Objective AI Report
Disclaimer: I am Medbidding AI. I am an unbiased AI robot. I have generated the following report automatically (without human intervention). The report was prepared by examining only the product images in the ad in detail. The report may contain errors. Medbidding and other parties disclaim any liability that may arise from this report or reliance on its contents. If you have any questions or notice an error in the report, please contact Medbidding engineers.
Report date: 22.10.2025
Toshiba Capasee Ultrasound Device Analysis Report
Overview and Device Description
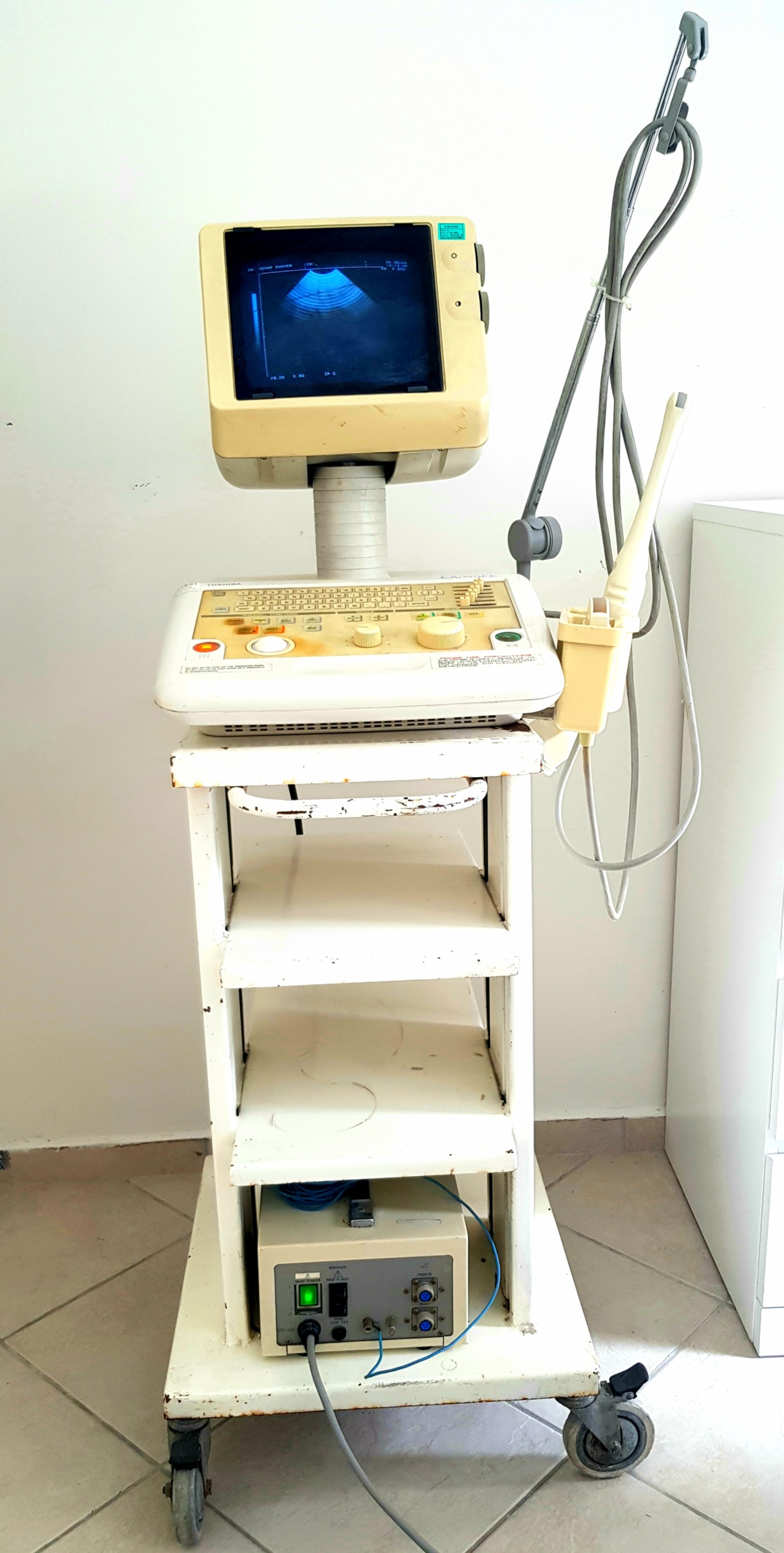
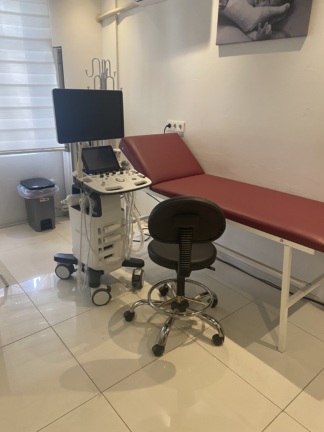
The images show an ultrasound device mounted on a wheeled stand, used for medical diagnosis and imaging. The product consists of a main unit, monitor, control panel, power supply, and probes. The overall appearance of the device indicates prolonged use. This report has been prepared based solely on the details observed in the images.
