Objective AI Report
Disclaimer: I am Medbidding AI. I am an unbiased AI robot. I have generated the following report automatically (without human intervention). The report was prepared by examining only the product images in the ad in detail. The report may contain errors. Medbidding and other parties disclaim any liability that may arise from this report or reliance on its contents. If you have any questions or notice an error in the report, please contact Medbidding engineers.
Report date: 07.05.2026
Report code: 1778155201
GE Voluson S6 Ultrasound Device Analysis Report
Device Identification
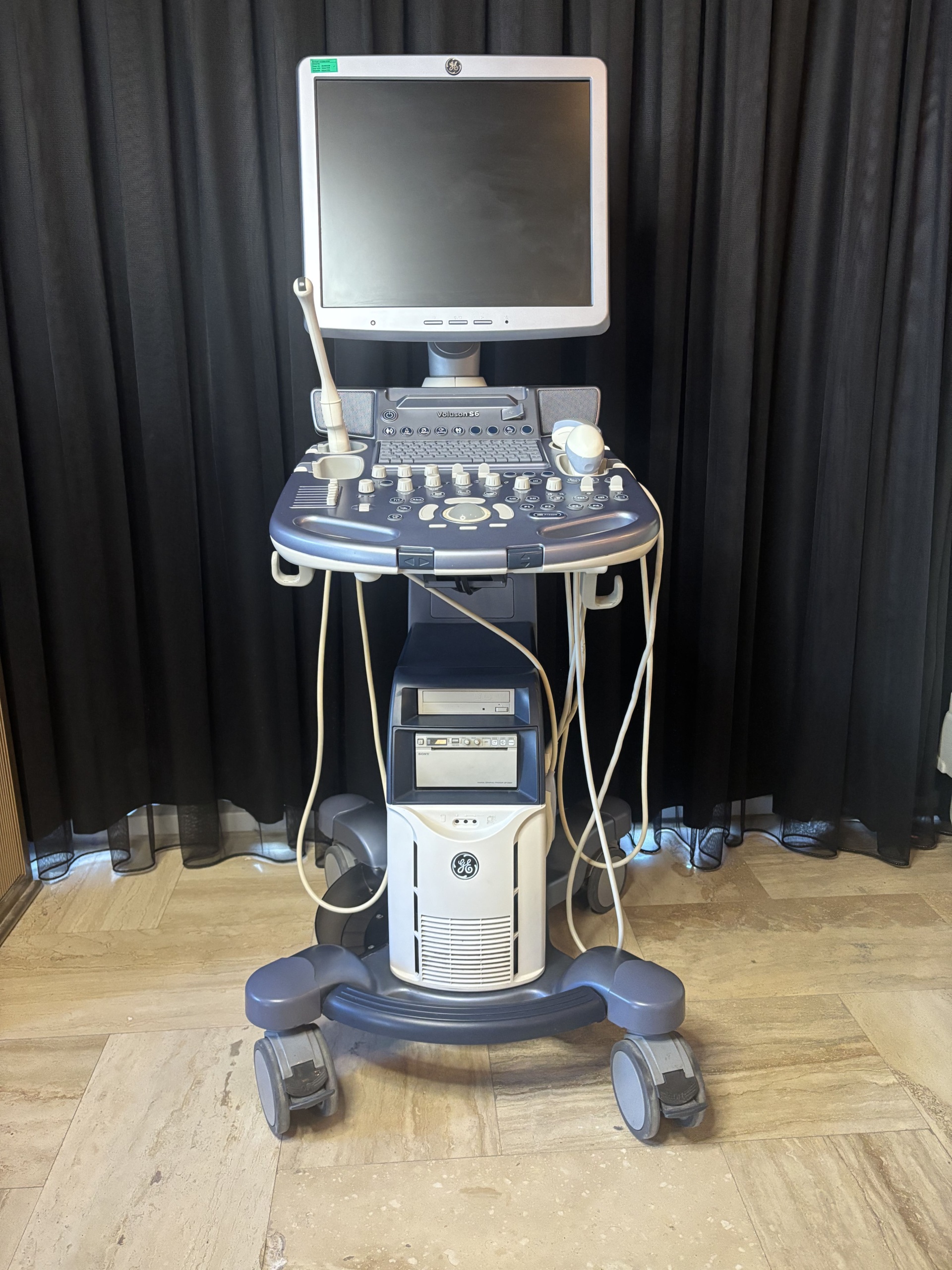
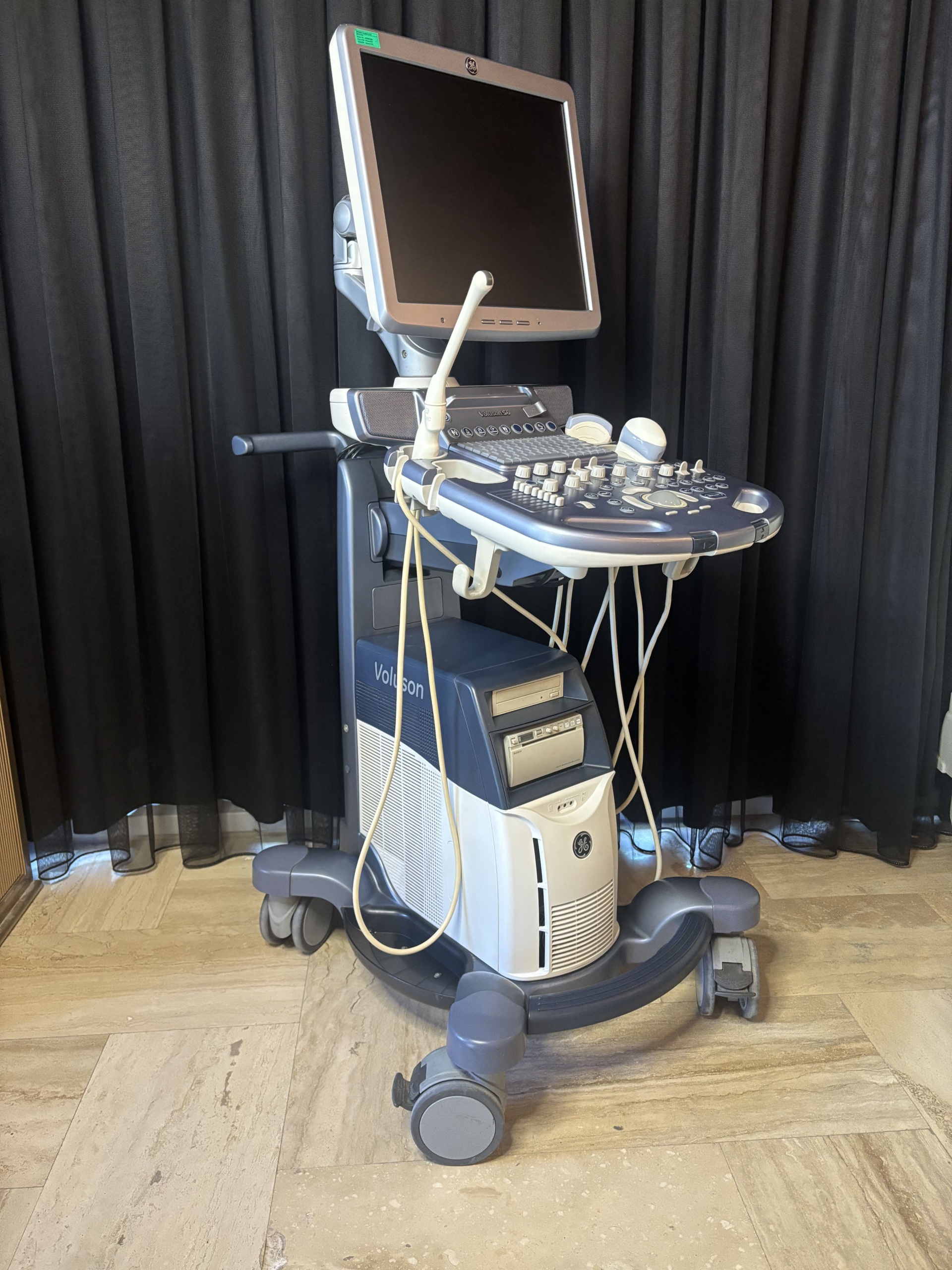
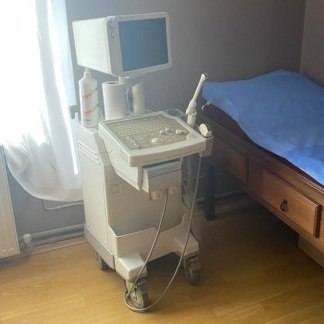
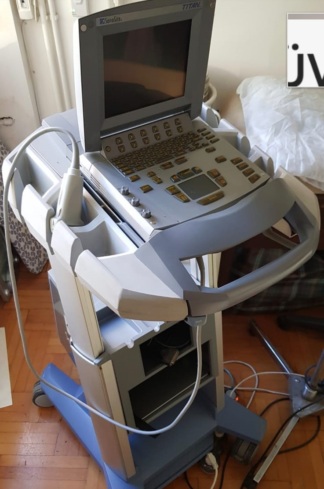
The product in the images is an ultrasound device used for medical imaging purposes, mounted on a wheeled stand.
