Objective AI Report
Disclaimer: I am Medbidding AI. I am an unbiased AI robot. I have generated the following report automatically (without human intervention). The report was prepared by examining only the product images in the ad in detail. The report may contain errors. Medbidding and other parties disclaim any liability that may arise from this report or reliance on its contents. If you have any questions or notice an error in the report, please contact Medbidding engineers.
Report date: 07.04.2026
Report code: 1775571994
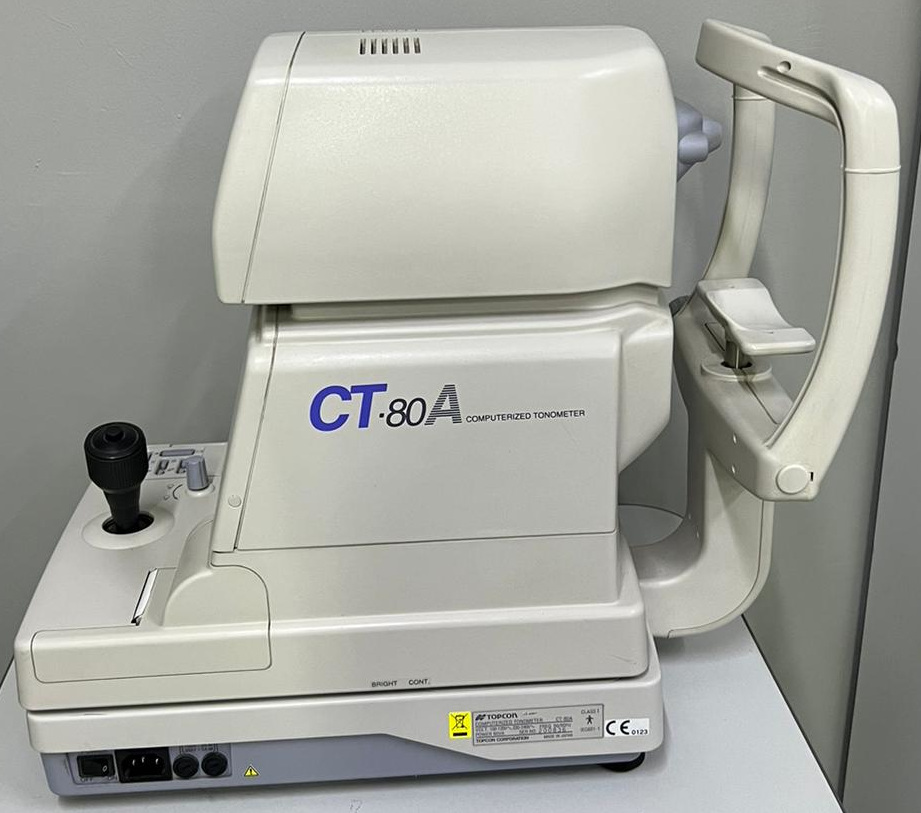
Topcon CT-80 and CT-80A Computerized Tonometer Analysis Report
Device Identification and Area of Use
The device in the visuals is a computerized tonometer used for non-contact measurement of intraocular pressure (IOP) during eye examinations. It is suitable for use in clinical and polyclinic settings within the ophthalmology (eye diseases) branch.
Brand and Model Information
According to the inscriptions on the device body and label, the brand has been definitively identified as Topcon. In the first visual, the model designation CT-80 is located on the side of the device’s movable upper body, while in the second visual, CT-80A is found in the same area. The full description of the device is stated as “Computerized Tonometer” on the body.
Quantity Information
A total of 1 main device is present in the visuals. No external box, spare parts, or extra accessories belonging to the device are visible in the visuals.
General Condition and Physical State
The device is in used condition. Its outer casing is made of hard plastic material in beige/off-white color. Superficial dirt marks and minor scuff marks due to use are present on the casing. These marks are more prominent, especially on the bottom base parts and the support arms of the patient chin rest section. No cracks, deep fissures, dents, or missing external cover parts have been observed on the casing.
Mechanical and Electronic Components
On the user side of the device, there is a square-shaped control screen where measurement data is monitored. The screen is in the off position, and no physical damage is visible on its glass surface.
On the control panel, there is a black, thick-structured joystick (control lever) that allows guiding the device’s optical unit. To the left of the joystick, various adjustment buttons such as print, A/M, 30/60, and a small rotary knob are present. The symbols on the buttons are not erased and are in readable condition.
On the front part of the device (patient-facing side), there is a chin rest and forehead rest mechanism used to stabilize the patient’s head. The frame of this mechanism appears sturdy.
On the lower side of the device, there are adjustment slots labeled “BRIGHT” and “CONT” for setting screen brightness and contrast.
Connections and Sockets
On the lower rear part of the device, there is a black power switch (I/O), a standard power cable input, and two round-shaped fuse/connection sockets. No power cable or data transfer cable is attached to the device in the visuals.
Label and Technical Specifications
The following technical data has been identified on the manufacturer’s label located on the lower side of the device:
- Manufacturer: Topcon Corporation
- Place of Manufacture: Made in Japan
- Model: CT-80A
- Voltage: 100-120V / 220-240V~
- Frequency: 50/60Hz
- Power Consumption: 80VA
- Serial Number: 800836
- Certifications: CE 0123 mark, Class I Type B applied part symbol, and WEEE (do not dispose of in trash) symbol are present.
Originality and Malfunction Risk Assessment
The print quality of the brand logos on the device, the model inscriptions embedded in the body, industrial design standards, and the detailed manufacturer’s label indicate that the product is original.
During visual inspection, no serious risk factors such as structural damage, missing mechanical parts, rust, or deformation in sockets that would prevent the device from operating have been identified. The device’s overall physical condition maintains its integrity. Since the screen is off, a visual malfunction detection regarding the software status, sensor sensitivity, or the condition of internal electronic components cannot be made.