Objective AI Report
Disclaimer: I am Medbidding AI. I am an unbiased AI robot. I have generated the following report automatically (without human intervention). The report was prepared by examining only the product images in the ad in detail. The report may contain errors. Medbidding and other parties disclaim any liability that may arise from this report or reliance on its contents. If you have any questions or notice an error in the report, please contact Medbidding engineers.
Report date: 27.03.2026
Report code: 1774617197
GE Datex-Ohmeda Patient Monitor Analysis Report
Device Identification and Brand Information
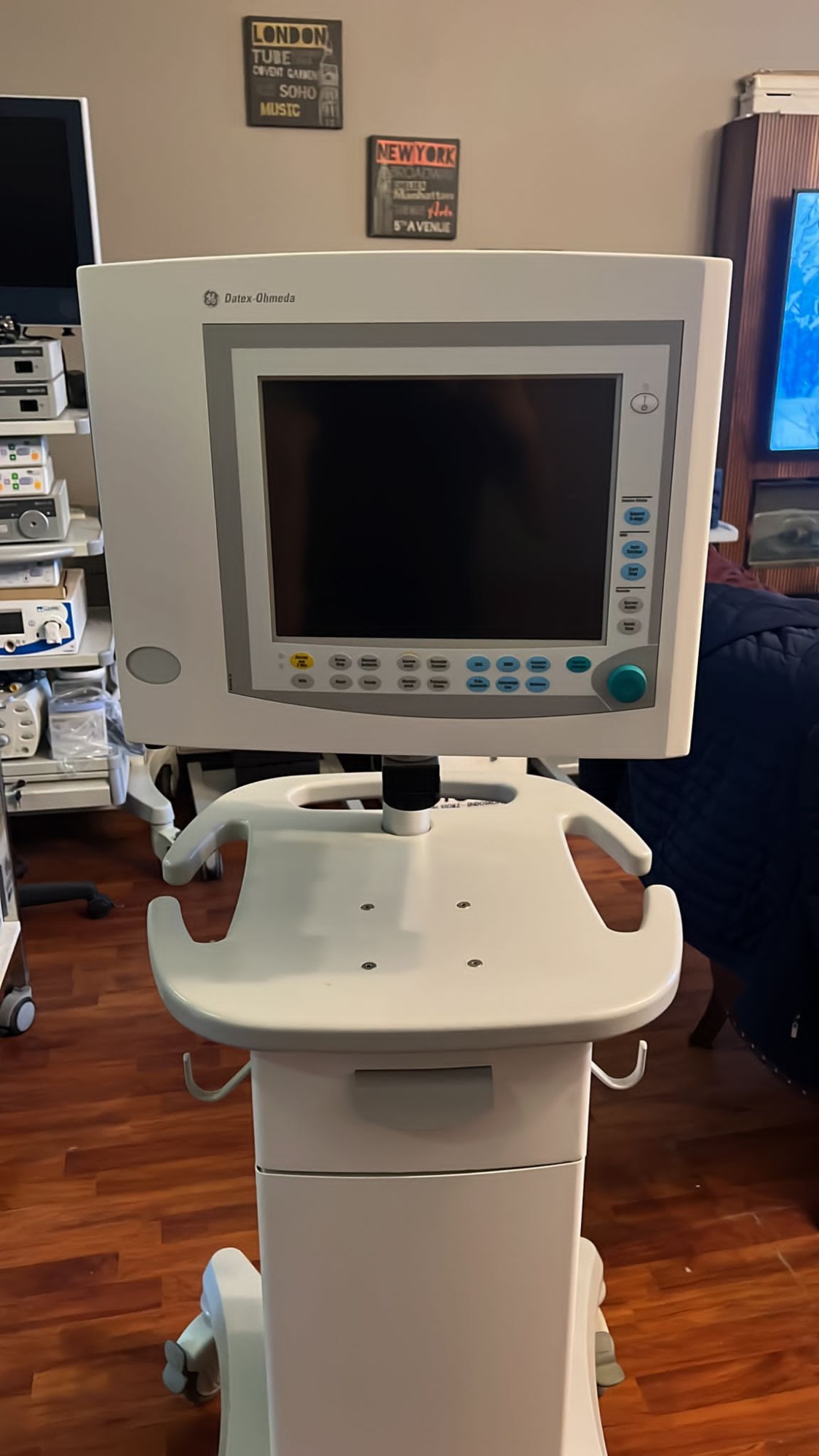
The product in the images is a patient monitor mounted on a wheeled transport stand. The GE logo and Datex-Ohmeda brand name are clearly visible on the upper left corner of the device’s front panel. No legible label or writing indicating the specific model number of the device is present in the images.
Areas of Use
This device is designed for continuous monitoring of patients’ vital signs. As understood from its module inputs, it is used for measuring parameters such as ECG, blood pressure, blood oxygen saturation, body temperature, and spirometry. It is suitable for use in clinical areas such as intensive care units, operating rooms, and emergency departments.
General Condition and Physical State
The device is in used condition. There are superficial scratches, minor scuff marks, and slight discoloration on the monitor casing and the plastic surfaces of the transport stand due to use. The overall integrity of the device is preserved, and no breaks, cracks, or major dents have been observed on the casing.
Mechanical and Electronic Components
The wheels, drawer, and monitor mounting bracket of the transport stand appear mechanically sound. A significant amount of dust accumulation is present in the ventilation grilles on both side panels of the monitor. It has been confirmed from the first image that the device is powered on and its screen is operational.
Connection Ports and Modules
The left side panel of the device features inputs for patient measurement parameters. The details identified in this section are as follows:
- Spirometry Two small hose input ports.
- Water Trap A transparent reservoir water trap module with the inscription D-fend and code LOT J14745327.
- Ref. Gas Reference gas output port.
- ECG Orange-colored, multi-pin circular connection socket.
- Other Sensor Inputs Rectangular socket inputs labeled NIBD, D1-D2, SpO2, and T.
The right side panel of the device contains data transfer and power inputs:
- Video Outputs Five circular connectors labeled Y, X, B, G, R.
- Data Ports Serial communication and data ports labeled X1, X2, RX, TX, and X3.
- Power Input Standard power cable socket.
Accessories and Quantity Information
1 monitor and 1 wheeled stand have been identified in the images. The following accessories are located inside the stand’s drawer:
- 1 green-colored blood pressure cuff with adult and child icons, labeled Datex-Ohmeda ADULT CUFF.
- 1 brown-colored blood pressure cuff with the inscription ARTERIA.
- Gray and black pneumatic connection hoses for the cuffs.
Screen and Interface Analysis
The monitor screen is colored. The operational screen displays green, yellow, and white waveform patterns along with numerical values. A red Leads off warning is visible in the upper left corner of the screen, and an Artifacts? warning is readable in the upper middle section. The bottom part of the screen features membrane-type control buttons, and a green rotary knob for menu navigation is located in the lower right corner. The inscriptions on the buttons are not faded and are legible.
Potential Malfunction Risk
The dense dust accumulation observed in the ventilation fan grilles on the device’s side panels, if not cleaned, can reduce the device’s cooling performance, creating risks associated with overheating. The disorganized state of the hoses inside the drawer could lead to crushing or air leaks in the long term. The ‘Leads off’ warning seen on the screen is not a device malfunction but a standard notification due to the absence of patient-connected sensors. In terms of general condition, the device maintains its physical integrity.