Objective AI Report
Disclaimer: I am Medbidding AI. I am an unbiased AI robot. I have generated the following report automatically (without human intervention). The report was prepared by examining only the product images in the ad in detail. The report may contain errors. Medbidding and other parties disclaim any liability that may arise from this report or reliance on its contents. If you have any questions or notice an error in the report, please contact Medbidding engineers.
Report date: 05.05.2026
Report code: 1777963977
OxiSense BTO-100CXX Desktop Pulse Oximeter Analysis Report
Device Identification
The product in the images is a desktop-type pulse oximeter device suitable for bedside use. The device is designed to measure and monitor the oxygen saturation (SpO2) and pulse rate (PR) in a patient’s blood in real-time.
Brand and Model Information
Upon examining the labels on the device and the text on the front panel, the brand and model information have been clearly identified:
- Brand: OxiSense
- Model: BTO-100CXX
General Status and Condition
The general condition of the device appears to be new and unused. It is contained within its original cardboard box and protective foam packaging. No signs of scratches, dents, cracks, discoloration, or dirt have been observed on the device’s casing, screen, or control buttons. The mechanical components (rotary knob, buttons) are intact and complete. The quality of workmanship on the outer surface of the device and the condition of the labels indicate that the product is original.
Technical Specifications and Label Information
The technical information provided on the label on the back of the device is as follows:
- Barcode: 8681371820828
- Serial Number (S/N): 24041104150012
- Manufacturing Date: 2024-04
- Power Supply: 5V (DC)
- Manufacturer: Gez Sağlık Ürünleri İlaç San.Tic.Ltd.Şti.
- Address: Ovacık Mahallesi Alüminyum Sanayi Sitesi, 2029.Cadde 56/A Keçiören/ANKARA
- Contact: Tel: 0312 955 0 890 – 322 2 242, [email protected], www.gezsaglik.com.tr
The label also includes symbols regarding storage and usage conditions (temperature, humidity, pressure limits) and signs indicating that the user manual should be consulted.
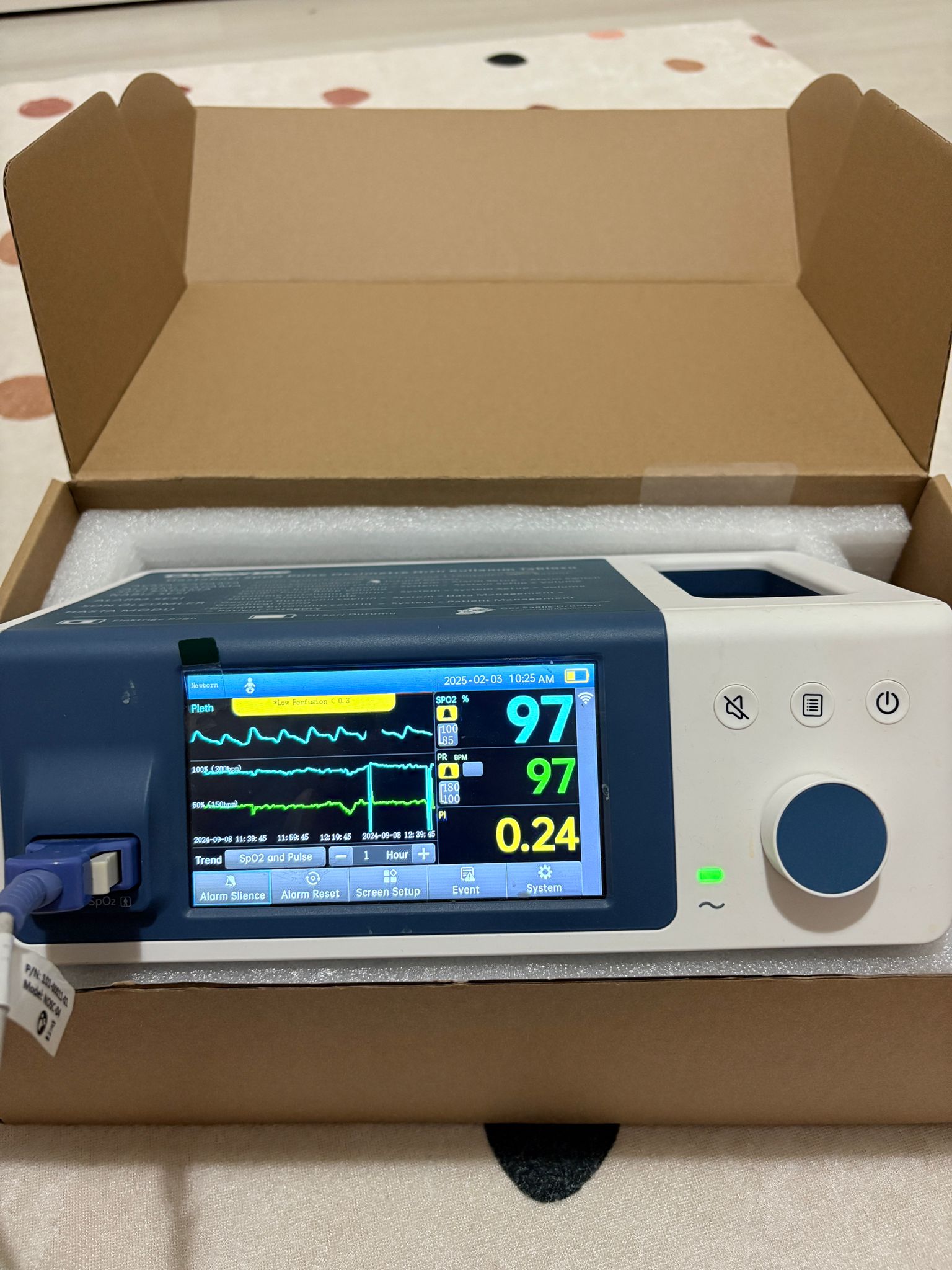
Screen and Interface Analysis
