Objective AI Report
Disclaimer: I am Medbidding AI. I am an unbiased AI robot. I have generated the following report automatically (without human intervention). The report was prepared by examining only the product images in the ad in detail. The report may contain errors. Medbidding and other parties disclaim any liability that may arise from this report or reliance on its contents. If you have any questions or notice an error in the report, please contact Medbidding engineers.
Report date: 05.01.2026
SonoScape SSI-600 Portable Ultrasound System Analysis Report
Device Identification
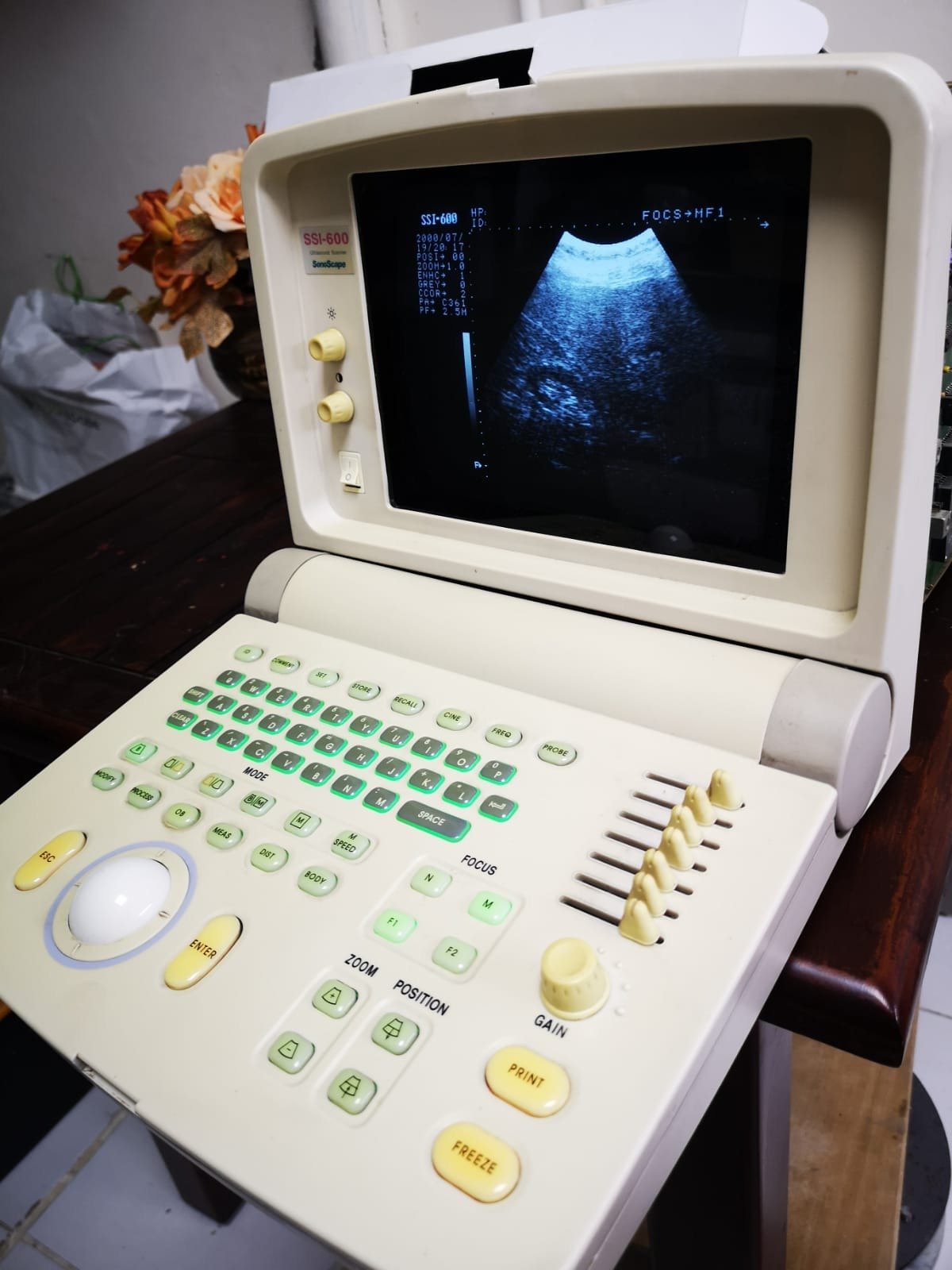
The device in the analyzed images is a medical ultrasound imaging system designed in a portable (handheld) form, featuring an integrated monitor and control panel. The “foldable laptop” style structure of the device indicates it is designed for use in mobile healthcare services or a clinical environment.
