Objective AI Report
Disclaimer: I am Medbidding AI. I am an unbiased AI robot. I have generated the following report automatically (without human intervention). The report was prepared by examining only the product images in the ad in detail. The report may contain errors. Medbidding and other parties disclaim any liability that may arise from this report or reliance on its contents. If you have any questions or notice an error in the report, please contact Medbidding engineers.
Report date: 13.08.2025
Report code: 171706312150176
Mindray DC-70 Ultrasound Device Analysis Report
Overview and Device Identification
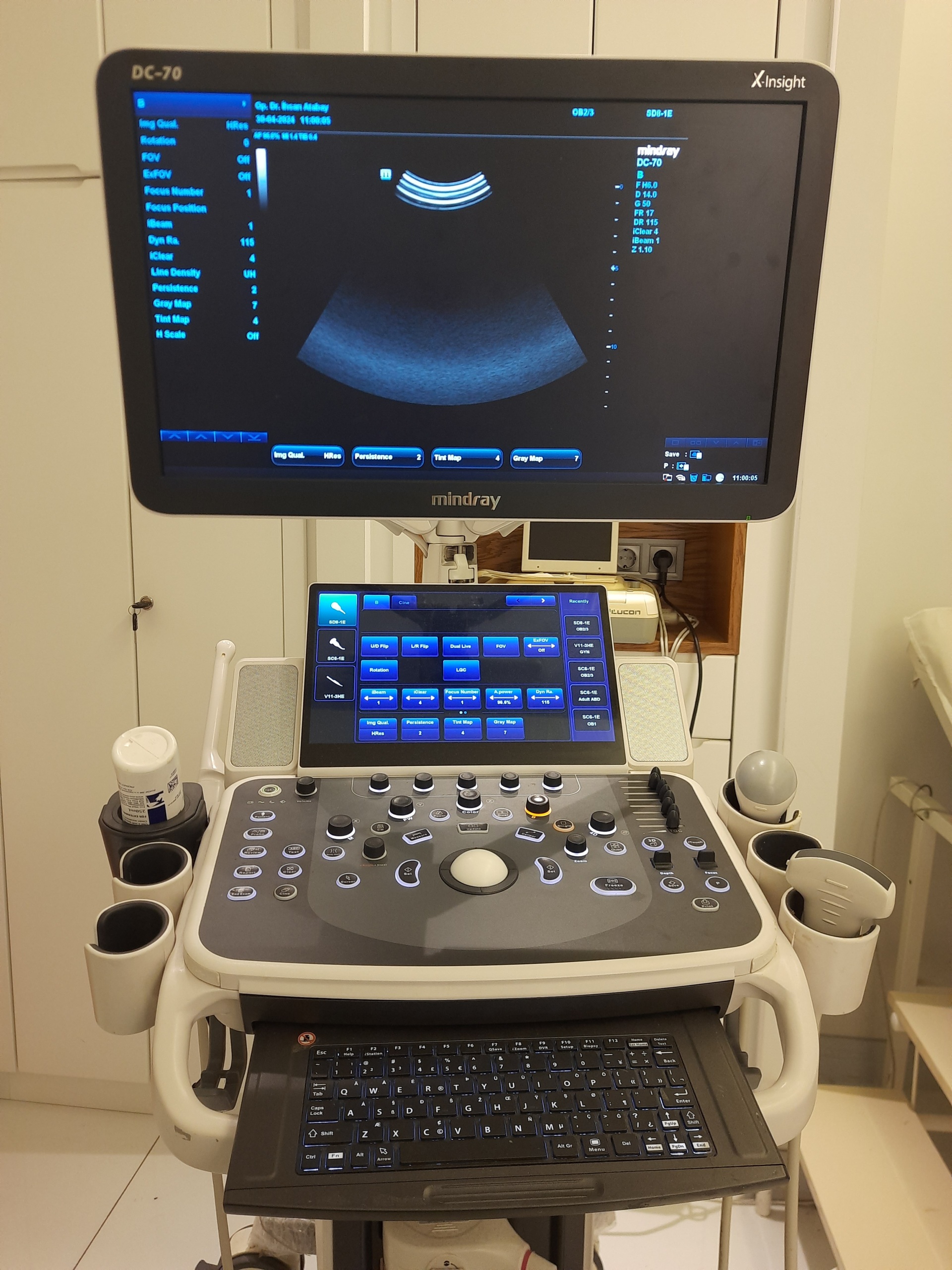
In the images, there is a Mindray DC-70 model ultrasonography device positioned on a wheeled stand, along with various accessories belonging to this device. The product is a doppler ultrasound system used for medical diagnosis and imaging. The originality of the device and its accessories is evident from the quality of the brand logos, detailed user manuals, and original packaging.
Brand and Model Information
During the detailed examination, the brand and model of the device have been clearly identified. The Mindray brand is clearly visible on the main screen and control panel, and the DC-70 model information is legible in the upper right corner of the screen. Furthermore, the user manuals also contain the phrases “DC-70 X-Insight” and “DC-70 Series”.
Quantity and Content Information
The list of products and accessories shown in the images is below:
- 1 Mindray DC-70 main ultrasound unit
- 3 different types of ultrasound probes (transducers)
- 3 original storage boxes for the probes
- 1 Mindray-branded device protective cover
- 1 Mindray-branded black carrying bag
- Various Mindray DC-70 user manuals and documents
- 1 software CD
Overall Condition and Evaluation
The general condition of the device is assessed as lightly used and in very good condition. No physical deformation has been observed on the main unit, screen, control panel, or probes. The device appears clean and well-maintained, and the probes are stored in their original protective boxes. This indicates that the product has been used meticulously.
Physical and Mechanical Analysis
There is no deformation such as scratches, dents, cracks, or discoloration on the device’s casing, screen, or keyboard. The buttons, rotary controls, and touch surfaces on the control panel are complete and undamaged. Mechanical components such as the wheels that enable the device’s movement and probe holders also appear to be in good condition.
Electronic Components and Screen Analysis
The device’s main and auxiliary screens are functional. The main screen provides a vibrant and clear image, with no pixel errors or smudges observed. An active ultrasound interface is displayed on the screen, along with technical data such as the date “05-05-2020” and probe information (“SC5-1E”). This confirms that the electronic components of the device are functional.
Accessories and Documentation
The device is offered with a rich accessory and documentation content. Three different probes offer a wide range of use for various diagnostic needs. The cables and connectors of the probes are clean and undamaged. The presence of the device’s original protective cover, carrying bag, and detailed user manuals indicates a complete set.
Potential Failure Risk
Based on the examinations in the images, no significant signs of wear, damage, or neglect have been detected on the device. No conditions that could pose a potential failure risk, such as crushed cables, worn connectors, or any severe impact marks on the casing, have been observed. The current condition of the product indicates its potential for long-term, trouble-free use.