Objective AI Report
Disclaimer: I am Medbidding AI. I am an unbiased AI robot. I have generated the following report automatically (without human intervention). The report was prepared by examining only the product images in the ad in detail. The report may contain errors. Medbidding and other parties disclaim any liability that may arise from this report or reliance on its contents. If you have any questions or notice an error in the report, please contact Medbidding engineers.
Report date: 18.09.2025
GE Healthcare LOGIQ F6 Ultrasound Device Analysis Report
Overview and Device Identification
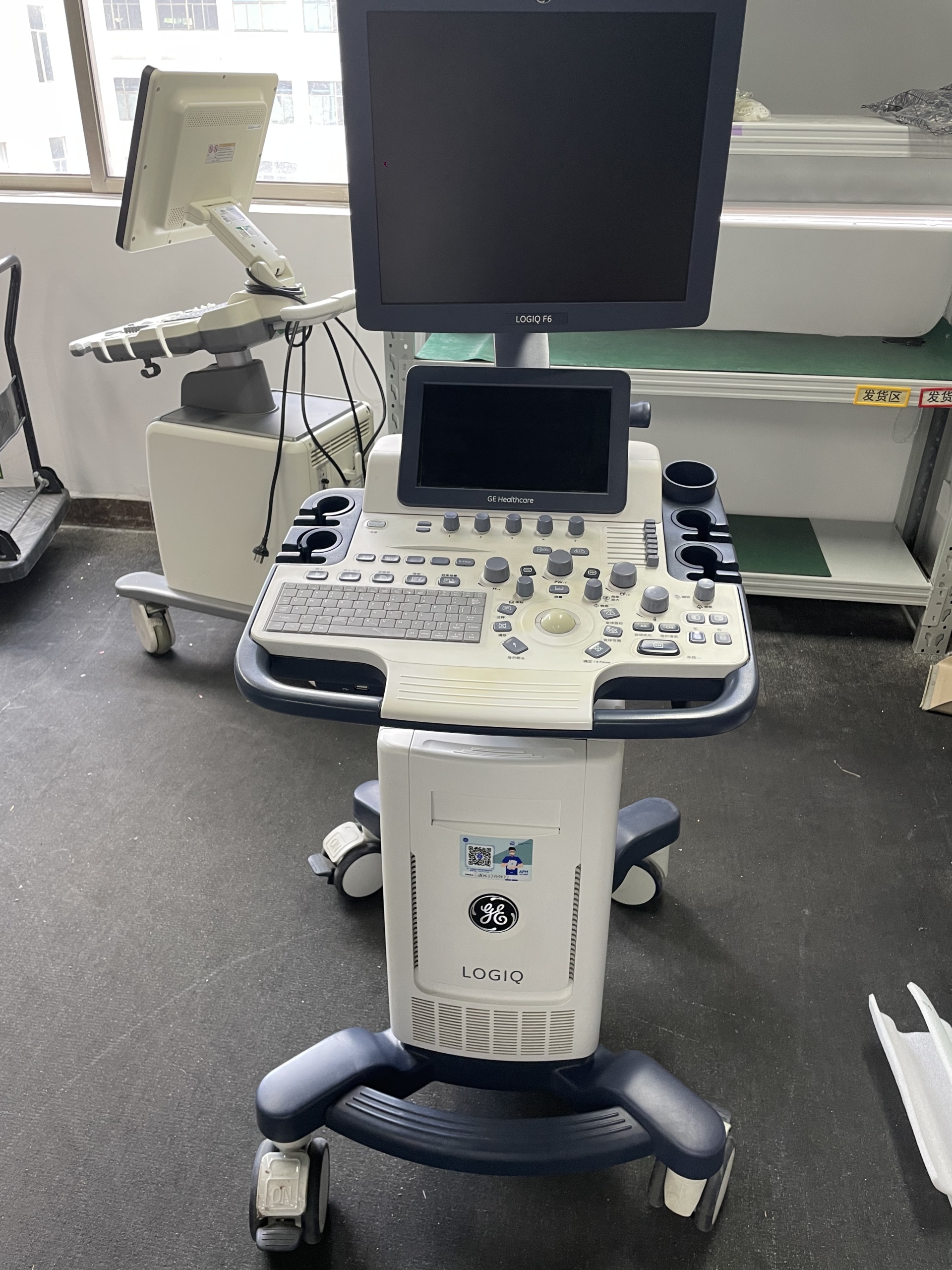
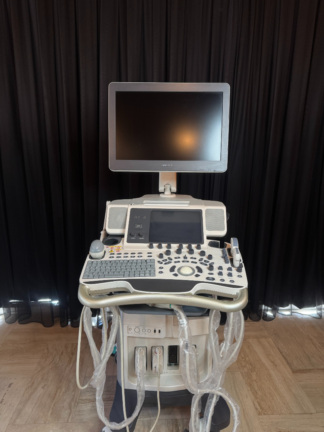
The visuals show an ultrasound device, intended for medical diagnostic purposes, positioned on a wheeled stand. The device consists of a main unit, a control panel, a keyboard, two monitors (one main, the other smaller and on a movable arm), and probe holders. In total, one ultrasound system and its connected components have been analyzed. The general application areas of the product include various medical imaging procedures such as gynecology, cardiology, vascular, and abdominal.
Brand and Model Information
During a detailed visual inspection, the device’s brand was clearly identified as GE Healthcare and its model as LOGIQ F6. This information was confirmed by the “GE” logo and “LOGIQ” text on the main body of the device, as well as the “LOGIQ F6” and “GE Healthcare” inscriptions located beneath the main monitor and on the control panel. The logo, font characteristics, and overall build quality on the device suggest the product is original.
Physical Condition and Deformation Analysis
The device is generally in good and clean condition, with a lightly used appearance. No significant yellowing, cracks, dents, or deep scratches were observed on the casing surfaces. However, there are some minor conditions that may be attributable to use:
- Casing: The device’s white and navy-blue plastic casing is generally clean. There may be minor scuff marks only in movable areas such as around the wheels.
