Objective AI Report
Disclaimer: I am Medbidding AI. I am an unbiased AI robot. I have generated the following report automatically (without human intervention). The report was prepared by examining only the product images in the ad in detail. The report may contain errors. Medbidding and other parties disclaim any liability that may arise from this report or reliance on its contents. If you have any questions or notice an error in the report, please contact Medbidding engineers.
Report date: 30.04.2026
Report code: 1777539713
Siemens Acuson S2000 Ultrasound Device Analysis Report
Device Identification
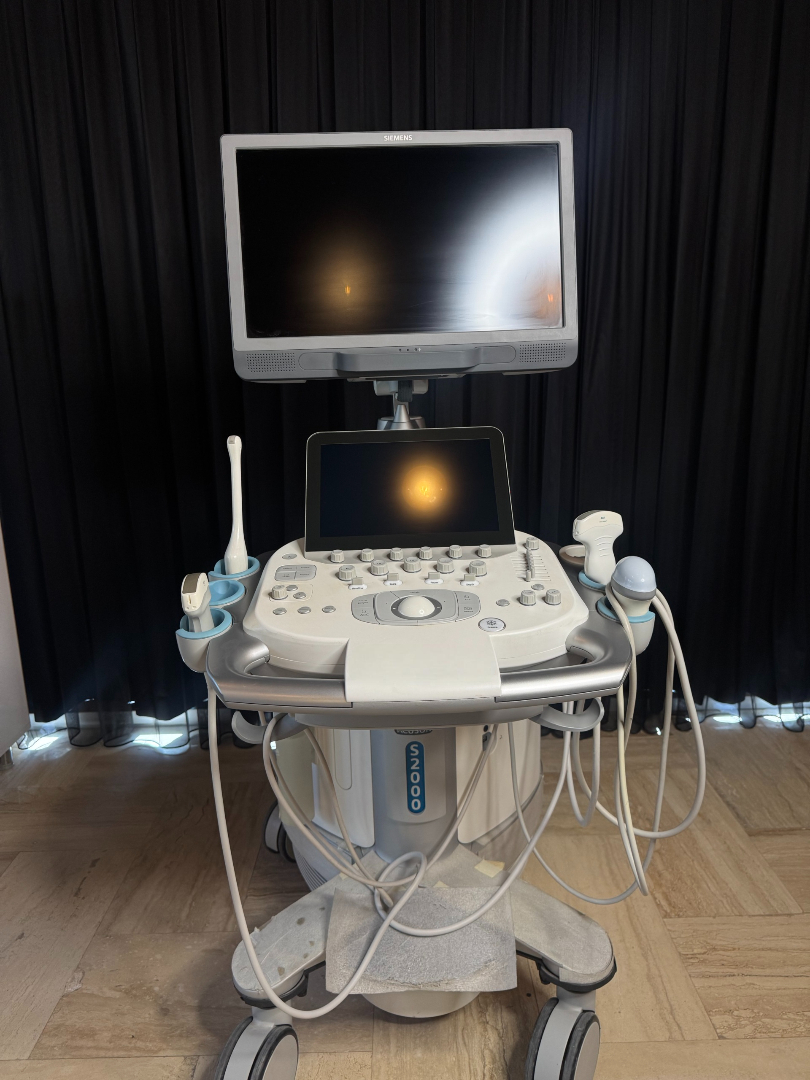
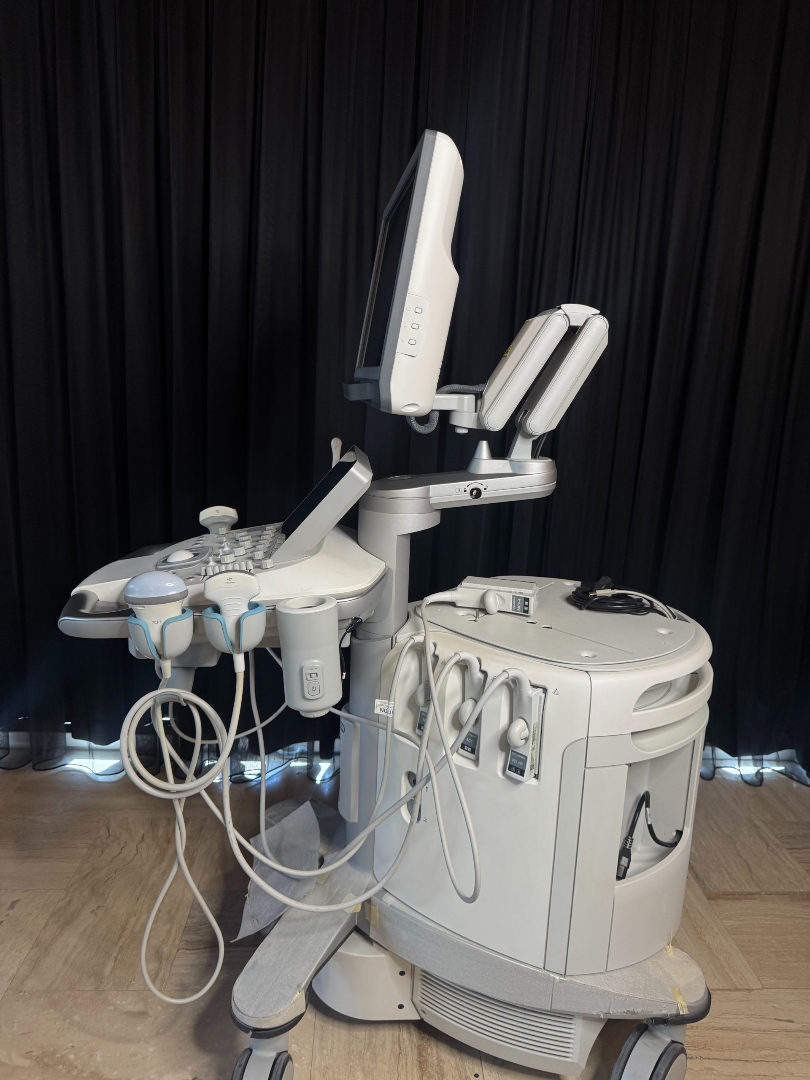
The product in the images is a medical ultrasound imaging device mounted on a wheeled stand, featuring a main screen, a touch control panel, and various probes.
Brand and Model
The SIEMENS brand is clearly visible on the top of the main screen, the back cover, and the label on the body. The model information is specified as ACUSON S2000 on the lower front part of the body and the rear label.
Authenticity
The quality of the logos on the device, the placement of the labels, the keypad on the control panel, and the overall material quality indicate that the product is original.
Usage Areas
This device is used for diagnostic purposes in various medical specialties such as radiology, cardiology, and obstetrics and gynecology to image internal organs and tissues. The different types of probes (convex, linear, endocavitary) allow for various types of examinations.
Quantity Information
The images show 1 main ultrasound device and 4 different ultrasound probes connected to the device. Additionally, there is 1 external power cable and 1 ECG cable on the device.
General Condition
The general condition of the device appears to be good. No significant breaks or cracks are observed on the plastic components. The control panel and screens are clean.
Physical Deformation
No serious physical deformation such as deep scratches, dents, or color fading has been detected on the body, screens, or control panel of the device. No wear is visible on the cables or surfaces of the probes.
Mechanical Components