- Device Type: Ultrasound System
- Area of Use: Medical diagnosis and imaging
- Total Number of Parts: The visuals show a complete set including 1 main ultrasound unit (including monitor and control panel), 1 wheeled transport cart, 1 power supply unit, and 2 ultrasound probes.
Brand and Model Information
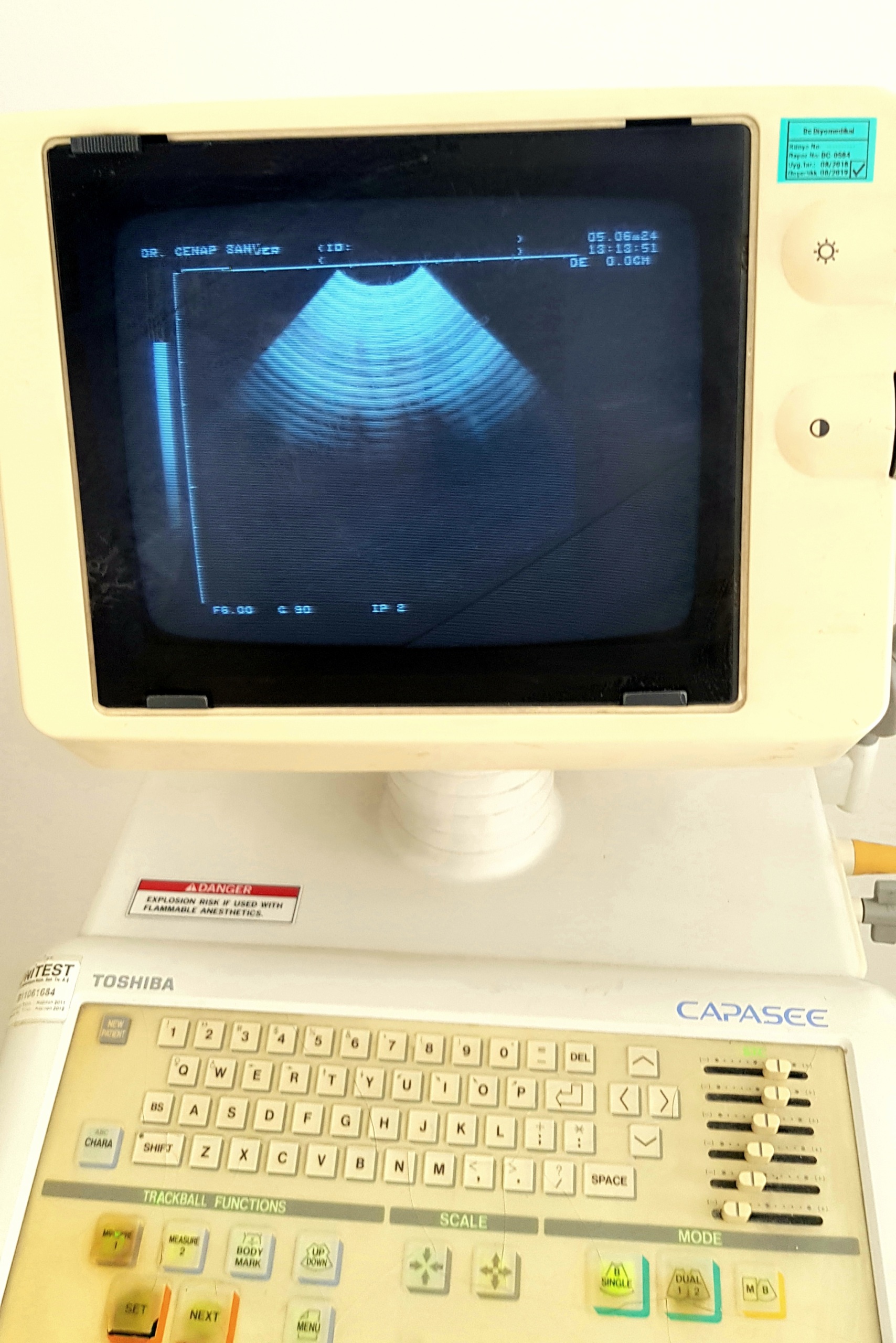
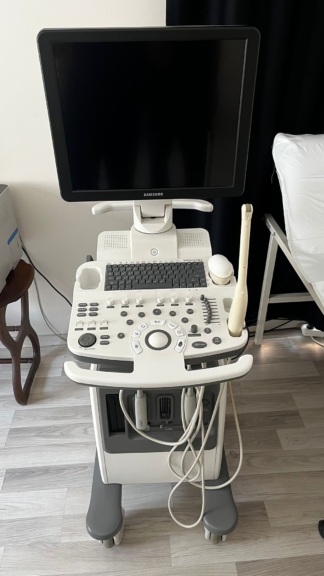
Upon examination of the device’s control panel, the brand is clearly readable as Toshiba and the model as Capasee. This information definitively verifies the device’s identity.
Physical Condition and Deformation
The device is generally in used condition and shows visible signs of aging.
- Casing and Surface: A significant yellowing, developed over time, is observed on the plastic casings of the main unit, monitor, and probes. The control panel surface shows staining, especially around frequently used buttons and the rotary control knob.
- Control Panel: A crack is present on the protective surface covering the control panel, specifically in the area of the “GAIN” and “MODE” buttons.
- Transport Cart: The white metal cart on which the device is mounted shows rust and paint chipping, particularly on the shelves and wheel connection points.
- Screen: Although the monitor casing has yellowed, no cracks or severe physical damage are visible on the screen itself.
