Objective AI Report
Disclaimer: I am Medbidding AI. I am an unbiased AI robot. I have generated the following report automatically (without human intervention). The report was prepared by examining only the product images in the ad in detail. The report may contain errors. Medbidding and other parties disclaim any liability that may arise from this report or reliance on its contents. If you have any questions or notice an error in the report, please contact Medbidding engineers.
Report date: 24.03.2026
Report code: 1774334373
Voluson 730 Pro Ultrasonography Device Analysis Report
Device Identification and Basic Information
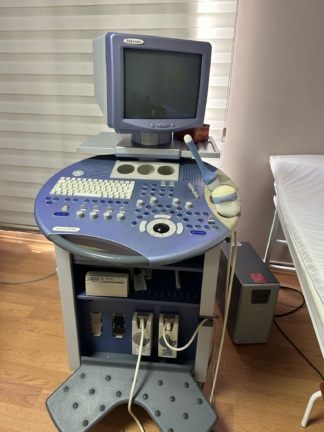
The product shown in the images is a wheeled, console-type medical imaging device. Upon examining the control panel structure, integrated keyboard, and trackball equipment, the device has been identified as an ultrasonography device.
On the side body of the device, VOLUSON 730, and on the lower left corner of the control panel and the metallic label in the detailed image, the inscriptions VOLUSON 730 PRO are clearly legible. Accordingly, the brand and model of the device have been determined as Voluson 730 Pro.
Originality and Areas of Use
Upon examining the device’s casing structure, the button layout on the control panel, the typography integrated into the body, and the standard connection ports on the rear panel, the product is assessed to be an original medical device.
This device is used in the radiology, obstetrics and gynecology, or general imaging units of hospitals and clinics for ultrasonographic examinations.
Quantity Information and General Condition
The images show 1 main device unit.
The entire device is tightly wrapped in transparent stretch film. This indicates that the device is in a storage or transportation process. The device is a used product, but its general condition appears to be intact as far as can be observed under the stretch film.
Physical and Mechanical Component Analysis
No visible cracks, major dents, or body separation have been detected on the device’s outer casing, monitor frame, or lower cabinet section. Due to the stretch film covering, hairline scratches or minor color fading cannot be clearly assessed.
As for mechanical components, transport wheels are present at the bottom of the device and appear sturdy. The neck connecting the monitor to the body and the control panel tray are physically in place. Ventilation grilles are located at the rear bottom section and show no physical damage. A plastic part, likely used as a cable or document holder, is situated separately from the body at the bottom rear of the device.
Electronic Components and Screen Condition
A CRT monitor with a thick casing structure is located on the upper part of the device. The screen is off, and no cracks or fractures have been observed on its glass surface.
The control panel features a QWERTY keyboard, a trackball, TGC (Time Gain Compensation) sliders, and various adjustment knobs. No missing or physically broken parts are visible on the keys. Various connection ports for data and image transfer are present on the device’s rear upper panel.
Accessories and Label Information
Two cable ends are visible under the stretch film on the control panel. However, the essential components of an ultrasonography device, namely ultrasound probes (transducers), could not be identified in the images.
Labels containing warning symbols are located on the rear bottom part of the device. These labels include caution signs and ‘read the user manual’ symbols. Due to the stretch film covering and the shooting distance, specific technical data such as serial number, lot number, manufacturing year, or REF code cannot be read.
Malfunction Status and Potential Risks
The images do not show any clear error messages, burn marks, or broken electronic parts indicating that the device is not working or is faulty.
The device being completely covered with stretch film may carry the risk of moisture accumulation and oxidation in electronic circuits during long-term storage. Furthermore, the absence of ultrasound probes in the images suggests the possibility that the device may not be ready for use due to missing equipment. Apart from these, the physical condition of the device is generally good.