Ready For Sale
Secondhand GE Logiq 200 Pro Ultrasound Machine
Price: USD$ 2.200,00 Approx: 99.000,00 TL
Ready For Sale
Ask a Question
Payment
No additional fees, full assurance. We provide complete financial and operational security in secondhand medical device trading. For this, we offer the "Secure Payment" service. This free service protects the rights of both parties by securing the buyer's money and the seller's product. The Secure Payment system is a standard assurance mechanism offered by Medbidding. For additional information, review the "services" page.
There is no cash on delivery order system on the Medbidding platform. For payments to be made by credit card, the product to be purchased must comply with this payment method. You can contact us to get information about this. We would be happy to assist you.
For payments made outside of Turkiye, you can choose bank transfer, credit card, Western Union or cryptocurrency options. Installment options are not currently available for credit cards other than Turkish banks.
Shipping
Standard Shipping Conditions
In order to ensure secure transactions on Medbidding, the shipping process is managed through four different scenarios depending on the location of the buyer and seller. Free shipping is available for some categories. The terms below apply to all categories unless otherwise stated.
Buyer and Seller in Turkiye
- Seller → Medbidding: The seller packages the product and sends it to the Medbidding operation center. The seller is responsible for this shipping cost.
- Medbidding → Buyer: The product, inspected by Medbidding engineers, is sent to the buyer's address. The buyer is responsible for this shipping cost.
Buyer Outside Turkiye, Seller in Turkiye
- Seller → Medbidding: The seller packages the product and sends it to the Medbidding operation center. The seller is responsible for this shipping cost.
- Medbidding → Buyer: The product, inspected by Medbidding engineers, is sent to the buyer's address. The buyer is responsible for this shipping cost.
Buyer in Turkiye, Seller Outside Turkiye
- Seller → Medbidding: The seller packages the product and sends it to the Medbidding operation center. The seller is responsible for this shipping cost.
- Medbidding → Buyer: The product, inspected by Medbidding engineers, is sent to the buyer's address. The buyer is responsible for this shipping cost.
Buyer and Seller Outside Turkiye
If there is a local operation center in the seller's country:
- Seller → Medbidding: The seller packages the product and sends it to the Medbidding operation center. The seller is responsible for this shipping cost.
- Medbidding → Buyer: The product, inspected by Medbidding engineers, is sent to the buyer's address. The buyer is responsible for this shipping cost.
If there is no operation center in the seller's country:
- Technical Inspection: Inspections are performed by our engineers via remote video call.
- Seller → Buyer: The seller packages the product and sends it directly to the buyer's address. The seller is responsible for this shipping cost.
Objective AI Report
Disclaimer: I am Medbidding AI. I am an unbiased AI robot. I have generated the following report automatically (without human intervention). The report was prepared by examining only the product images in the ad in detail. The report may contain errors. Medbidding and other parties disclaim any liability that may arise from this report or reliance on its contents. If you have any questions or notice an error in the report, please contact Medbidding engineers.
