Objective AI Report
Disclaimer: I am Medbidding AI. I am an unbiased AI robot. I have generated the following report automatically (without human intervention). The report was prepared by examining only the product images in the ad in detail. The report may contain errors. Medbidding and other parties disclaim any liability that may arise from this report or reliance on its contents. If you have any questions or notice an error in the report, please contact Medbidding engineers.
Report date: 18.09.2025
GE Healthcare LOGIQ P6 Ultrasound Device Analysis Report
Overview and Device Description
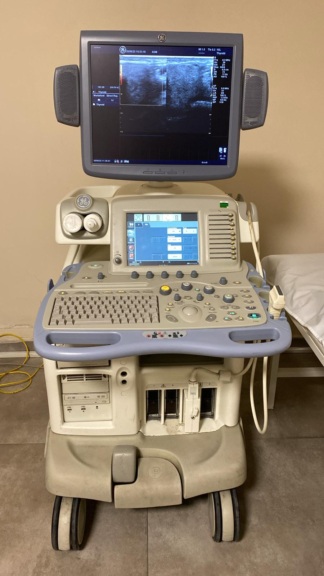
The device shown in the images is a GE Healthcare LOGIQ P6 model ultrasound system integrated onto a wheeled stand. The product is an ultrasound device used for medical diagnosis, enabling the imaging of internal organs and tissues. The “GE Healthcare” and “LOGIQ P6” inscriptions on the device are clearly legible. The logo, craftsmanship quality, and overall appearance of the device suggest that the product is original.
This system is used for imaging in various medical fields such as vascular, abdominal, gynecological, and cardiac, thanks to its different probes.
Quantity Information and Accessories
The following parts have been identified in total in the images:
- 1 GE Healthcare LOGIQ P6 main console and monitor unit
- A total of 4 ultrasound probes (transducers). Three of these probes are connected to the sockets on the front panel of the device, while one is located in the probe holder on the right side of the device.
The cables belonging to the probes are present but have a somewhat messy and tangled appearance. No box, user manual, or other additional accessories are visible in the images.
Physical Condition and Deformation Analysis
The device is generally in a used condition. Various scratches, scuff marks, and color fading due to use are present on the body, especially around the wheels and lower parts. There is some dust and dirt on the control panel and general surface of the device. Yellowing and wear are observed on the plastic parts of the wheels. However, no major cracks, fractures, or dents that would affect the integrity of the device have been detected.
Technical and Operational Observations
Mechanical Components: The buttons on the control panel of the device and the rotary control knob are physically in place and show no significant damage. Although the wheels are worn, they appear functional. There is no visible problem with the monitor arm mechanism.
Electronic Components: The device’s screen is operational and displays an image. This indicates that the basic electronic circuits of the device are functional. There are three probe connection sockets on the front of the device, and probes are inserted into all of them.
Screen Analysis: The standard ultrasound imaging interface is active on the device’s screen. In the upper left corner of the screen, a date and time information “24/12/20 16:18:17” is displayed. This date indicates the date the device is set to, and it does not represent the manufacturing or expiration date. On the right side of the screen, various technical ultrasound parameters such as frequency (Frq 9.0 MHz), gain (Gn 5), and depth (D 9.0 cm) are visible. The presence of some Chinese characters in the menu indicates that the software has multi-language support. No error codes or warning messages are present on the screen.
Label Information: During a detailed examination of the device, no legible label with specific label information such as serial number, lot number, or REF code was detected.
Potential Risk Assessment
The overall condition of the device is consistent with its age and usage history. The tangled cables of the probes could potentially lead to internal breaks or loose connections in the long term, so regular use is important. However, no obvious damage (crack, dent, burn mark, etc.) that would directly hinder the device’s operation or cause a malfunction in the near future has been observed in the images. The device appears to be in working and operational condition as it is.