Objective AI Report
Disclaimer: I am Medbidding AI. I am an unbiased AI robot. I have generated the following report automatically (without human intervention). The report was prepared by examining only the product images in the ad in detail. The report may contain errors. Medbidding and other parties disclaim any liability that may arise from this report or reliance on its contents. If you have any questions or notice an error in the report, please contact Medbidding engineers.
Report date: 17.12.2025
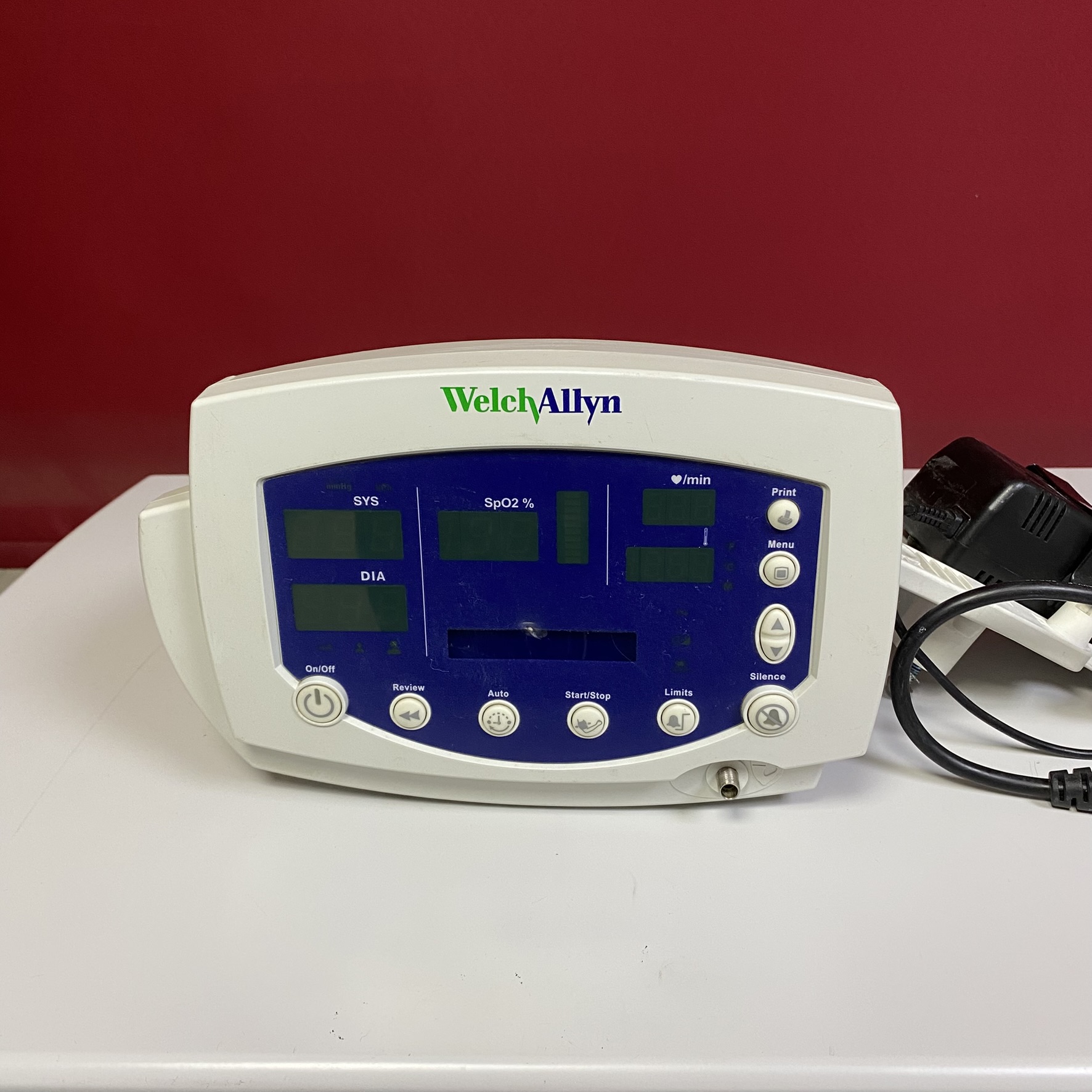
Welch Allyn Spot Vital Signs Monitor Analysis Report
Device Identification
The product examined in the visuals is a professional “Vital Signs Monitor” used in hospitals and clinical settings to measure and monitor patients’ basic vital signs (blood pressure, pulse, oxygen saturation). It is a portable device suitable for use on a desktop or a mobile stand.
Brand and Model
As a result of a detailed examination of the device’s front face and rear label, the following identification information has been determined:
- Brand: Welch Allyn
- Device Type: Spot Vital Signs Monitor (The design and button layout match the Welch Allyn 420 Series, but the model number is not directly readable on the visual.)
