive Airway Pressure) group, commonly used in the treatment of sleep apnea or respiratory failure.
Brand and Model
As a result of a detailed examination of the image, the text and logos on the device are clearly legible. The brand and model identification are as follows:
- Main Brand: LÖWENSTEIN Medical (Also known as Weinmann formerly, but LÖWENSTEIN is clearly written on the device).
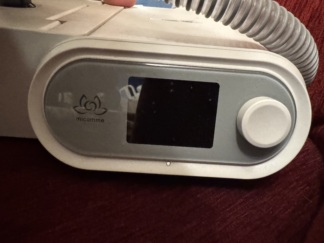
- Device Model: The inscription prisma25ST is clearly visible around the power button of the main control unit. This model is typically a BiLevel (BPAP) device with Spontaneous/Timed (S/T) modes.
- Humidifier Model: The inscription prismaAQUA is located on the humidifier module integrated into the left side of the device.
Originality
The font structure of the logo on the product, the placement of model designations, case quality, and industrial design (the characteristic black casing and vertical screen structure of the Löwenstein prisma series) confirm that the product is an original Löwenstein Medical device.
Areas of Use
The Löwenstein Medical prisma25ST model is used in the treatment of patients suffering from obstructive sleep apnea, complex sleep apnea, or respiratory failure. The “ST” designation indicates that the device supports both spontaneous breathing and activates in a timed manner if the patient’s breathing stops (Spontaneous/Timed). The prismaAQUA humidifies the inhaled air during this treatment to prevent the patient’s airways from drying out.
Quantity Information
The following parts have been identified in the image:
- 1 prisma25ST Main Device
- 1 prismaAQUA Humidifier Unit (Attached to the main device)
- 1 Respiratory Hose (Circuit)
General Condition and Physical Deformation
The general cosmetic condition of the device has been evaluated as “Used”. Noteworthy details regarding its physical condition are as follows:
- Cosmetic Deformation: On the front surface of the device, particularly on the glossy panel where the brand logo and hose outlet are located, a protective gelatin or sticker layer is observed. However, this layer is quite worn, has bubbled in places, formed air bubbles, and started to peel at the edges. This situation indicates that the device’s original protective gelatin was either used for a long time without being removed, or a subsequently applied protector has deformed. This appearance creates an aesthetically unclean impression.
- Case Cleanliness: General dustiness is present on the casing. No deep damage such as cracks, breaks, or dents could be detected from the image.
- Hose Connection: The hose’s entry point into the device appears sturdy, but the sticker deformation around the connection point indicates that the area is frequently touched.
Mechanical and Electronic Components
Mechanical Components: The humidifier unit (left part) and the main unit (right part) are perfectly fitted together. No gap is visible between the detachable parts. No externally visible tears or dents were detected in the corrugated structure of the respiratory hose.
Electronic Components: The device’s power button (bottom right) appears physically intact. Since the screen is currently off (black), it could not be analyzed whether there are any dead pixels or screen malfunctions.
Accessories
The image shows 1 grey respiratory hose connected to the device. The cuff (adapter) connecting the hose to the device appears sturdy. The image frame does not include a power adapter (power supply), carrying bag, or face mask.
Label and Technical Information
The information legible in the image is limited to the front face of the device:
- Brand: LÖWENSTEIN medical
- Model: prisma25ST and prismaAQUA
- Specific information such as Serial Number (SN), REF code, or Lot number is usually located on the bottom or back of the device, so it is not visible from this angle.
Potential Malfunction Risk and Evaluation
The most prominent flaw of the device is the excessive deformation of the film/coating on the front panel. If this is a factory-applied protective film and has been exposed to heat/humidity for a long time, it might have adhered to the surface or left marks; however, this is not a technical malfunction that would affect the device’s operation. No critical risks that would impede operation, such as broken parts, corrosion, or detached cables, are visible in the image. The product’s condition is rated as “Medium/Good,” and it can be said that it only requires detailed external cleaning.