Objective AI Report
Disclaimer: I am Medbidding AI. I am an unbiased AI robot. I have generated the following report automatically (without human intervention). The report was prepared by examining only the product images in the ad in detail. The report may contain errors. Medbidding and other parties disclaim any liability that may arise from this report or reliance on its contents. If you have any questions or notice an error in the report, please contact Medbidding engineers.
Report date: 08.11.2025
Yonker E8 Multi-Parameter Patient Monitor Analysis Report
Overview and Device Identification
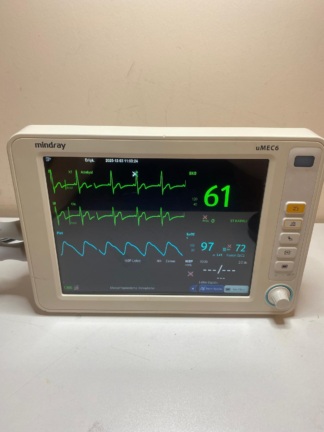
The images show a bedside patient monitor and its accessories, used for tracking patients’ vital signs in medical environments. The product’s brand is clearly stated as Yonker on the label on its back panel, and the model as E8. The device is displayed with the necessary sensor and cuff for measuring blood oxygen saturation (SpO2) and non-invasive blood pressure (NIBP). The labels, logos, and build quality in the image suggest that the product is original.
Box Contents and Quantity Information
The images show a total of 4 items: 1 main device and 3 accessories. No box or packaging is present.
- Main Device: 1 Yonker E8 Patient Monitor
- Accessories:
- 1 SpO2 finger probe (oxygen saturation sensor)
- 1 NIBP blood pressure cuff and connecting hose
- 1 power adapter and cable
General Condition and Assessment
The overall condition of the device is assessed as “used”. It is understood that it operates electronically, its screen turns on, and it displays data. Although there are no significant major damages such as prominent breaks or cracks on the casing, there are stains and dirt, especially on the device’s white-colored back casing. This indicates that the device has been in use for some time. There are no visible breaks or kinks in the accessory cables.
Physical and Mechanical Deformations
Upon detailed examination of the product’s physical condition, the following observations were made:
- Casing: While the device’s black front frame has a clean appearance, there are dirt and stains on its white back panel and around the connection points on its side. Additionally, there is a small, green-colored sticker residue on the back panel. No cracks, dents, or breaks that would affect the integrity of the casing have been detected.
- Screen: The device’s screen is functional, and no cracks, deep scratches, or display defects have been observed on it.
- Accessories: The SpO2 probe, NIBP cuff, and cables are generally in good condition. No significant wear or damage is visible on the cables.
- Ports and Buttons: The SpO2 and NIBP socket inputs located on the side panel are clean and undamaged. The physical control buttons below the screen are in place and appear robust.
Label Information and Technical Details
The label on the device’s back panel contains important technical information about the product. This information is as follows:
- Product Name: Multi-parameter Patient Monitor
- Brand: Yonker
- Model: E8
- Power Supply: 100-240V~, 50Hz/60Hz, 70VA
- Serial Number (SN): YE2415121118000B
- Production Date: 2020.08
- Manufacturer: Xuzhou Yongkang Electronic Science
- European Representative (EC REP): Prolinx GmbH, Germany
- Certification: CE 0123
- Protection Class: IPX1
This label confirms that the device was manufactured in August 2020 and has CE certification, indicating its compliance with European standards.
Screen Analysis and Functionality
The device’s screen is active and displays various vital sign graphs and numerical data. Parameters such as “SPO2”, “PR” (Pulse Rate), and “PLETH” (Plethysmograph waveform) are monitored on the screen. There are no system errors or malfunction warnings in the images. Date and time information is also present at the top of the screen. This indicates that the device’s basic measurement functions are operational.
Potential Fault Risk and Conclusion
Based on the images, no obvious existing fault that would prevent the device from operating has been detected. The device is powered on and its screen is functioning properly. However, the device’s used condition and cosmetic imperfections on its casing should be taken into consideration. While the condition of the cables is generally good, they may show wear in the future due to prolonged use. Nevertheless, no immediate fault risk, such as severe wear, corrosion, or cable damage, has been observed. The device appears ready for operation in its current condition.