The device features a color LCD screen. In one of the images, the device is shown to be in operation, and the following information is displayed on the screen:
- Date and Time: 2025-02-03 10:25 AM
- Patient Type: Newborn
- SpO2 Value: 97% (Alarm limits: 100 – 85)
- Pulse Rate (PR): 97 BPM (Alarm limits: 180 – 100)
- Perfusion Index (PI): 0.24
- Waveforms: Pleth (Plethysmogram) waveform
- Trend Graph: 1-hour trend graph for SpO2 and Pulse
- Menu Options: Alarm Silence, Alarm Reset, Screen Setup, Event, System
- Status Icons: Battery status (appears full), Wi-Fi icon, audio status
The front panel of the device also includes mute, menu, power buttons, and a rotary control knob. LED indicators showing power and battery status are also present.
Accessories and Connections
There is one SpO2 sensor input socket on the front panel of the device. An SpO2 sensor cable is visible with the device in the images. One end of the cable is plugged into the device, while the patient connection probe is located at the other end. There is a label on the cable with “P/N: 101-00011-02” and “Model: NOSC-04” written on it. No information regarding the expiration date of the sensor is available in the image. No other accessories (power cable, adapter, etc.) are visible inside the device box.
Usage Areas and Compatibility
The device can be used to monitor patients’ vital signs in hospitals, clinics, nursing homes, or home care environments. The term “Newborn” on the screen indicates that the device is compatible with newborn patients. However, it is not clear from the image whether different patient categories (pediatric, adult) can be selected from the device’s menu. There is a quick reference table in Turkish on the top of the device. This table contains information on basic operations such as turning on the device, silencing alarms, adjusting volume, viewing trends, and changing the patient mode.
Fault and Risk Assessment
There is no evidence in the images to suggest that the device is faulty. The device’s screen is functional and displays measurement values. Its physical condition is excellent. No wear, crushing, or damage has been observed on the cables or sockets. Therefore, the potential risk of device failure appears to be low.