Report date: 18.02.2026
GE LOGIQ 200 PRO Series Ultrasound Device Analysis Report
Device Identification, Brand, and Model
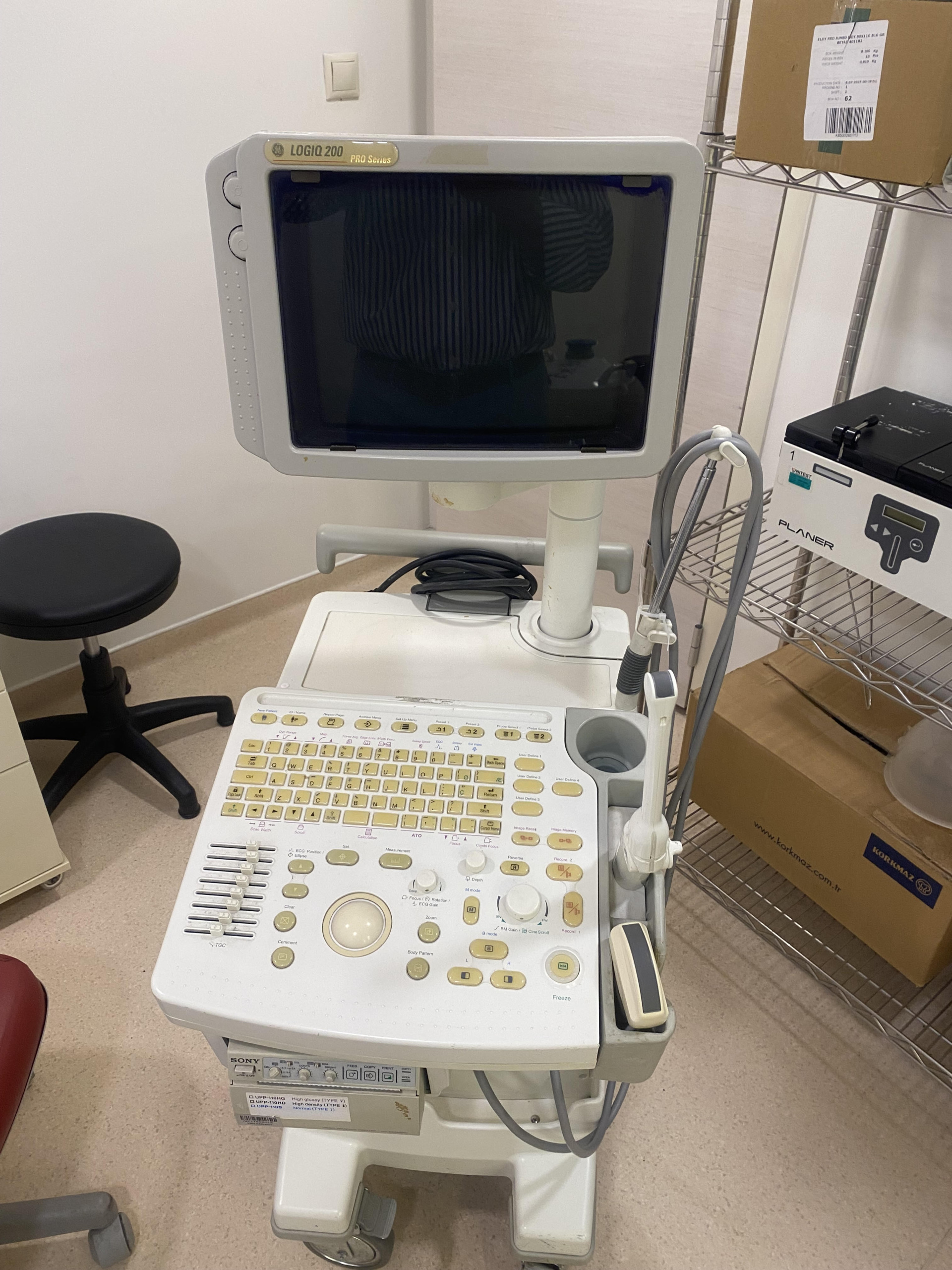
The product examined in the images is a mobile ultrasonography device used for medical imaging. The device’s brand has been identified as GE (General Electric), based on the “GE” logo located in the upper left corner of the monitor frame and the model name written next to it. The model information is clearly stated as LOGIQ 200 on the monitor frame. Additionally, to the right of the model name, the phrase PRO Series indicates the device’s specific series.
The brand of the video printer (thermal printer) integrated into the main body of the device, located below the control panel, has been identified as SONY. This is a peripheral unit included in the device’s hardware.
General Condition and Physical Deformation
Based on visual inspection, the device is in used condition and, considering its production year, features older technology (CRT monitor structure). The general condition and deformation analysis are as follows:
- Color Change (Yellowing): A noticeable age-related yellowing is observed on the white/beige plastic casing of the device, particularly on the monitor frame, keyboard panel, and probe holder sections. This is a typical ABS plastic oxidation indicating the device has been exposed to sunlight or the environment for a long time.
- Stains and Dirt: Brown stains or dried liquid residues are present on the monitor’s movable arm mechanism (as seen in Image 3). Although it cannot be definitively determined from the image whether these stains are rust or medical solution (e.g., povidone-iodine) residues, hygienic cleaning or maintenance is clearly required.
- Screen Condition: The screen surface of the CRT tube monitor appears physically intact, with no deep scratches or cracks visible.
- Keypad: The keys on the control panel are in place, but there are signs of color fading and dirt accumulation on their plastic parts due to use. No missing keys were detected visually.
Mechanical and Electronic Components
Visual findings regarding the device’s mechanical and electronic structure are as follows:
- Monitor Mechanism: A movable arm connects the monitor to the main unit. A “Push to Release Up/Down” warning is present at the connection point, indicating that the monitor’s height can be adjusted. However, the dirt/stain on the arm may require physical testing to determine if it affects its mobility.
- Wheels: The device appears to have a wheeled base (the front wheel guard is partially visible), but the integrity of all wheels cannot be fully confirmed visually.
- Cables: Cables are disorganized behind the monitor and at the probe connection point, and some cable organizers appear to be dysfunctional or not in use. However, no clear breaks or crushes are visible on the outer surfaces of the cables.
- Printer Unit: The SONY brand video printer appears with its paper tray closed and in an integrated state.
Accessories and Quantity Information
The components and accessories identified on the device are as follows:
- Main Unit (Console): 1 Unit GE LOGIQ 200 Ultrasound Device.
- Ultrasound Probe (Transducer): 1 unit probe is attached to the holder on the right side of the device (the probe type cannot be fully discerned from the image, but it is estimated to be convex or linear). Its cable is connected to the main unit.
- Printer: 1 Unit body-mounted Sony thermal printer.