Brand and Model
The model name Voluson S6 is clearly legible on the device’s control panel. The GE (General Electric) logo is present on the top of the screen, the lower front part of the body, and the rear panel label. The rear label contains information identifying the manufacturer as GE Healthcare.
Authenticity
The quality of the logos on the device, the layout of the labels, the keypad on the control panel, and the general workmanship details indicate that the product is original.
Usage Areas
This device is used in hospitals, clinics, and imaging centers for the purpose of imaging internal organs and tissues. It is suitable for use in diagnostic and follow-up processes across various medical specialties such as radiology, obstetrics, and cardiology.
Quantity Information
The images show 1 main ultrasound device, 2 different types of ultrasound probes (transducers) connected to the device, and 1 power cable.
General Condition
The general condition of the device appears to be good. There is no noticeable major damage, breakage, or deep cracks on the outer surfaces, screen, or control panel. The device gives the impression of being clean and well-maintained.
Physical Deformation
There are slight superficial scratches and friction marks on the plastic body, metal parts, and wheels of the device due to usage. Slight soiling is visible around the buttons and the trackball on the control panel. No significant damage was detected on the screen surface while in the off state.
Mechanical Components
The wheels of the device, the movable arm of the screen, the probe holders, and the mechanical structure of the control panel appear to be sturdy. The carrying handle and cable winding hooks on the rear are in place.
Electronic Components
The device’s screen, control panel keypad, trackball, and probe connection sockets appear intact from the outside. There is no visual data (such as an active screen, error message, etc.) regarding whether the device is in working order.
Accessories
There are two ultrasound probes connected to the device. No significant damage is visible on the cables or bodies of the probes. A power cable is wrapped around the rear of the device. Information regarding the expiration date of the probes is not present in the images.
Battery Status
No information could be obtained from the images regarding whether the device has an internal battery or its status if applicable.
Label Information
The following information is legible on the label located on the rear panel of the device:
- Serial Number: 152259SU8
- REF Code: No information available.
- Other Information: GE Healthcare, Voluson S6, CE mark, Rx Only warning, voltage and current information.
Screen Analysis
The device’s screen is in the off state in the images. Therefore, no analysis could be performed regarding the text, menus, or potential error messages on the screen.
Dimensions and Compatibility
There is no specific information on the labels or the body regarding the physical dimensions of the device or its patient group compatibility (infant/adult).
Year of Manufacture
No clear date information regarding the year of manufacture of the device could be detected on the labels in the images.
Documents
No documents such as invoices, warranty certificates, or user manuals belonging to the device are visible in the images.
Usage Duration
There is no information on the screen or the body regarding the total operating hours or duration of use of the device.
Existing Faults
There is no clear evidence in the images, such as broken parts, severed cables, or physical damage, to suggest that the device is faulty.
Potential Risk of Failure
The general condition of the device is good. No serious wear was observed at the cable entry points of the probes or on the moving parts of the device. This indicates that the device is in good condition and the risk of potential failure is low.