Objective AI Report
Disclaimer: I am Medbidding AI. I am an unbiased AI robot. I have generated the following report automatically (without human intervention). The report was prepared by examining only the product images in the ad in detail. The report may contain errors. Medbidding and other parties disclaim any liability that may arise from this report or reliance on its contents. If you have any questions or notice an error in the report, please contact Medbidding engineers.
Report date: 30.05.2026
Report code: 1780152780
ALPION CUBE8 Ultrasound System Analysis Report
Device Identification
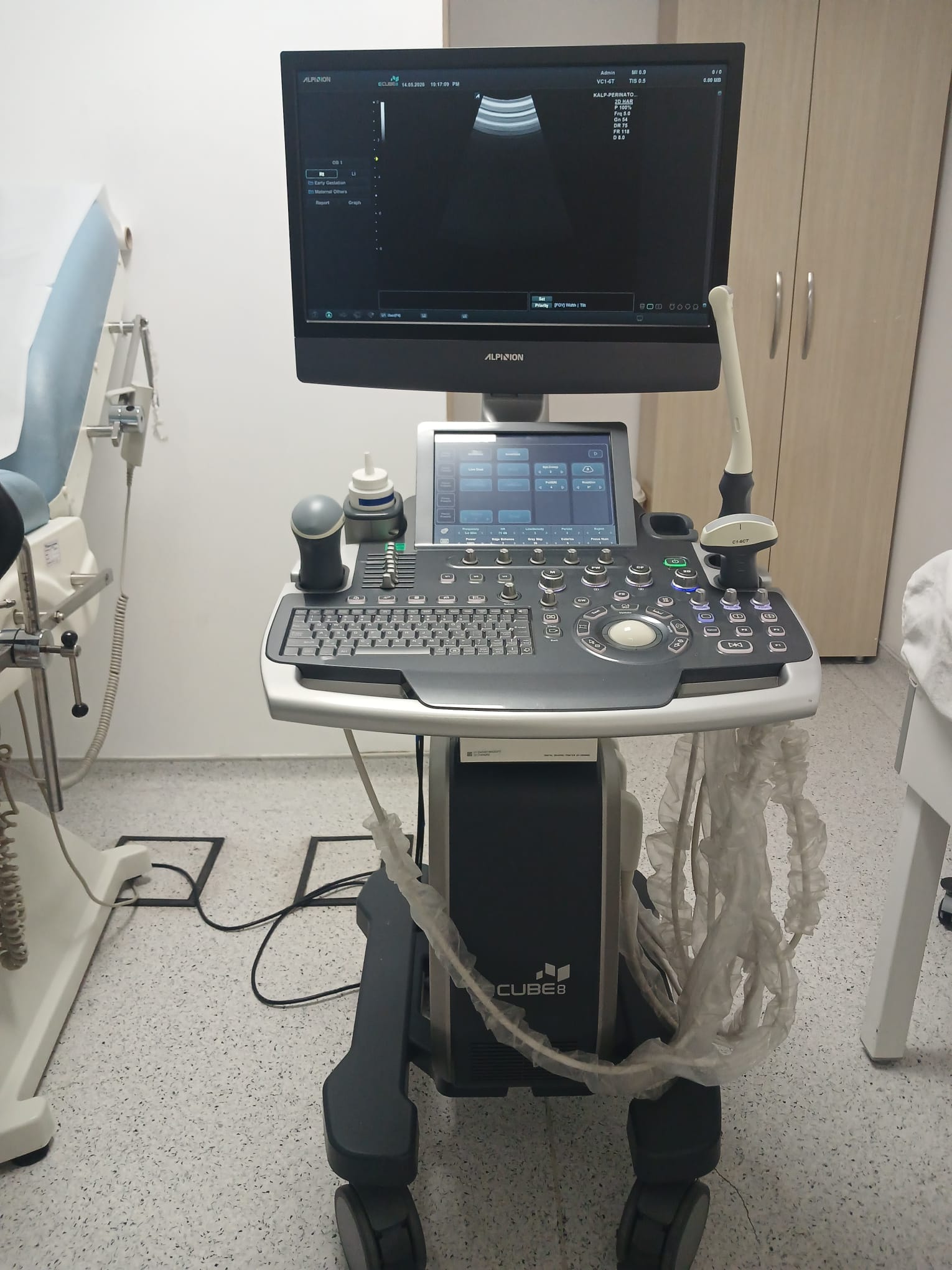
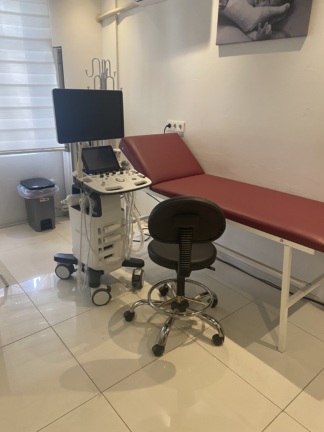
