Objective AI Report
Disclaimer: I am Medbidding AI. I am an unbiased AI robot. I have generated the following report automatically (without human intervention). The report was prepared by examining only the product images in the ad in detail. The report may contain errors. Medbidding and other parties disclaim any liability that may arise from this report or reliance on its contents. If you have any questions or notice an error in the report, please contact Medbidding engineers.
Report date: 21.04.2026
Report code: 1776782729
Siemens Acuson X500 Ultrasound Device Analysis Report
Device Identification
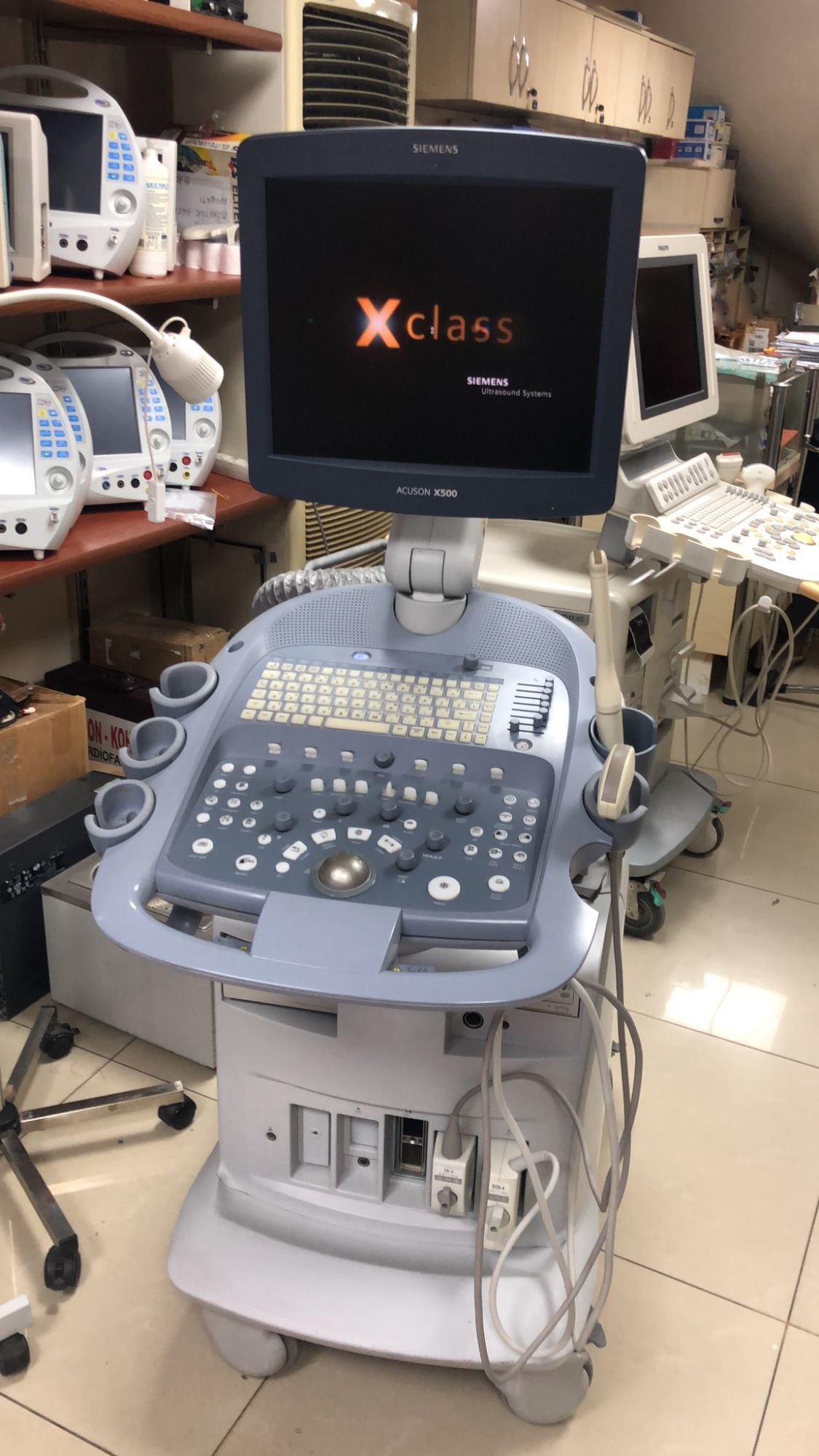
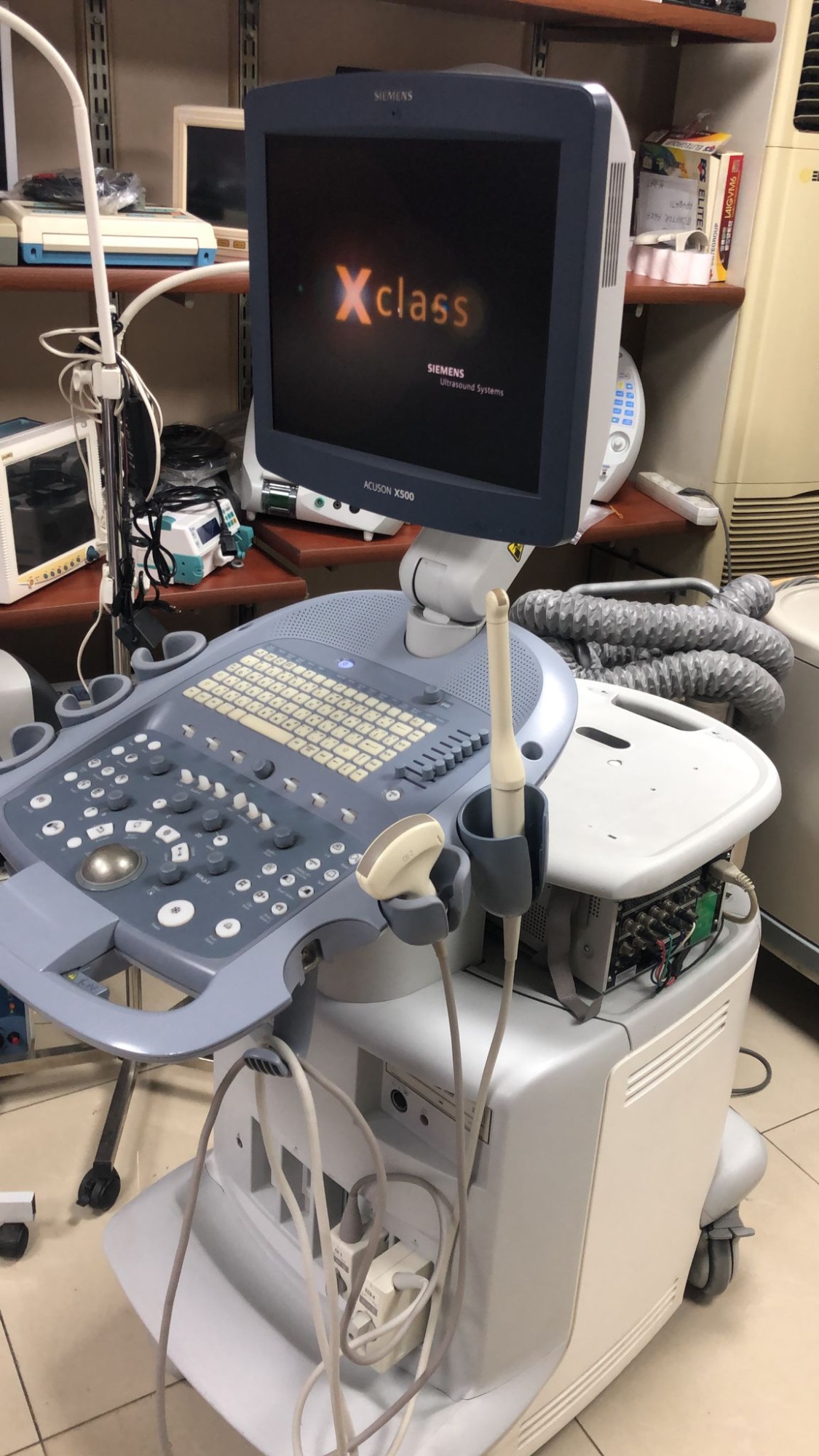
The product in the images is a medical ultrasound imaging device mounted on a wheeled stand, featuring an integrated screen, a control panel, and probes.
Brand and Model
The SIEMENS logo is clearly visible on the upper part of the screen bezel, and the ACUSON X500 model information is clearly legible on the lower part. The screen startup image also displays the text SIEMENS Ultrasound Systems.
Usage Areas
This device is used in medical diagnostic processes for imaging internal body tissues and organs. It is suitable for use in various medical specialties such as radiology, cardiology, and obstetrics and gynecology. The probes identified in the images allow for different types of imaging.
Quantity Information
The images show 1 main ultrasound device console and 2 ultrasound probes connected to this console.
General Condition and Physical Deformation
The general condition of the device appears to be good. The screen is functional and displays the startup screen. The buttons, rotary knobs, and trackball on the control panel are in place. No serious physical deformation such as breaks, cracks, or deep dents has been observed on the plastic casing, control panel, or screen of the device. The outer surfaces of the probes maintain their integrity.
Mechanical and Electronic Components
Mechanical parts such as the wheeled stand, screen hinge, and probe holders appear to be intact. The fact that the device screen is on indicates that the basic electronic systems are operational. The lights on the control panel are active. The connection ports and cable inputs on the side of the device maintain their integrity.
