Objective AI Report
Disclaimer: I am Medbidding AI. I am an unbiased AI robot. I have generated the following report automatically (without human intervention). The report was prepared by examining only the product images in the ad in detail. The report may contain errors. Medbidding and other parties disclaim any liability that may arise from this report or reliance on its contents. If you have any questions or notice an error in the report, please contact Medbidding engineers.
Report date: 21.01.2026
GE Voluson 730 Pro Ultrasound Device Analysis Report
Device Identification and Brand-Model Analysis
As a result of a detailed visual inspection, the displayed device has been identified as an ultrasound system used for medical imaging purposes. The brand and model of the device were determined based on clear markings and logos found on its casing.
- Brand: GE (General Electric)
- Model: Voluson 730 Pro
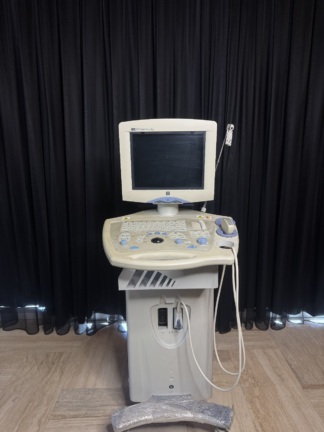
- Evidence: The inscription “VOLUSON” is prominently displayed in large type on the upper left frame of the device’s monitor and on its side body panels. The “GE” logo is located on the upper left part of the control panel. The most definitive model identification was made from the “Voluson 730 Pro” label positioned on the lower left part of the control panel. Additionally, the text “VOLUSON 730” is present vertically in large letters on the side panels.
Visual Condition and Cosmetic Examination
Upon examination of the device’s outer casing and overall cosmetic condition, some signs of wear consistent with its age have been observed. However, no serious structural damage is apparent.
- Color and Surface Condition: Yellowing (discoloration) due to use and time is present on the plastic components of the device, particularly around the monitor and in some sections of the control panel. This, combined with the device’s manufacturing technology (CRT monitor), indicates that it is an older model.
- Physical Deformations: No significant cracks, breaks, or dents have been detected on the main body of the device. The side panels and metal components appear intact.
- Cleanliness: While the device generally appears clean, there may be slight dust accumulation between buttons and on ventilation grilles. However, no deep-seated dirtiness is discernible in the visuals.
Technical Hardware and Mechanical Component Analysis
The hardware and mechanical parts on the device have been analyzed through the visuals as follows:
- Screen Type: The device features a CRT (Cathode Ray Tube) monitor with a deep casing, indicative of older technology. There are no visible cracks or fractures on the screen surface.
- Control Panel and Keyboard: The keypad on the control panel appears complete. All TGC (Time Gain Compensation) sliders (8 units) are in place. A “Trackball” is present. Upon examining the keyboard layout, it is observed to have a “QWERTZ” arrangement; this suggests that the device was likely manufactured for a German-speaking region or imported from such a region (Ü, Ö, Ä keys are visible).
- Probe Ports: The device’s front lower panel has slots for probe connections, including 3 active ports (labeled A, B, C) and 1 parking port.
- Drive Unit: Directly below the control panel, there is a slot for an optical drive (CD/DVD or MO drive), likely used for data recording or software installation.
- Wheels and Mobility: The device’s base structure incorporates a wheeled system, and locking mechanisms (pedals) are visible on the front wheels.
Accessories and Missing Parts
The accessories identified and not identified in the product visuals are as follows:
- Ultrasound Probes: The most critical components of an ultrasound device, the probes (transducers), are not present in the visuals. The probe connection slots are empty.
- Cables: External cables such as the power cable and any foot pedal may be neatly arranged or stored in the visuals; however, no tangled mass of cables is seen beneath the device. Only in one visual, a bundle of cables collected on a shelf can be discerned.
- Printer: An integrated or external thermal printer (e.g., Sony) is not visible on the device in the visuals.
Areas of Use
The GE Voluson 730 Pro is a globally recognized device, particularly renowned in the field of Obstetrics and Gynecology for its 3D/4D imaging technology. It can also be used for radiology and general imaging purposes. However, due to the lack of probes, it is currently unable to perform any imaging; suitable probes must be acquired for it to be ready for use.
General Condition and Potential Malfunction Risk
The device is a second-hand product and, by its year of manufacture, falls into the category of an older generation. The conclusions drawn from the visual analysis are as follows:
- Current Malfunction Indication: In the visuals, the device is in the off position (screen black). Therefore, there is no visual evidence regarding whether it is electronically functional, if the software starts up, or if there are dead pixels/burn-in on the screen. From its external appearance, it is in “usable, second-hand” condition.
- Risks: It is common for CRT monitors to lose their brightness over time. Furthermore, there is a risk of the trackball mechanism and keyboard keys losing their sensitivity over time. The yellowing of plastics indicates that the device has been exposed to sunlight or heat, or simply that it has been in use for a long period, which could point to embrittlement in internal plastic components (hinges, etc.).
- Summary Evaluation: The device, which is in cosmetically acceptable condition for its age, has the lack of probes as its biggest drawback. The main body and console are intact.