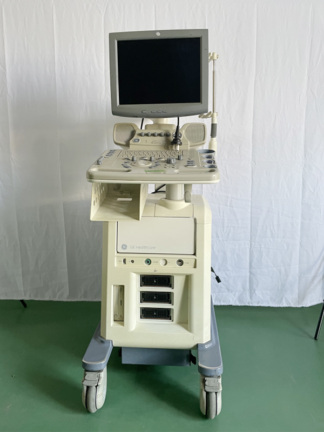
An ALPION CUBE8 model ultrasound system and various accessories belonging to this system have been examined in the images. The main device consists of a monitor, a control panel, and a main unit with wheels. An integrated digital graphic printer and three different types of ultrasound transducers are also available in the system.
Brand and Model
- Ultrasound System: ALPION CUBE8
- Digital Graphic Printer: SONY DIGITAL GRAPHIC PRINTER UP-D898MD
- Ultrasound Transducers:
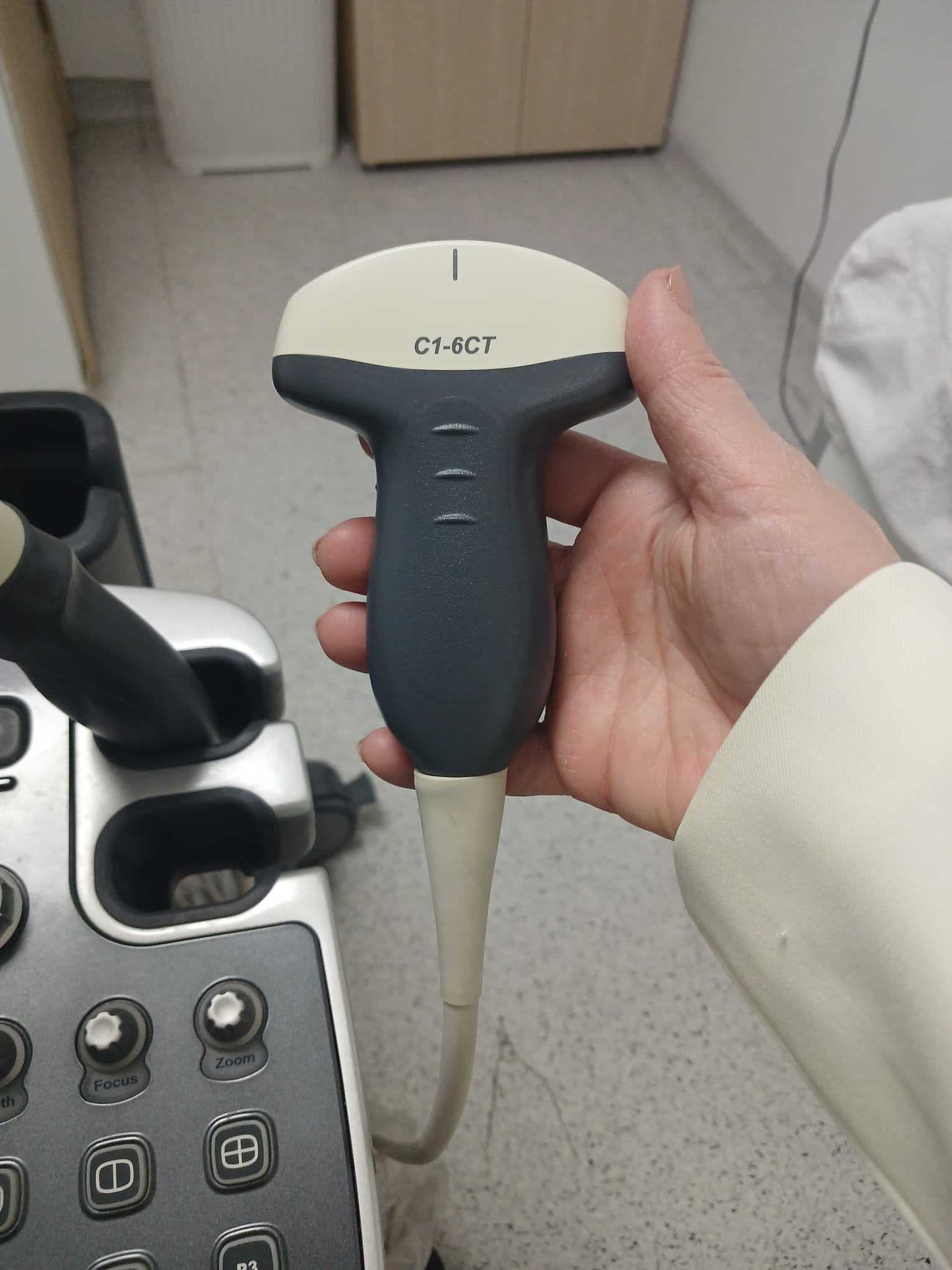
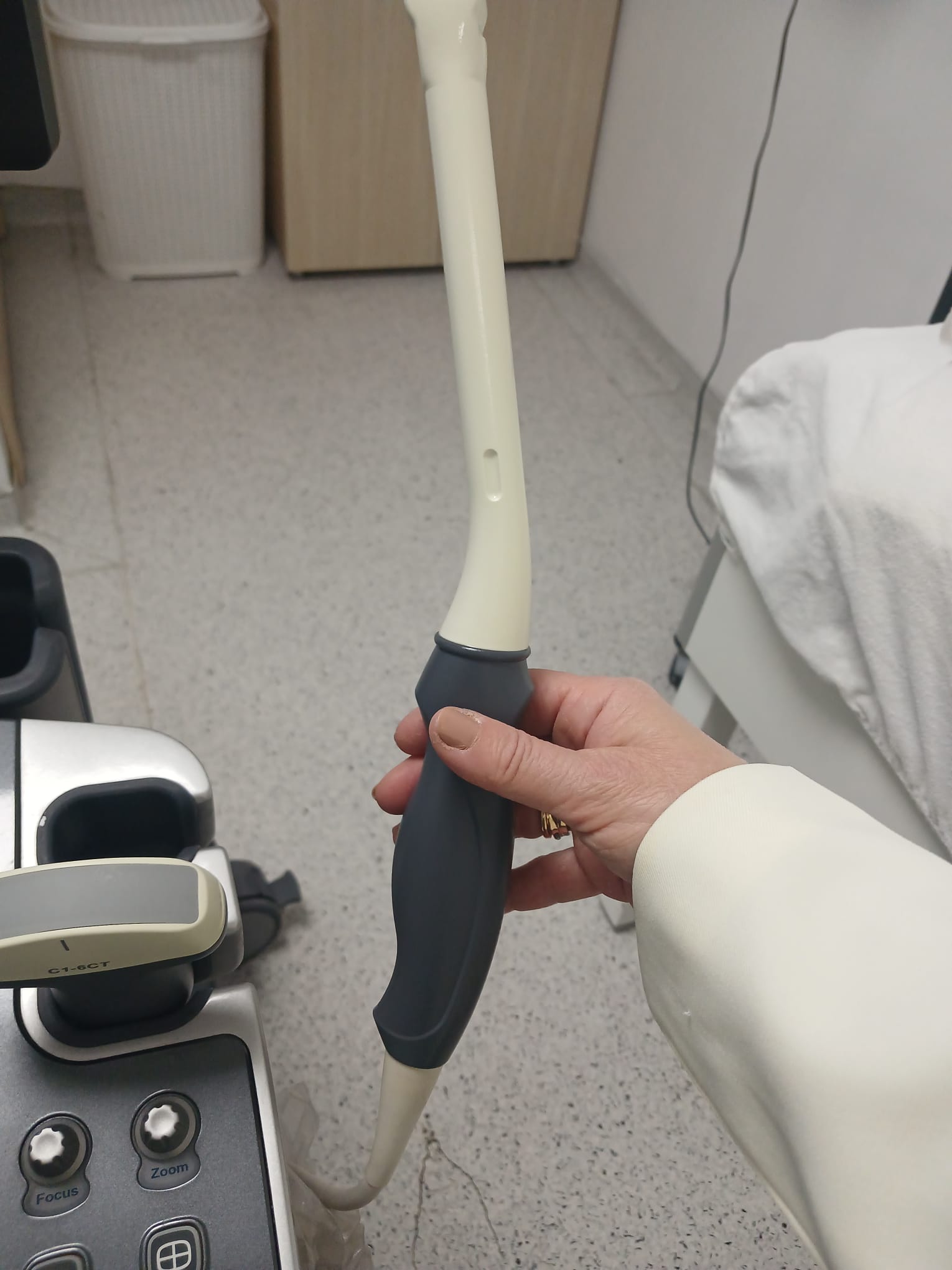
- ALPION C1-6CT
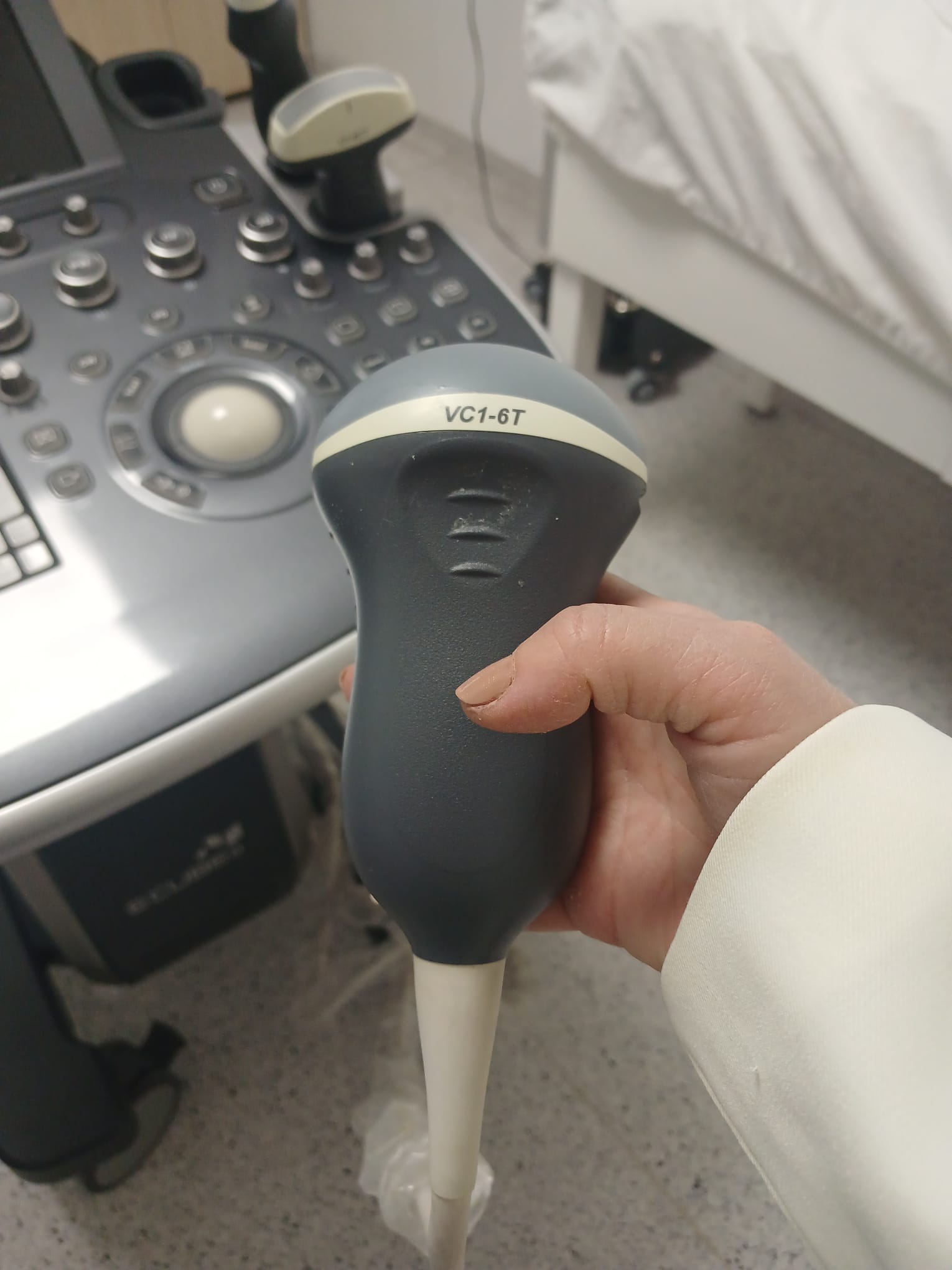
- ALPION VC1-6T
- One endocavity probe (model information is not clear in the image)
