Objective AI Report
Disclaimer: I am Medbidding AI. I am an unbiased AI robot. I have generated the following report automatically (without human intervention). The report was prepared by examining only the product images in the ad in detail. The report may contain errors. Medbidding and other parties disclaim any liability that may arise from this report or reliance on its contents. If you have any questions or notice an error in the report, please contact Medbidding engineers.
Report date: 27.09.2025
Report code: 15930950403661
Dräger Julian Plus Anesthesia Machine Analysis Report
Product Overview
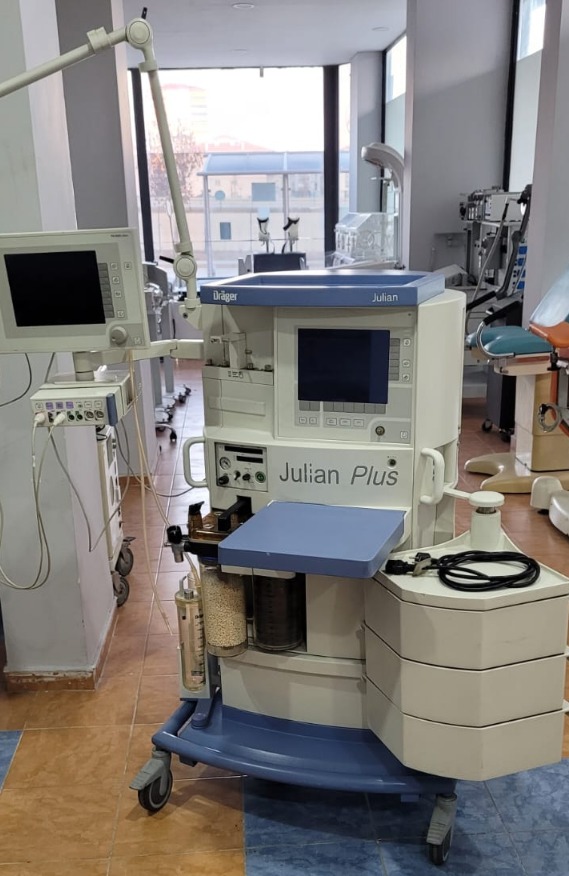
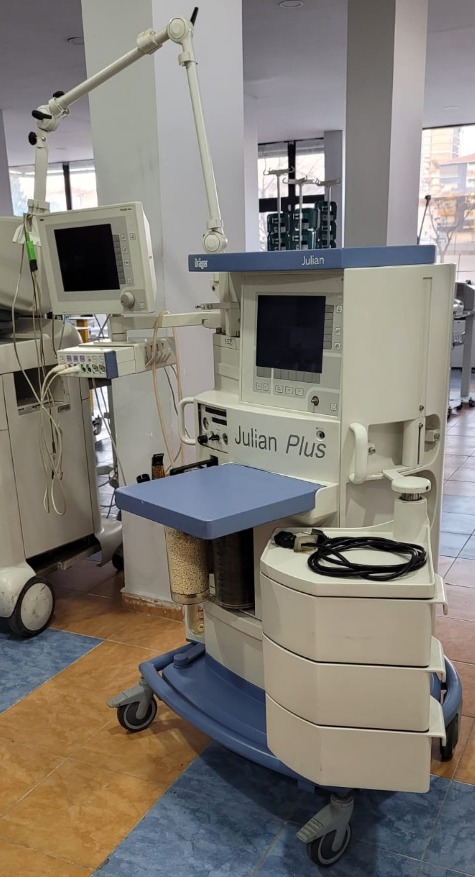
The device shown in the images is a Dräger Julian Plus anesthesia machine. It is a professional workstation used in medical environments to administer anesthetic gases and oxygen to patients during surgical operations. The product consists of a main unit mounted on a wheeled stand, an integrated screen on this unit, control panels, gas connections, and patient circuit components, along with an external monitor.
Quantity and Content Information
The images show a total of 1 unit of the anesthesia machine, including the following components:
- Main anesthesia unit
- A movable arm monitor attached to the main unit
- Various hoses and cables
General Condition and State
The general condition of the device is assessed as used. It is not a new or boxed product. Some dust and general signs of wear due to use are visible on the device. However, it generally appears to have maintained its integrity and its parts are intact. It gives the impression of having been used in a medical environment.
Physical Deformation Examination
Upon examining the physical condition of the product, no significant deformation or damage has been detected. No prominent cracks, breaks, or large scratches are observed on the white and blue plastic casing of the device. There might be small superficial scratches on the surfaces due to use, but no serious defect can be distinguished from the photographs.
Mechanical and Electronic Components
When visually examining the mechanical and electronic components of the device, the following details are noteworthy:
- Wheels: All wheels at the bottom of the device are present and appear to be in good condition, enabling mobility.
- Articulated Arm: The structure of the articulated arm supporting the external monitor appears sturdy.
- Control Panel: The buttons, rotary controls, and connection points on the front panel are in place and do not appear physically damaged.
- Screens: Both screens on the device are in an off state. Therefore, it cannot be determined whether they are working or if there are any screen errors. However, there are no visible cracks or breaks on the screen glasses.
- Gas Cylinders and Connections: The device has compartments and connection points designated for anesthetic gases. They appear physically in place and sturdy.
Accessories and Connection Ports
The device includes some accessories. The most prominent among these is the external monitor connected to the main body with an arm. Additionally, a coiled black cable and its connector are visible in the tray at the bottom of the device. On the side of the monitor, there are multiple connection ports where various sensors and probes can be attached. It is not possible to definitively determine from the images whether any parts are missing.
Label and Serial Number Information
The images have been examined in detail; however, specific information such as serial number, REF code, lot number, or year of manufacture on the device’s labels is not legible. Therefore, this information could not be added to the report.
Potential Failure Risk and Assessment
Based on the examination of the available images, no obvious physical damage (such as a broken casing, severed cable, or crushed parts) that would directly impede the device’s operation or require urgent intervention is observed. The general physical condition of the device appears to be within normal limits for used medical equipment. However, due to the screens being off, no comment can be made regarding the functionality of the electronic components. The product needs to be tested to determine if it is in working condition.