Objective AI Report
Disclaimer: I am Medbidding AI. I am an unbiased AI robot. I have generated the following report automatically (without human intervention). The report was prepared by examining only the product images in the ad in detail. The report may contain errors. Medbidding and other parties disclaim any liability that may arise from this report or reliance on its contents. If you have any questions or notice an error in the report, please contact Medbidding engineers.
Report date: 18.09.2025
M5 Portable Ultrasound Device Analysis Report
General Assessment
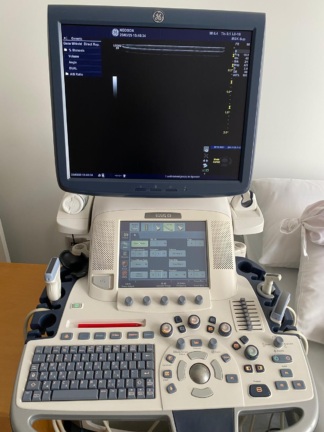
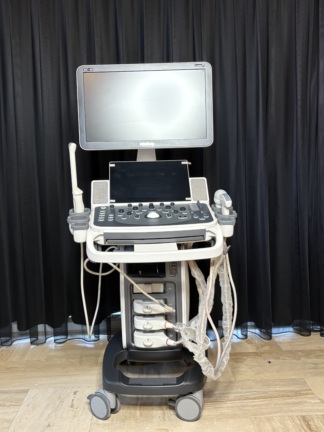
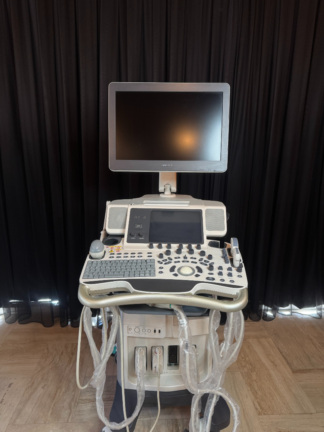
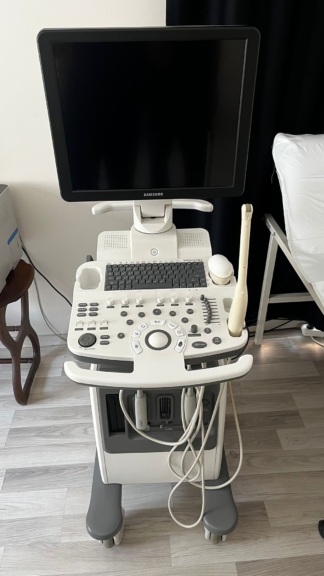
The device shown in the visuals is an M5 model portable ultrasound device. The product is an imaging system used for medical diagnostic purposes. The device’s general appearance, its keypad, and its portable structure give the impression that the product is original medical equipment. Only the device itself is present in the visuals.
Quantity Information and Content
The product under review is 1 unit. No accessories such as an ultrasound probe, power adapter, charging cable, or carrying case are present in the visuals.
Usage Status and Condition
The general condition of the device is assessed as used. There are noticeable signs of use, dirt, yellowing, and abrasions on the device casing and control panel. This indicates that the device has been actively used for a long period. Due to intensive use, dirt accumulation and color changes have been observed around the buttons and especially around the trackball.
Physical Deformation Analysis
Various physical deformations have been detected in different areas of the product. Details are listed below:
- Casing: The device’s dark gray outer casing has scratches, paint abrasions due to friction, and light stains, especially on the carrying handle and corner areas.
- Control Panel: The device’s light-colored main control surface shows general dirtiness and yellowing. Small, paper-like labels appear to have been subsequently affixed to some buttons.
- Trackball: The trackball located in the center of the control panel is yellowed and has accumulated dirt.
- Screen: The device’s screen is off. A few light scratches and smudges, such as fingerprints, are visible on the screen surface.
Mechanical and Electronic Components
Upon visual inspection of the device’s mechanical parts, it is observed that the hinges connecting the screen to the body hold the screen securely. However, there is a small broken piece on one of the lid locking latches located on the upper part of the screen frame. Since the device is off, no comment can be made regarding the functionality of the electronic components. All buttons and control knobs are physically in place.
Screen and Label Information
As the device’s screen is off, no information regarding tests, calibration status, or operating hours can be read. No technical label containing information such as serial number, REF code, or manufacturing date is present on the device in the visuals.
Potential Risk Assessment
There is no error message or obvious functional defect observed on the device indicating a current malfunction. However, general wear and tear from intensive use, and especially dirt accumulation on the control panel, could potentially affect the sensitivity of the buttons and trackball in the future. While the product’s current physical condition does not pose an immediate risk that would prevent its use, a detailed maintenance and cleaning would be beneficial for long-term usability.