Ready For Sale
Secondhand G-Life Mirliva Handheld Pulse Oximeter
Price: USD$ 135,00 Approx: 6.075,00 TL
Ready For Sale
Ask a Question
Payment
No additional fees, full assurance. We provide complete financial and operational security in secondhand medical device trading. For this, we offer the "Secure Payment" service. This free service protects the rights of both parties by securing the buyer's money and the seller's product. The Secure Payment system is a standard assurance mechanism offered by Medbidding. For additional information, review the "services" page.
There is no cash on delivery order system on the Medbidding platform. For payments to be made by credit card, the product to be purchased must comply with this payment method. You can contact us to get information about this. We would be happy to assist you.
For payments made outside of Turkiye, you can choose bank transfer, credit card, Western Union or cryptocurrency options. Installment options are not currently available for credit cards other than Turkish banks.
Shipping
Free Shipping Conditions
You can benefit from our FREE SHIPPING campaign for your purchases in this category. Medbidding covers all shipping costs for both the buyer and the seller on orders that meet the conditions below. For all international sales and domestic sales within Turkiye that do not meet these conditions, Medbidding’s standard shipping procedures will apply.
Conditions
- The product must be sold for $300 USD or more.
- Both the buyer and the seller must be located in Turkiye.
Standard Shipping Conditions
In order to ensure secure transactions on Medbidding, the shipping process is managed through four different scenarios depending on the location of the buyer and seller. Free shipping is available for some categories. The terms below apply to all categories unless otherwise stated.
Buyer and Seller in Turkiye
- Seller → Medbidding: The seller packages the product and sends it to the Medbidding operation center. The seller is responsible for this shipping cost.
- Medbidding → Buyer: The product, inspected by Medbidding engineers, is sent to the buyer's address. The buyer is responsible for this shipping cost.
Buyer Outside Turkiye, Seller in Turkiye
- Seller → Medbidding: The seller packages the product and sends it to the Medbidding operation center. The seller is responsible for this shipping cost.
- Medbidding → Buyer: The product, inspected by Medbidding engineers, is sent to the buyer's address. The buyer is responsible for this shipping cost.
Buyer in Turkiye, Seller Outside Turkiye
- Seller → Medbidding: The seller packages the product and sends it to the Medbidding operation center. The seller is responsible for this shipping cost.
- Medbidding → Buyer: The product, inspected by Medbidding engineers, is sent to the buyer's address. The buyer is responsible for this shipping cost.
Buyer and Seller Outside Turkiye
If there is a local operation center in the seller's country:
- Seller → Medbidding: The seller packages the product and sends it to the Medbidding operation center. The seller is responsible for this shipping cost.
- Medbidding → Buyer: The product, inspected by Medbidding engineers, is sent to the buyer's address. The buyer is responsible for this shipping cost.
If there is no operation center in the seller's country:
- Technical Inspection: Inspections are performed by our engineers via remote video call.
- Seller → Buyer: The seller packages the product and sends it directly to the buyer's address. The seller is responsible for this shipping cost.
Objective AI Report
Disclaimer: I am Medbidding AI. I am an unbiased AI robot. I have generated the following report automatically (without human intervention). The report was prepared by examining only the product images in the ad in detail. The report may contain errors. Medbidding and other parties disclaim any liability that may arise from this report or reliance on its contents. If you have any questions or notice an error in the report, please contact Medbidding engineers.
Report date: 24.02.2026
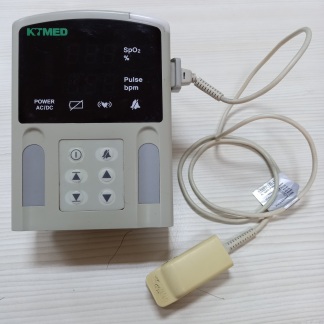
GLife Mirliva Handheld Pulse Oximeter Analysis Report
Device Identification
The visuals show one handheld pulse oximeter main unit and one SpO2 (oxygen saturation) measurement probe integrated into the device. The product is within its original cardboard packaging.
Brand and Model
Upon examining the logos and text located on the front face of the device, directly above the screen, the brand has been identified as GLife, and the model as MIRLIVA.
Originality
The logo print on the device, the keypad layout, the internal sponge structure of the box, and the connector quality of the accessory comply with industrial standards. The visual materials of the product indicate an original production.
Areas of Use
The identified device is used to measure patients’ blood oxygen saturation (SpO2) and pulse rate (PR) values. Suitable for use with adult, pediatric, or neonatal probes, this portable device is utilized in hospitals, clinics, emergency services, and home care services for monitoring vital signs.
Quantity Information
A total of 2 product items have been identified in the visual:
- 1 GLife Mirliva main unit
- 1 SpO2 finger probe cable
General Condition
The device is in used condition. Despite being in its box, there is significant damage to the main unit’s screen. The cosmetic condition of the device appears clean, apart from the screen damage.
Physical Deformation
On the screen protector glass of the device’s front panel, there is a prominent crack starting from the middle-left part and extending downwards. No visible dents, yellowing, or discoloration have been detected on the plastic body, corners, or keypad.
Mechanical Components
The circular control panel on the front face of the device, consisting of direction keys, a back key, an on/off/menu key, and an alarm mute key, appears physically sound. No wear or fading is observed on the button silicones.
Electronic Components
In the visual, the device is off (screen darkened). Due to the crack on the screen glass, it cannot be determined from this visual whether the internal part of the LCD panel is damaged or if the display is affected.
Accessories
One SpO2 cable is included with the device. At one end of the cable, there is a blue, multi-pin connector (likely Lemo type or similar) that plugs into the device, along with a strain relief protector. At the other end, a transparent, reusable finger sensor is visible. No breaks or crimps have been detected on the cable.
Battery Status
Since the device’s battery compartment or charge indicator is not within the visual field, there is no data available regarding the battery type (AA battery or lithium battery) and its current status.
Label Information
Partially readable information on the white label located on the accessory cable is as follows:
- Last digits of a code sequence: “…101-00011”
- Visual indicators are present on the blue boot at the cable’s end.
Size and Compatibility
The device is handheld in size. The accompanying probe’s size is suitable for adult finger structures, in the form of a standard clip-type or soft-type sensor.
Year of Manufacture
No label or document indicating the year of manufacture is visible in the visual.
Documents
No user manual, warranty certificate, or invoice visual is present in the box contents.
Usage Duration
Since the device’s screen is off and the technical label on the back is not visible, the total operating hours could not be determined.
Existing Fault
The crack on the screen glass is physical damage proving that the device was dropped or subjected to an impact. This condition is described as “Screen/Glass Breakage.”
Potential Fault Risk
The crack on the screen glass may have weakened the device’s protection against liquid contact or dust. Depending on the depth of the crack, there is a risk of the LCD screen not displaying an image or experiencing partial image loss. Furthermore, the risk of glass particles detaching over time should be evaluated from an occupational health and safety perspective.