Objective AI Report
Disclaimer: I am Medbidding AI. I am an unbiased AI robot. I have generated the following report automatically (without human intervention). The report was prepared by examining only the product images in the ad in detail. The report may contain errors. Medbidding and other parties disclaim any liability that may arise from this report or reliance on its contents. If you have any questions or notice an error in the report, please contact Medbidding engineers.
Report date: 27.12.2025
Icare ONE Intraocular Pressure Measuring Device (Tonometer) Analysis Report
Device Identification and Description
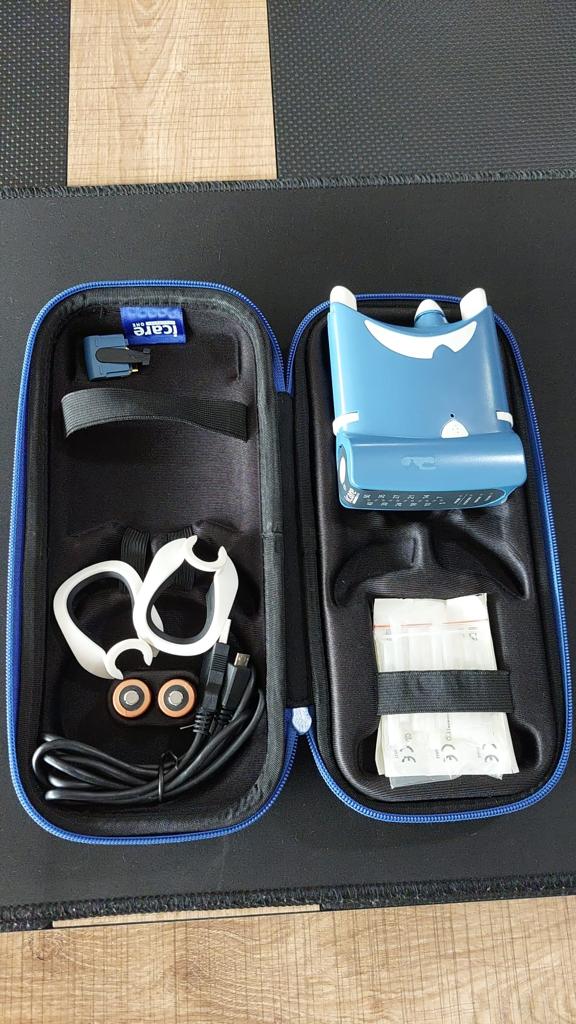
The device shown in the visuals is a portable medical measuring instrument used in the field of ophthalmology. The ergonomic design of the device, its digital display panel, and the specific probe tips provided indicate that it is a rebound tonometer used for measuring intraocular pressure (IOP). The product comes as a complete set with its custom foam-padded carrying case and measurement accessories.
Brand and Model Information
As a result of examinations made on the front face of the device, just above the screen, and on the back label, the brand and model information has been definitively identified as follows:
- Brand: Icare (The logo is rendered as ‘icare’ with ‘ONE’ inscribed below it)
- Model: ONE (Clearly written on the front panel)
- Type/Technical Model Code: Type TA02 (The phrase “Type TA02” is present on the side label of the device)
- Manufacturer: Icare Finland Oy (Stated on the side label)
- Origin: Made in Finland (Stated on the label as Made in Finland)
Areas of Use
This device is designed to measure intraocular pressure by contacting the eye (using “rebound” technology). It is used by patients requiring glaucoma (eye pressure) monitoring or by healthcare professionals. The “Icare ONE” model is a simplified version, typically designed for patients to perform self-measurements at home.
Authenticity Status
The overall material quality of the product, plastic injection details, regular placement of logos, and especially the “Icare Finland Oy” markings on the label along with the CE 0044 certification mark, demonstrate that the product is an original Icare production. The “icare ONE” logo on its case and the original accessories also support this status.
Package Contents and Quantity Information
The total parts and accessories visible within the carrying case in the visual are as follows:
- 1 Icare ONE Tonometer Device (Blue colored)
- 1 Original Zippered Carrying Case (Hard-protected)
- 1 White Forehead Support (The curved part attached to the tip of the device)
- 1 USB Connection Cable (Black)
- 1 Wrist Strap (Black)
- 2 Alkaline AA Batteries (Duracell-looking, next to the device compartment)
- 1 pack of Sterile Probe Set (In sealed packaging, spare probes are present although the exact number cannot be clearly counted)
- 1 Plastic Part (In the mesh pocket of the case’s top lid, likely a probe base or battery cover key)
Condition and Cosmetic Evaluation
The outer casing of the device appears to be in “Excellent” condition. No discoloration, yellowing, or texture degradation has been detected on the blue plastic casing. The markings on the keypad (5, 10, 50, etc.) are extremely clear and legible; there is no fading. The white forehead support parts are clean and have not yellowed. The device gives the impression of being “like new” or very lightly used.
Physical Deformation Check
Upon examining the angles in the visuals, no cracks, breaks, deep scratches, or impact-related dents are present on the device. No scratch reflection that would obstruct the view is visible on the plexi/glass surface of the screen. No deformation has been observed on the battery compartment cover or the locking mechanism.
Technical Specifications and Indicator Analysis
The vertical indicator panel on the front face of the device displays the measured intraocular pressure value (in mmHg). The scales on the indicator are as follows:
- Numerical Values: 5, 7, 10, 14, 18, 21, 24, 27, 30, 35, 40, 50
- Warning LEDs: PROBE, POSITION, REPEAT, CLEAN
This interface indicates that the device does not have an LCD screen but rather displays the measurement result with LED lights. Warnings such as “CLEAN” and “CHANGE” remind the user of maintenance time.
Label and Certification Information
The silver-colored label on the side surface of the device contains the following information:
- Symbols: Consult instructions for use symbol (letter i), Type BF applied part symbol (stick figure icon).
- CE Mark: CE 0044 (Along with the notified body number, compliance with medical device regulations).
- Reference: Type TA02
- Company: Icare Finland Oy
Battery and Power Status
2 batteries are visible inside the case. Although 4 battery symbols appear on the device’s side label, its physical size and equivalents typically operate with 2 AA or CR123 batteries (the batteries in the visual resemble AA size). No leakage, corrosion, or deformation is observed in the batteries. The device is suitable for wireless use.
Connection Ports
There is one “Mini-USB” port on the lower side of the device. The port’s input is clean; no dust accumulation or bent pins have been observed inside. This port is typically used for data transfer.
Current Fault and Risk Analysis
There are no indications in the visuals suggesting that the device is not working or is faulty (e.g., error codes on the screen, broken parts, corrosion). However, the front area where the probe (needle) is inserted has a very sensitive mechanical structure; although this hole appears clean in the visual, its mechanical functionality should be tested. As a potential risk, it is vital that the probe tip housing remains dust-free; it appears clean in the visual.
Conclusion
The examined Icare ONE Type TA02 device is a complete set with all its accessories. It is cosmetically flawless, free from deformation, and original. It has been manufactured in Finland in compliance with medical standards (CE 0044). Its physical condition indicates that the product has been used or stored very carefully.