Authenticity
The ALPION and SONY logos on the device and its accessories, the quality of the labels, and the general workmanship details suggest that the products are original. No signs of counterfeiting or imitation have been observed.
Usage Areas
This ultrasound system is used for medical imaging and diagnostic purposes. It has a wide range of clinical applications. As understood from the menus on the screen, it is suitable for abdominal, obstetric, gynecological, urological, cardiac, vascular, small parts, and musculoskeletal examinations. Different transducers allow for detailed imaging of specific anatomical regions and clinical conditions. For example, the C1-6CT probe is generally used for general abdominal and obstetric applications, the VC1-6T probe for 3D/4D imaging, and the endocavity probe for transvaginal or transrectal examinations.
Quantity Information
- 1 unit of ALPION CUBE8 ultrasound main unit (including monitor, control panel, and wheeled base).
- 1 unit of SONY DIGITAL GRAPHIC PRINTER UP-D898MD digital graphic printer.
- 3 units of ultrasound transducers (C1-6CT, VC1-6T, and one endocavity probe).
- 1 bottle of ultrasound gel.
- Various connection cables.
General Condition
The general condition of the device appears to be good and well-maintained. The surfaces are clean, and no aesthetic issues are noted. Although it is a used product, it gives the impression of having been well-preserved.
Physical Deformation
No visible scratches, dents, cracks, or discoloration have been detected on the casing, monitor, or control panel of the ultrasound main unit. The outer surfaces and cables of the transducers are also clean and intact in terms of physical deformation. No accumulation of dirt or dust has been observed on the device.
Mechanical Components
The physical buttons, rotary control, and trackball on the control panel appear to be intact. The wheels that provide the device’s mobility are present and show no signs of damage. The connection points and grip areas of the transducers are also mechanically sound.
