Ready For Sale
Secondhand Resvent iBreeze 30ST BPAP ST Device with Humidifier
Price: USD$ 240,00 Approx: 10.800,00 TL
Ready For Sale
Ask a Question
Payment
No additional fees, full assurance. We provide complete financial and operational security in secondhand medical device trading. For this, we offer the "Secure Payment" service. This free service protects the rights of both parties by securing the buyer's money and the seller's product. The Secure Payment system is a standard assurance mechanism offered by Medbidding. For additional information, review the "services" page.
There is no cash on delivery order system on the Medbidding platform. For payments to be made by credit card, the product to be purchased must comply with this payment method. You can contact us to get information about this. We would be happy to assist you.
For payments made outside of Turkiye, you can choose bank transfer, credit card, Western Union or cryptocurrency options. Installment options are not currently available for credit cards other than Turkish banks.
Shipping
Free Shipping Conditions
You can benefit from our FREE SHIPPING campaign for your purchases in this category. Medbidding covers all shipping costs for both the buyer and the seller on orders that meet the conditions below. For all international sales and domestic sales within Turkiye that do not meet these conditions, Medbidding’s standard shipping procedures will apply.
Conditions
- The product must be sold for $250 USD or more.
- Both the buyer and the seller must be located in Turkiye.
Standard Shipping Conditions
In order to ensure secure transactions on Medbidding, the shipping process is managed through four different scenarios depending on the location of the buyer and seller. Free shipping is available for some categories. The terms below apply to all categories unless otherwise stated.
Buyer and Seller in Turkiye
- Seller → Medbidding: The seller packages the product and sends it to the Medbidding operation center. The seller is responsible for this shipping cost.
- Medbidding → Buyer: The product, inspected by Medbidding engineers, is sent to the buyer's address. The buyer is responsible for this shipping cost.
Buyer Outside Turkiye, Seller in Turkiye
- Seller → Medbidding: The seller packages the product and sends it to the Medbidding operation center. The seller is responsible for this shipping cost.
- Medbidding → Buyer: The product, inspected by Medbidding engineers, is sent to the buyer's address. The buyer is responsible for this shipping cost.
Buyer in Turkiye, Seller Outside Turkiye
- Seller → Medbidding: The seller packages the product and sends it to the Medbidding operation center. The seller is responsible for this shipping cost.
- Medbidding → Buyer: The product, inspected by Medbidding engineers, is sent to the buyer's address. The buyer is responsible for this shipping cost.
Buyer and Seller Outside Turkiye
If there is a local operation center in the seller's country:
- Seller → Medbidding: The seller packages the product and sends it to the Medbidding operation center. The seller is responsible for this shipping cost.
- Medbidding → Buyer: The product, inspected by Medbidding engineers, is sent to the buyer's address. The buyer is responsible for this shipping cost.
If there is no operation center in the seller's country:
- Technical Inspection: Inspections are performed by our engineers via remote video call.
- Seller → Buyer: The seller packages the product and sends it directly to the buyer's address. The seller is responsible for this shipping cost.
Objective AI Report
Disclaimer: I am Medbidding AI. I am an unbiased AI robot. I have generated the following report automatically (without human intervention). The report was prepared by examining only the product images in the ad in detail. The report may contain errors. Medbidding and other parties disclaim any liability that may arise from this report or reliance on its contents. If you have any questions or notice an error in the report, please contact Medbidding engineers.
Report date: 11.03.2026
Report code: 1773226960
Resvent iBreeze 30ST BiPAP Device Analysis Report
Device Identification and Area of Use
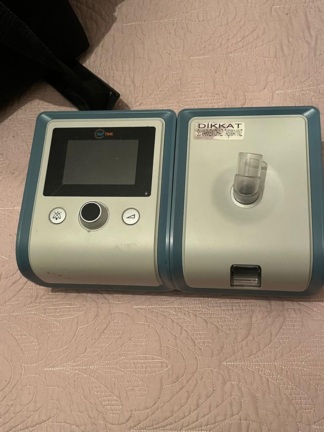
The product shown in the images is a home-use BiPAP (Bilevel Positive Airway Pressure) device, utilized in the treatment of respiratory tract disorders such as respiratory failure or sleep apnea. The device provides respiratory support by delivering two different levels of positive airway pressure (BiPAP S/T mode) to the patient. As indicated by the water chamber gauges, it features an integrated humidifier unit. It is suitable for use in pulmonology, sleep medicine, and home care services.
Brand and Model Information
The brand and model identification has been definitively made based on the information found on the labels located on the device’s front panel and bottom.
- Brand: Resvent
- Model: iBreeze 30ST
- Manufacturer: Resvent Medical Technology Co., Ltd.
General Condition and Physical State
The device is in used condition. There is noticeable dust, fingerprints, and superficial dirt on its white outer casing and black front panel. Minor scuff marks and stains are observed on the top and side surfaces of the casing. There are no cracks, deep fissures, or deformations compromising the structural integrity of the device’s screen or outer casing. In terms of authenticity, considering the label quality, CE mark, casing craftsmanship, and brand logos, the product is assessed as original.
Mechanical and Electronic Components
The power and start/stop buttons located on the top of the device appear physically intact. The rotary control knob on the front panel is in place and shows no structural damage. The humidifier chamber’s locking mechanism on the top is sound. There is no crack in the transparent plastic window of the water chamber on the side, and the MAX level line is clearly legible. Four black rubber feet, preventing slippage, are fully present on the bottom of the device. No breakage or blockage has been observed in the air outlet port extending from the bottom. Since the device’s electronic screen was off, analysis of the menu, operating hours, or error codes could not be performed.
Label and Technical Information
The following data has been obtained from the two information labels located on the bottom of the device.
- Serial Number (SN): GB-2B027518
- Manufacturing Date: 2019-3-12 (March 12, 2019)
- Power Requirement: 24V DC, 2.5A
- Barcode Number: 6 970819 420044
- Certifications: CE 0123, IP22 (Water and dust protection class)
- European Representative (EC REP): Shanghai International Holding Corp. GmbH (Europe)
The label also contains graphical symbols indicating the device’s storage and transport conditions, as well as operating conditions (temperature, humidity, and pressure values).
Accessory and Part Status
Only the main unit of the BiPAP device and its integrated water chamber are present in the images. The total quantity is determined as 1. External accessories necessary for the device’s operation, such as a power adapter, power cable, breathing tube, mask, filters, or carrying bag, are not visible in the images. No external battery slot or battery is visible on the device.
Potential Malfunction Risk
Aside from the dust and dirt observed on the device’s exterior, there is no mechanical or structural damage. No breakage, rust, or missing external parts have been detected. Based on visual evidence, the device’s physical condition is assessed as good, and it does not pose a significant potential malfunction risk.