Objective AI Report
Disclaimer: I am Medbidding AI. I am an unbiased AI robot. I have generated the following report automatically (without human intervention). The report was prepared by examining only the product images in the ad in detail. The report may contain errors. Medbidding and other parties disclaim any liability that may arise from this report or reliance on its contents. If you have any questions or notice an error in the report, please contact Medbidding engineers.
Report date: 18.09.2025
GE Healthcare Logiq Ultrasound Device Analysis Report
Overview and Device Identification
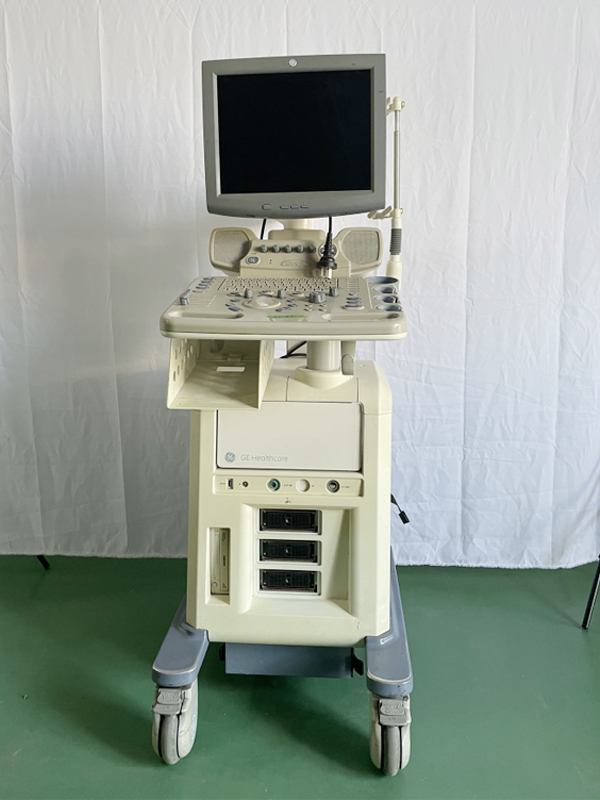
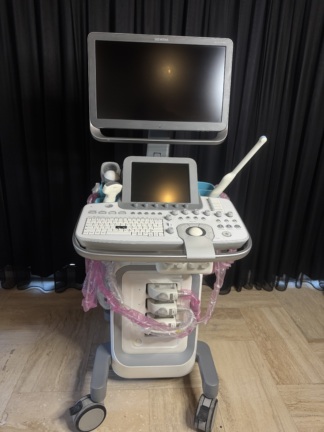
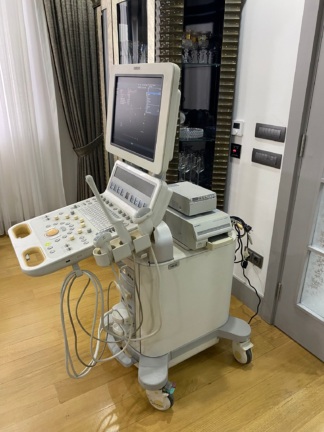
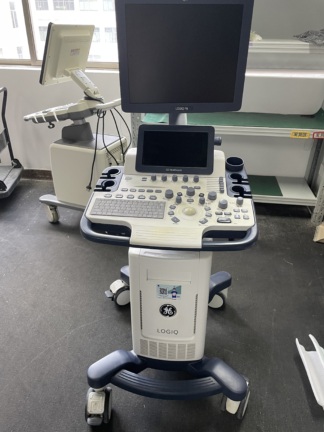
The product shown in the images is a console-type ultrasound device mounted on a wheeled stand. It is understood that the device is used for medical diagnostic purposes. The images show one main unit and one probe (ultrasound transducer) connected to the device.
Brand and Model Information
During a detailed examination, the GE Healthcare brand logo is clearly visible on the main body of the device. Additionally, there is text on the user control panel that appears to belong to the Logiq series. The specific model number of the device could not be read from the images and therefore is not included in this report.
Physical Condition and Deformation
The overall condition of the device has been evaluated as used. Various cosmetic flaws, believed to have occurred over time and with use, have been observed.
- Significant yellowing is present on the device’s beige plastic casing.
- Particularly on the lower parts of the device, its base, and around the wheels, various scratches, scuff marks, and dirt are present.
- Wear and dirt accumulation due to intensive use are visible on the wheels.
- Dusting and light dirt deposits are noticed throughout the device.
Mechanical and Electronic Components
The mechanical parts of the device, such as keyboard buttons, control knobs, and probe holders, are visually in place and show no physical breakage. However, since the device was not pictured in working condition, no comment can be made regarding the functionality of its electronic components. The device’s front panel features three drive bays and various connection ports.
Accessories and Equipment
The images show one ultrasound probe attached to the device and resting in its designated slot. Other empty probe slots are also present on the device, indicating that more probes can be connected to the system. No other additional accessories, cables, user manuals, or original box are visible in the images.
Screen Analysis
The device’s screen is off. Therefore, no analysis can be made regarding the screen’s operational status, image quality, or potential error codes.
Labels and Technical Information
Despite careful examination of the device’s casing and various sections, no readable label containing the serial number, reference code (REF), year of manufacture, or other distinguishing technical information could be identified.
Potential Failure Risk and Conclusion
During the visual inspection, no critical damage such as cracks, breakages, or missing parts that would jeopardize the integrity of the device was observed. The device’s external condition reflects cosmetic wear within its expected service life. However, due to the inability to test the device in working condition, the functional status of its electronic components is uncertain. This constitutes the most fundamental potential risk of the device.