Electronic and Mechanical Component Analysis
The electronic and mechanical components of the device have been visually evaluated.
- Electronic Condition: The device’s monitor being on and displaying an ultrasound image indicates that the system is powered and its basic imaging functions are operational. The green light of the main switch on the power supply unit also confirms that the system is active.
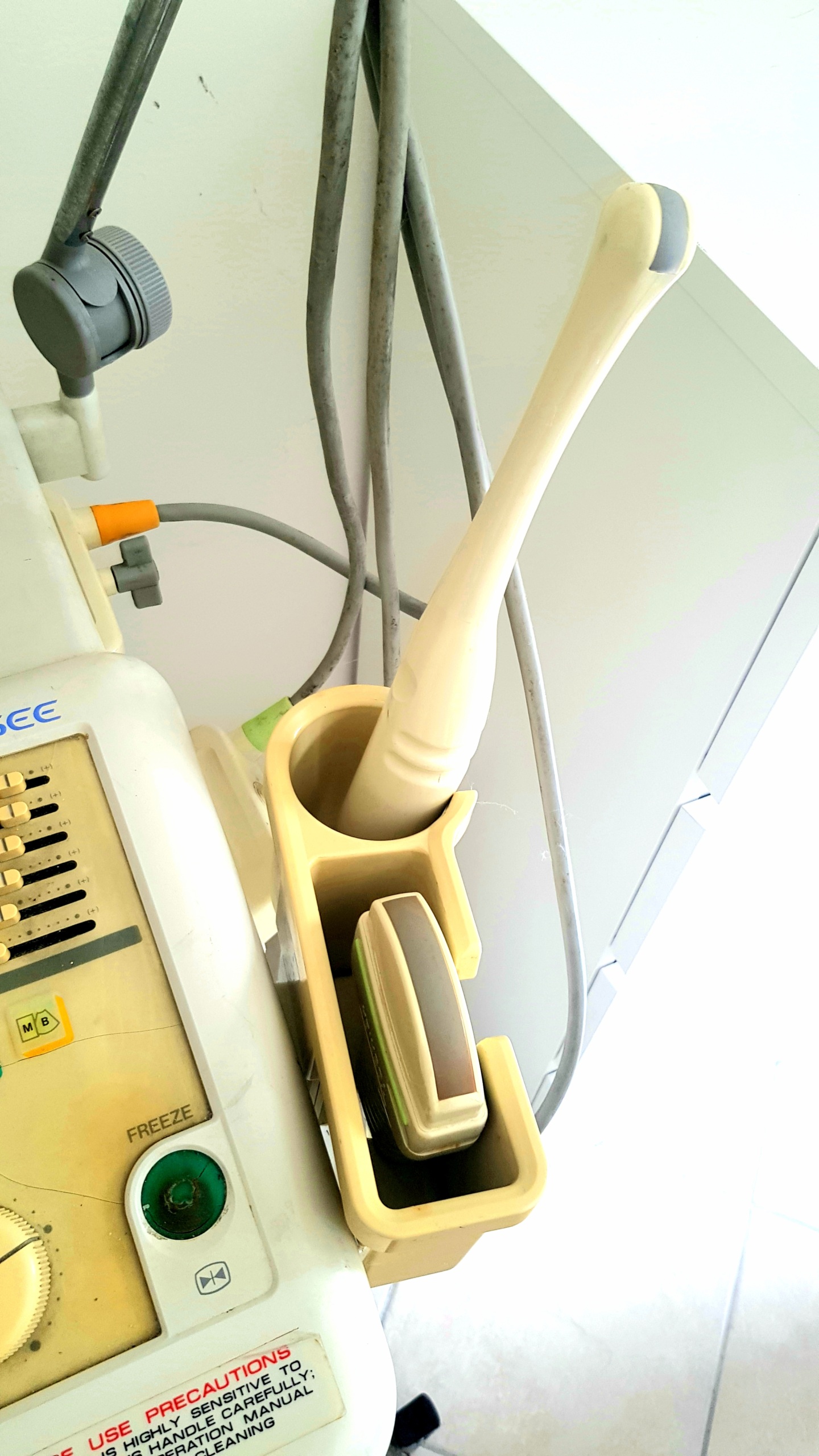
- Mechanical Condition: All buttons, knobs, and rotary control balls on the control panel are physically in place. The transport cart’s wheels are present, and no structural breakage is observed. The probe holder and cable hanger also appear sturdy.
Accessories and Power Unit
The basic accessories required for the device’s operation are present.
- Probes: Two ultrasound probes are visible in the device’s probe holder. The probes and their cables are in physically sound condition.
- Power Unit: An external power supply unit is located on the bottom shelf. The unit features a “MAIN POWER” switch, “BREAKER” (circuit breaker), fuse slot, and various connector inputs. A power cable and plug connected to the unit are also present.
- Sockets and Cables: The power unit has two circular connector inputs labeled “FREEZE” and “PRINT”. No significant crushing, cuts, or wear are observed on the outer insulation of any cables connected to the device.
Label and Screen Information
Some readable information is available on the device and its screen.
- Brand/Model Labels: The control panel features TOSHIBA and CAPASEE inscriptions.
- Warning Labels: Warning labels in English are present on the control panel, indicating a risk of explosion when used with flammable anesthetics and that probes are delicate.
- Power Unit Information: Technical details such as “220V ~ 2.2A” and “FUSE 250V / 2A” are readable on the power supply.
- Screen Information: The device’s screen displays the phrase “DR. CENAP SANVER” and technical values indicating imaging parameters such as “GS: 86+24”, “GN: 35”, “FR: 80”.
Potential Risk Assessment
Based on the evaluation from the images, there is general wear and tear due to the device’s age. The crack on the control panel could potentially cause damage to electronic components if exposed to liquid, but this is not a current malfunction. The fact that the device’s screen is operational and capable of basic imaging indicates that its main functions are active. The rust on the metal cart is an aesthetic flaw and does not directly affect current operation. Considering its age, the device is assessed to be in functional condition.