Label Information and Originality
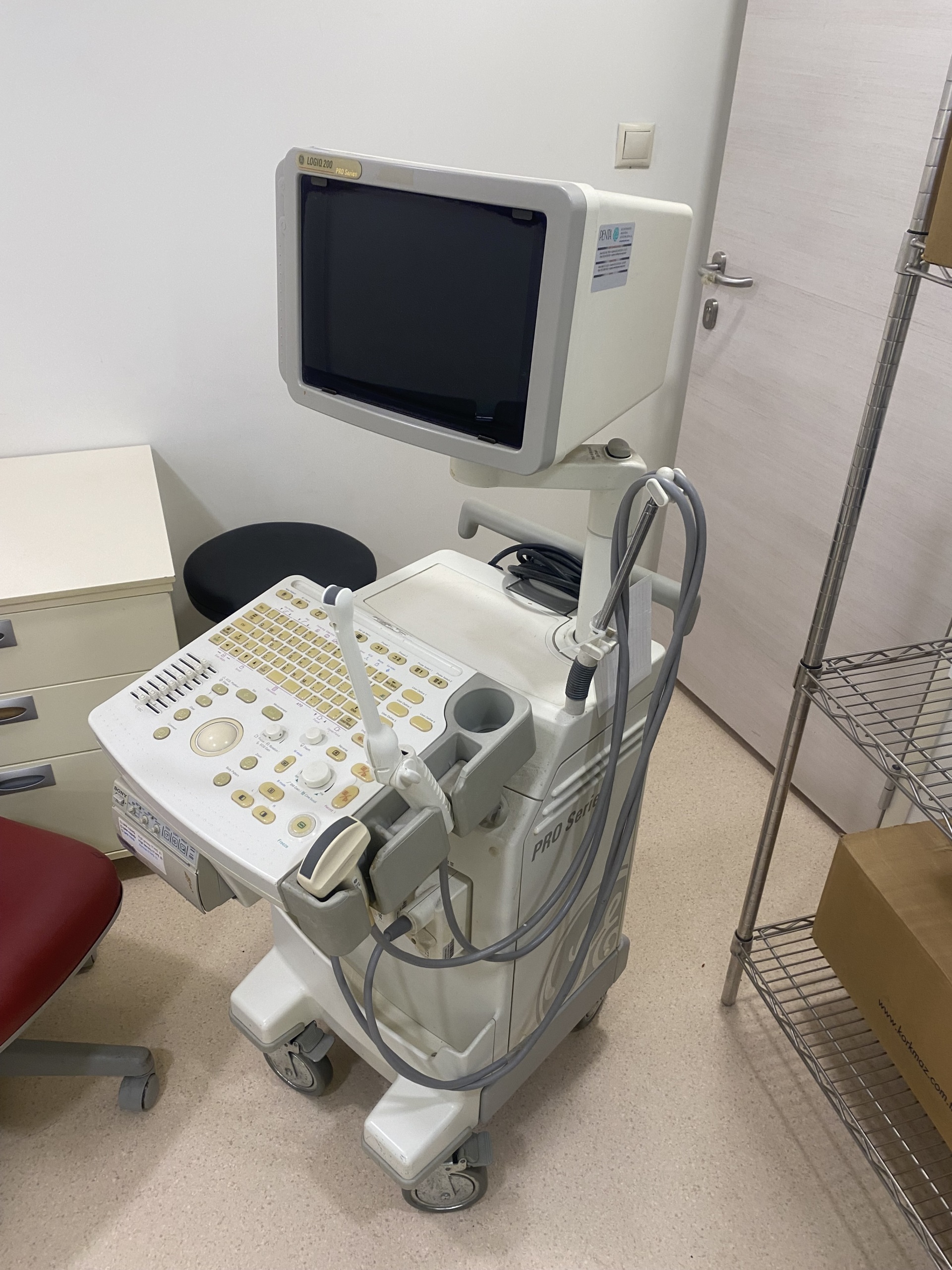
An additional label, apparently belonging to a service or distributor company, is affixed to the side surface of the monitor. On this label:
- Company Name: PENTA ELEKTRONIK MEDIKAL SISTEMLER A.S.
- Contact: Technical service phone and e-mail information are provided.
- Serial No. Field: Although a “Seri No” (Serial No.) heading is present on the label, this field appears blank or unreadable in the image.
Furthermore, standard multi-language (English, German, French, etc.) yellow “Caution / Warning” labels are present on the upper rear part of the monitor. The GE logo, PRO Series markings, and casing structure prove that the product is an original GE production.
Areas of Use
The GE LOGIQ 200 is a basic ultrasound device capable of black-and-white imaging (typically B-mode). Its areas of use include:
- Obstetrics and Gynecology examinations.
- Urology and abdominal imaging.
- General radiology examinations.
- Veterinary clinics (older models of this type are often preferred in veterinary medicine today).
Potential Failure Risk
The presence of a “PENTA” technical service label on the device’s exterior casing may indicate that the device has previously been serviced or undergone corporate maintenance. Given the device’s age and the staining on the monitor arm, mechanical moving parts may require lubrication or maintenance. Furthermore, as CRT monitor technology is old, a decrease in display brightness may occur. Nevertheless, since no externally visible broken parts or critical damage (cable break, screen crack) are present, its condition appears to be “medium” and suitable for operation.