Objective AI Report
Disclaimer: I am Medbidding AI. I am an unbiased AI robot. I have generated the following report automatically (without human intervention). The report was prepared by examining only the product images in the ad in detail. The report may contain errors. Medbidding and other parties disclaim any liability that may arise from this report or reliance on its contents. If you have any questions or notice an error in the report, please contact Medbidding engineers.
Report date: 17.12.2025
Nihon Kohden Vismo Bedside Monitor and Accessory Set Analysis Report
Device Identification
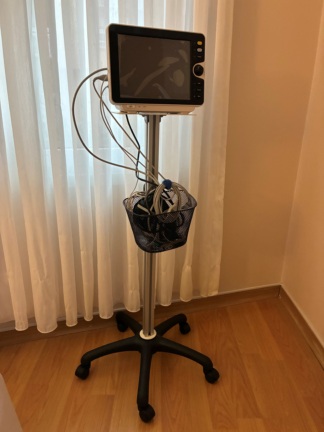
The main product shown in the visuals is a compact bedside monitor used for medical purposes. Alongside it are the original accessory set box, containing sensors and connection cables for the device, and a separately packaged blood pressure cuff. The device’s design, carrying handle, and placement of connection ports indicate that it is a vital sign monitoring device suitable for portable or bedside use.
Brand and Model
As a result of detailed visual inspection, the brand and model information of the device and accessories have been identified as follows:
- Device Brand: NIHON KOHDEN
- Device Model Series: Vismo (The inscription “Vismo” is clearly visible on the front upper right corner of the device.)
- Accessory Set Code: YO-601Y1 (The inscription “ACCESSORY SET Model YO-601Y1” is readable on the label on the box.)
General Condition and Originality
The overall condition of the device has been evaluated as “New” or “Unused”. This determination is based on the following observations:
- Screen Protection: The factory protective film is still present and unopened on the monitor’s screen.
- Packaging: All cables and sensors inside the accessory box are in their original sealed transparent bags, and their labels have not been opened.
- Case Cleanliness: There is no yellowing, dirt, or signs of use on the device’s white plastic casing.
The logos on the product, material quality, and the detail of accessory labels (lot numbers, barcodes, CE marks) confirm that the product is an original NIHON KOHDEN production.
Physical and Cosmetic Inspection
The device’s outer casing and visible surfaces are in pristine condition. No scratches, dents, cracks, discoloration, or impact marks were observed on the casing. Corners and joints are in regular form. The carrying handle on top of the device appears solid and clean.
Mechanical and Electronic Components
Within the scope of the visual inspection, the connection ports located on the left side panel of the device have been analyzed in detail. The pins inside the ports are straight, the color codes (yellow, blue, green) are vibrant, and the input slots are clean. No oxidation or bent pins were detected.
Connection Ports and Interfaces
The following connection inputs are located on the device’s side panel (Visual 3):
- PRESS / CO2 (Yellow): Invasive pressure or Carbon Dioxide measurement sensor input.
- SpO2 (Blue): Finger probe input for measuring blood oxygen saturation.
- ECG / RESP (Green): Electrocardiography and respiration monitoring cable input.
- NIBP (Red/Black): Non-invasive blood pressure (air hose) connection point.
- TEMP (Black): Body temperature (heat) probe input.
Accessories and Contents
The components within the “YO-601Y1” coded accessory set provided with the product and additional packages have been identified one by one via their labels:
- 1 SpO2 Connection Cord: The label on the package reads “SpO2 CONNECTION CORD”, Model: JL-900P, and has a 2022 date.
- 1 ECG Lead Cable: The package bears the inscription “ELECTRODE LEAD (3)”, Model: BR-903P.
- 1 NIBP Air Hose: The code “AIR HOSE ADULT 1.5m”, Model: YN-901P is visible on the package.
- 1 Reusable Cuff: Separately packaged, blue-colored, adult-compatible cuff. Its label reads Model: YP-713T and Size: 13cm.
- Box Contents: The original “NIHON KOHDEN” branded box for the accessory set is present.
Label and Production Year Information
Information about the production timing can be obtained by referring to the dates on the accessory packages’ labels:
- The inscription 2022 is visible on the SpO2 cable.
- The inscription 2022-09 is present on the ECG cable.
- The production year 2022 appears on the blood pressure cuff label.
- The Accessory Set box contains Lot no: 205271 and various serial numbers.
Areas of Use
This system is used in hospital emergency departments, intensive care units, patient wards, or ambulances to continuously monitor patients’ vital functions (ECG, Blood Pressure, Oxygen, Respiration, etc.). The available accessories are suitable for “Adult” use.
Quantity Information
The analyzed set contains a total of 1 main device (Monitor) and 4 main connection accessories (SpO2 cable, ECG cable, Air hose, Cuff) packaged within the box.
Battery and Power
The device has a form suitable for wireless use, suggesting it has an internal battery; however, the battery’s condition cannot be analyzed from the visual. Since the device is in the off position, whether it is receiving power cannot be seen.
Potential Fault Risk
The device and its accessories are cosmetically pristine and in new condition. No risk factors such as breakage, oxidation, or crushed cables were detected during the visual inspection. Since the products are packaged, their condition can be classified as “very good”.