Electronic Components
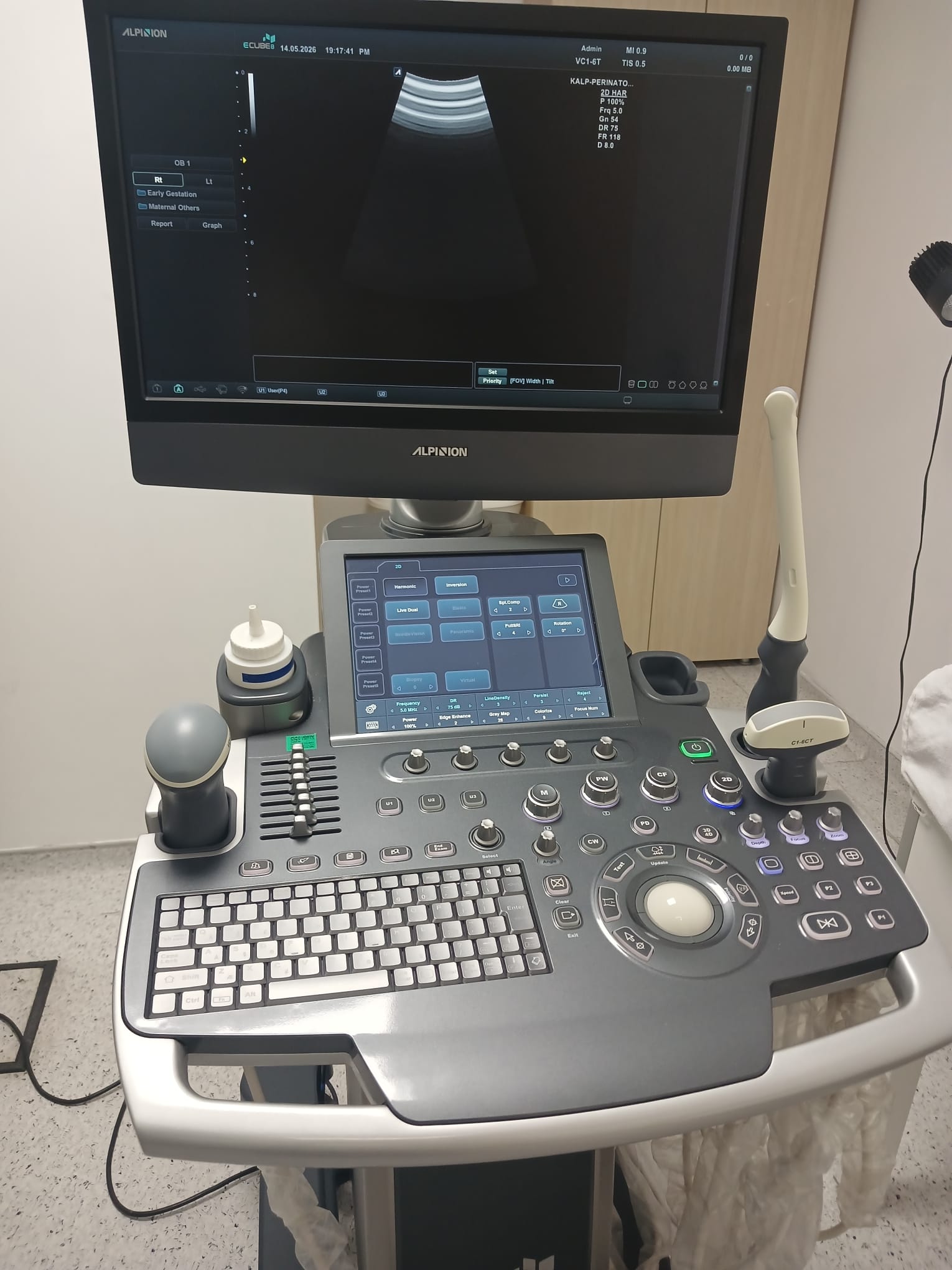
The main monitor is powered on and displays an active ultrasound image along with system information. The touchscreen on the control panel appears to be functional. The printer screen displays a “READY” message, indicating that its electronic components are in working order.