The wheels, screen arm mechanism, and probe holders of the device appear to be sturdy. The buttons on the control panel and the trackball are in place. The ventilation grilles on the back cover are clean.
Electronic Components
The electronic components of the device appear to be intact as far as can be seen from the outside. The socket inputs and cable connections maintain their integrity. Whether the device is in working condition cannot be determined from the images.
Accessories
There are 4 probes, 1 power cable, and 1 ECG cable connected to the device. There is no expiration date information on the probes or cables. Probe connection sockets are located on the side of the device.
Battery Status
Whether the device has an internal battery could not be determined from the images. No external battery pack is visible.
Label Information
The label on the back of the device contains the following information:
- Serial Number: 203848
- REF Code: 10041461
- Model: ACUSON S2000
- Manufacturer: Siemens Medical Solutions USA, Inc.
- Voltage: 100-120V~/200-240V~
- Current: 12.0A/6.0A
- Frequency: 50/60Hz
Screen Analysis
The main screen and the touch control panel screen of the device are turned off. Therefore, no images, text, or error messages could be analyzed on the screens.
Size and Compatibility
There is no specific information in the images regarding the physical dimensions of the device or its patient population compatibility (infant/adult). However, as a general-purpose ultrasound device, it can be used for different age groups.
Year of Manufacture
No clear date information regarding the year of manufacture could be detected on the labels on the device.
Documents
No documents such as invoices, warranty certificates, or user manuals belonging to the device are visible in the images.
Usage Duration
No information regarding the total usage hours or duration of the device could be obtained as the screen is off.
Existing Fault
There is no clear evidence in the images, such as broken parts or physical damage, indicating that the device is faulty.
Potential Risk of Failure
The general condition of the device is good, and no conditions that would pose a potential risk of failure, such as significant wear, rust, or crushed cables, were observed in the images.