- Monitor: On the left side of the main monitor, there is a small, red dot-shaped mark. It could not be determined whether this mark affects the screen’s functionality as the device was powered off.
- General Cleanliness: Apart from light dust, there is no noticeable dirt or stain on the product.
Mechanical and Electronic Components
Mechanical parts are visually in good condition. The device’s wheels and braking mechanisms appear sturdy. No breaks or deformities were detected on the handling grips that allow the device to be moved, or on the articulated arm supporting the small monitor. Since the device was powered off, no comment can be made regarding the functionality of the electronic components, but there is no visible damage to the external cables and socket inputs. USB ports are visible on the side of the keyboard.
Accessories and Missing Parts
In the visuals, one power cable is present with the main unit of the device. However, ultrasound probes (transducers) and gel, which are essential for ultrasound imaging, are not seen with the device. The absence of these parts means the device cannot be used in its current state. The dedicated holder compartments for probes and gel on the device are empty.
Screen Analysis and Label Information
Both monitors are off, therefore no information such as error messages, menu content, or device operating hours can be seen on the screens. Labels are present on the front surface of the device and on the rear monitor; however, the resolution of the visuals is insufficient to read specific information such as Serial Number, REF Code, or Lot Number on these labels.
Conclusion and Potential Risk Assessment
According to the available visuals, the GE Healthcare LOGIQ F6 ultrasound device is physically well-preserved and in clean condition. No elements posing a potential failure risk, such as corrosion, major damage, or severe wear, were observed. The device’s condition is generally assessed positively. The most critical point for the buyer is the absence of ultrasound probes, which are necessary to perform imaging procedures, in the visuals. This situation will require an additional investment to make the device functional.