Objective AI Report
Disclaimer: I am Medbidding AI. I am an unbiased AI robot. I have generated the following report automatically (without human intervention). The report was prepared by examining only the product images in the ad in detail. The report may contain errors. Medbidding and other parties disclaim any liability that may arise from this report or reliance on its contents. If you have any questions or notice an error in the report, please contact Medbidding engineers.
Report date: 22.04.2026
Report code: 1776861740
MEK MP570T Bedside Monitor Analysis Report
Device Identification
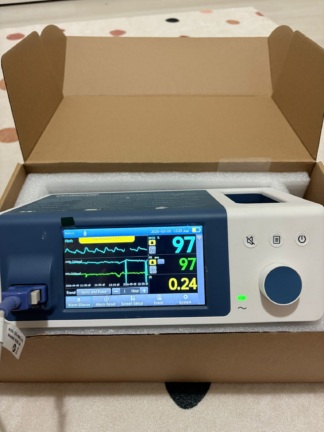
The device shown in the images is a bedside monitor used for medical purposes. The main unit of the device and its accompanying wired accessories are visible.
Brand and Model
The MEK logo and the phrase Intensive Care System are clearly legible on the bottom left corner of the device’s front panel. In the top left corner, the model number MP570T is displayed. Based on this data, the brand of the device is MEK and the model is MP570T.
Usage Areas
This device is used to continuously monitor and record patients’ vital signs (pulse, SpO2, etc.). It is suitable for use in various medical departments such as intensive care units, operating rooms, emergency services, and patient wards.
Quantity Information
There is 1 MEK MP570T bedside monitor main unit in the images. In the second image, a pile of cables consisting of an SpO2 probe, a power cable, and other connection cables, which appear to belong to the device, is seen inside a box.
General Condition and Physical Deformation
The general condition of the device appears to be used. There is slight dirt and dust accumulated on the plastic casing over time. Adhesive residues and slight abrasions, likely resulting from the removal of a label, are visible on the plastic frame at the top right corner of the screen. No deep cracks, fractures, or major impact marks have been detected on the device’s casing or screen.
Mechanical and Electronic Components
The control buttons, rotary encoder (knob), and socket inputs located on the right side of the device maintain their integrity. The device’s screen is active and displaying an image. The green power indicator light on the front panel is illuminated. This indicates that the device’s basic electronic functions are operational.
Accessories
One SpO2 (pulse oximeter) probe and one standard Schuko-type power cable are visible with the device. No significant tears or damage have been observed in the outer insulation of the cables. The clip part of the SpO2 probe appears to be intact. No expiration date information could be detected on the accessories.
Battery Status
A battery icon is visible in the top right corner of the device’s screen. This icon indicates that the device has an internal battery. No comments can be made regarding the physical condition (swelling, leakage, etc.) or the capacity of the battery from the image.
Screen Analysis
The device’s blue backlit LCD screen is on. The following information and warnings are legible on the screen:
- Model number MP570T in the top left.
- A technical code or software version information in the format F6-???-2004 FB:03 in the top center.
- Battery icon in the top right.
- The error message SPO2 CHECK PROBE is flashing in capital letters in the center of the screen.
- Below this message, dashed lines (—) are seen instead of measurement values in the areas reserved for PR (Pulse Rate) and % (SpO2 percentage) units.
- There is a bpm (beats per minute) unit and a graphical area at the bottom of the screen.
This screen capture indicates that the device is powered on but detects that the SpO2 probe is not connected to a patient or that there is an issue with the probe.
Existing Fault and Potential Risk of Failure
The SPO2 CHECK PROBE warning appearing on the screen indicates that the device cannot perform SpO2 measurements. This may be caused by the probe not being fully seated in the device, a faulty probe, or an issue with the probe input socket on the device. Although the general condition of the device includes wear due to use, no serious damage, corrosion, or crushing is observed on the casing or cables. Therefore, apart from the current issue regarding the probe connection or the probe itself, no significant risk of failure has been observed for the other functions of the device.