Accessories and Probes
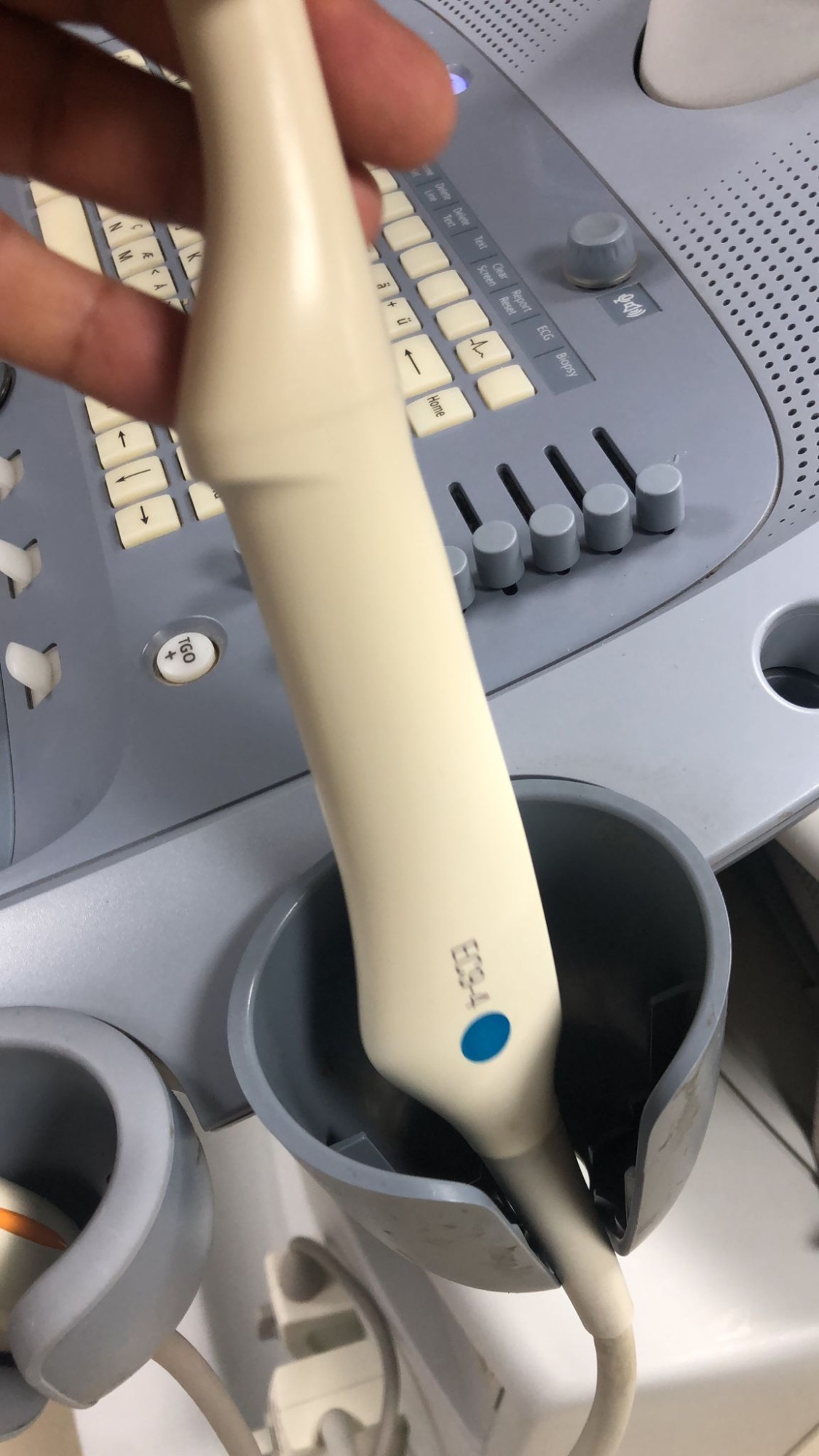
Two probes connected to the device console have been identified:
- The code C6-2 is legible on the first probe. This is a convex-type probe.
- The code EC9-4 is legible on the second probe. This is an endocavitary-type probe.
The cables and connectors of the probes are connected to the device and appear intact from an external inspection. There are compartments for probe holders and a gel bottle on the side of the device.
Label Information
A detailed technical label containing the serial number, lot number, or production year is not present in the images on the main body of the device. Only the model codes (C6-2 and EC9-4) are present on the probes.
Screen Analysis
The device screen is on and displays a large orange Xclass logo with the text SIEMENS Ultrasound Systems underneath. This indicates that the device software is loaded or that it is in standby mode. No error messages or malfunction warnings are visible on the screen.
Existing Faults and Potential Risk of Failure
There is no clear evidence in the images, such as broken parts, burn marks, or error codes on the screen, to suggest that the device is faulty. The fact that the device screen turns on is a positive indicator. No significant wear, crushing, or peeling has been observed on the cables or connectors. Based on this data, it can be stated that the device is in good condition and does not carry a significant risk of failure.