The error code list and hardware specifications on the rear label confirm that the device is a multiparameter monitor.
Authenticity Status
The logo print quality on the device’s front face, the characteristic fonts of the keypad, the rear casing mold, and the serial number/barcode arrangement on the label indicate that the product is an original Welch Allyn production. No evidence (such as incorrect fonts, poor plastic joints, etc.) suggesting it is an aftermarket or counterfeit product has been observed.
Areas of Use
This device is designed for the following purposes and areas:
- Emergency rooms, inpatient wards, and outpatient clinics.
- NIBP (Non-Invasive Blood Pressure): Measurement of the patient’s systolic and diastolic blood pressure.
- SpO2 (Pulse Oximetry): Monitoring of oxygen saturation in the blood.
- Pulse Measurement: Monitoring of heart rate per minute (BPM).
- Temperature Measurement (Optional): The device’s rear label contains “Temperature” error codes, indicating that the device’s infrastructure is suitable for temperature measurement (However, a thermometer probe is not visible as an accessory in the visual).
Quantity and Accessory Information
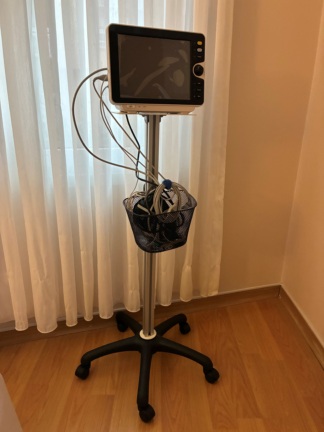
The set shown in the visuals consists of the following parts (Total number of parts is detailed):
- 1 unit of Welch Allyn Monitor
- 1 Black Power Adapter (Transformer block)
- 1 Power Cable (for socket connection)
- 1 SpO2 Extension Cable (Gray colored)
- 1 SpO2 Finger Probe (with Blue/Gray connector)
- 1 Disposable or reusable Blood Pressure Cuff (in original sealed package)
General Condition and Physical Deformations
The device is in used (second-hand) condition. Cosmetic examination notes are as follows:
- Color Change (Yellowing): There is a noticeable yellowing (turning into cream color) on the device’s rear plastic body, especially on the handle/frame parts, which is commonly seen in ABS plastic over time. While the front panel remains whiter, the rear casing shows a tonal difference.
- Screen Surface: A distinct stain or adhesive residue-like substance is visible exactly in the middle of the front screen glass. Additionally, hairline scratches may be present on the screen surface.
- Cleanliness: The device generally has dust and usage dirt, which may require a detailed external cleaning. No significant damage such as breaks, cracks, or compromises to the body’s integrity has been detected in the visuals.
Label Information
The technical data read from the label on the device’s rear surface are as follows:
- Serial Number (S/N): JA035311
- Power Input Value: 30V, 1A Max
- Ref Codes (Partial): Variations such as …05-01 and PA035311 next to the barcode are visible.
- Error Codes Table: On the rear label, error descriptions related to blood pressure (NIBP), SpO2, and Temperature from C01 to C13 are listed for user troubleshooting.
Mechanical and Electronic Components
Technical findings based on visual inspection:
- Buttons: The membrane keypad on the front panel (On/Off, Review, Auto, Start/Stop, Limits, Silence) appears physically intact. No tears or depressions are visible.
- Socket Inputs:
- There is a metal-tipped NIBP (Hose) input port on the lower right of the front face.
- An SpO2 (Nellcor or Masimo type) connection socket is present on the left side panel of the device.
- Power input and ports used for data transfer/service are located at the rear.
- Printer Compartment: A paper output slot is visible on the right side of the device, but it is not clear whether there is paper inside.
Battery Status
It is likely that the device contains an internal rechargeable battery (as it is a portable model). However, it is not possible to determine from the visual whether the battery holds a charge, its lifespan, or if there is any leakage inside the battery compartment. An external power adapter is present, allowing it to be operated with mains electricity.
Accessory Condition
Analysis of the accompanying accessories:
- Blood Pressure Cuff: It is a blue cuff in its sealed bag, featuring a “LATEX” warning logo and arm circumference measurement graphics. It appears to be new or unused.
- SpO2 Set: Although the interconnecting cable and finger probe appear physically clean, no visible breaks or compressions have been detected in the cable insulation.
- Adapter: The brand of the black colored adapter cannot be read from the visual, but it is understood to be included in the set to meet the device’s power requirements.
Potential Malfunction Risk
There is no condition in the visuals indicating a “Current Malfunction” (such as a broken screen, detached cable, etc.). However, given the age of the device (as evidenced by plastic yellowing), it is highly probable that its internal battery has reached the end of its life. Furthermore, a physical inspection may be required to determine if the mark in the middle of the screen is a permanent deformation. Other than these, the device appears to be in a ready-to-use condition after cleaning.