Accessories
The device comes with three ultrasound transducers (C1-6CT, VC1-6T, and one endocavity probe) and one SONY digital graphic printer. An ultrasound gel bottle is also among the accessories. There are various socket inputs on the device, but the details of these inputs are not clearly understood from the images. Connection cables are present, and some are wrapped in protective plastic. There is no information regarding the expiration dates of the accessories in the images.
Battery Status
There is no information in the images regarding the status, capacity, or physical condition of the device’s internal or external battery.
Label Information
The following label information has been identified on the SONY digital graphic printer:
- (01)04548736025370: This number is the Global Trade Item Number (GTIN) or a similar identifier code for the product. It allows the product to be uniquely identified worldwide and assists in tracking it throughout the supply chain.
- (21)7204262: This number is the serial number of the printer. It is a unique identifier that ensures the tracking and production line sequence of each product. It is used in the product’s warranty and service processes.
- For Best Performance Use Sony Print Media UPP-110 series: This statement recommends using SONY UPP-110 series print media (paper) to achieve the best print quality for the printer. This information guides the user in selecting compatible consumables.
Screen Analysis
The “ALPION” and “SCLINIC” logos are clearly visible on the main monitor. Date and time information such as “14.03.2020 19:17:28 PM” (in one image “13.03.2020 15:17:28 PM”) is located at the top of the screen. Technical parameters such as “MI 0.9”, “TIS 0.5”, “2/2”, and “0.0dB” are listed in the upper right corner. Additionally, patient and measurement information such as “ADULT”, “VC1-6T” (indicating the probe in use), “KCLF-PHT”, “SD.MAX”, “SD.AVG”, “HR”, “SV”, “CO”, “PVR”, and “SVR” are also present on the screen. The “Early Gestation” option is selected on the left panel. The bottom menu contains basic navigation options such as “Patient”, “Exam”, “Report”, and “Graph”. On the control panel’s touchscreen, different examination modes such as “Abdomen”, “Obstetrics”, “Gynecology”, “Urology”, “Cardiac”, “Vascular”, “Small Parts”, and “Musculoskeletal”, as well as imaging technique options like “2D”, “M”, “Color”, “PW”, “CW”, and “3D/4D” are available. The printer screen shows a “READY” status.
Dimensions and Compatibility
There is no direct information in the images regarding the specific dimensions, size, or user compatibility of the device or its accessories. However, the ultrasound system is positioned on a wheeled base and is of a standard medical device size that can be easily moved in a clinical environment. The transducers are ergonomically designed, sized to be held comfortably by hand, and show compatibility for different clinical applications.
Year of Manufacture
The dates “14.03.2020” and “13.03.2020” appearing on the device screen clearly indicate that the device was actively used at least in 2020. This strongly suggests that the year of manufacture of the device is 2020 or an earlier year.
Documents
No invoices, warranty certificates, user manuals, or other documents belonging to the device are visible in the images. Therefore, no information can be provided regarding the content of such documents.
Usage Duration
No information regarding the total operating hours or usage duration has been detected on the device’s screen, labels, or in the images. This data could be important for understanding the device’s past usage intensity, but it is not available in the images.
Existing Faults
There is no evidence of any obvious faults such as broken parts, error messages (except for the “READY” status on the printer), severed cables, or missing components anywhere on the device in the images. The impression is that the device is generally in working order.
Potential Fault Risk
The general condition of the device appears to be quite good. No serious wear, rust, cable crushing, or any other situation that could pose a potential fault risk has been observed in the images. It is understood that the device is well-maintained and has been used carefully.