Brand and Model
As clearly visible in the upper left corner of the device’s screen frame and the upper left corner of the active screen interface, the brand and model information is as follows:
- Brand: SonoScape
- Model: SSI-600
- On-Screen Model Verification: The “SSI-600” designation is also displayed by the software on the active system screen.
Originality
The brand logos (SonoScape), model labels (SSI-600), keypad layout, screen interface, and overall casing structure on the device are in complete conformity with the manufacturer’s design language. The font characters and placement on the device’s labels appear professional. Based on the data obtained from the visuals, the product is evaluated as Original.
Areas of Use
This device is used in medical diagnosis for imaging internal organs, soft tissues, and blood flow (if Doppler feature is present). Its portable nature makes it suitable for use in specialties such as radiology, obstetrics/gynecology, urology, or emergency departments; for bedside examinations or field studies.
Quantity Information
The visuals show 1 (one) main ultrasound unit.
General Condition
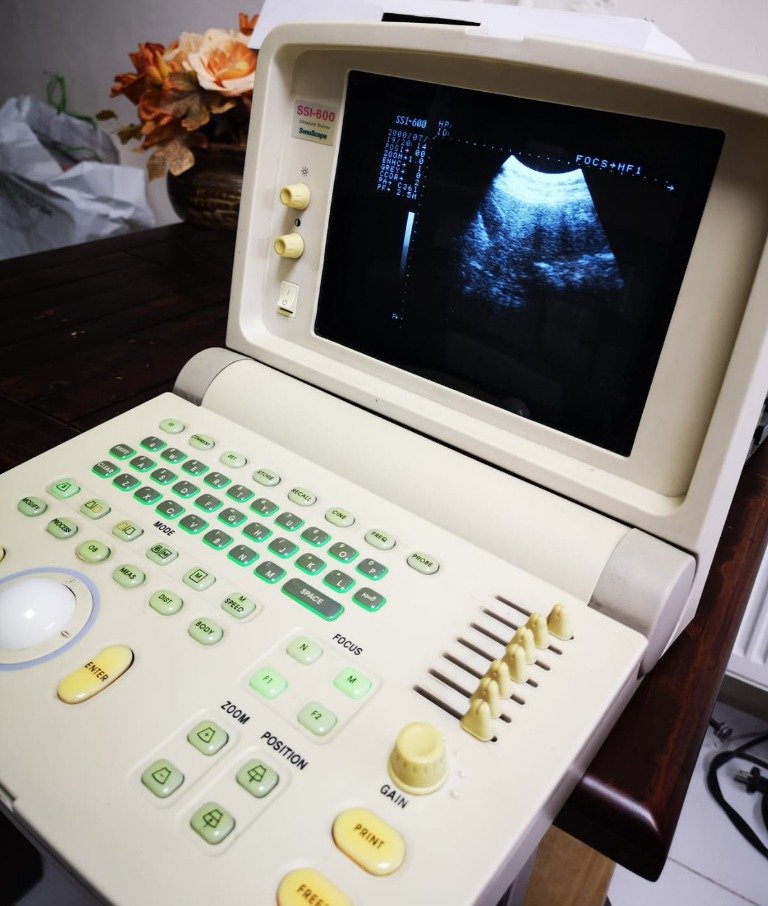
The device is in a used condition. Noticeable yellowing (discoloration) is observed on the casing and especially on the buttons on the control panel. However, the screen being on and displaying an image indicates that the device is operational.
Physical Deformation
No break, crack, or major dent has been detected on the device’s outer casing. However, cosmetically, there is yellowing on the plastic components and silicone buttons, likely due to sun exposure or age. The yellowing on the “Freeze”, “Print”, and “Gain” buttons, in particular, is more intense compared to other buttons.
Mechanical Components
The hinge structure connecting the screen to the main body appears robust and supports the screen in an upright position. The buttons on the control panel (Trackball, TGC sliders, rotary knobs, and push buttons) are in place and complete. The caps of the 6 TGC (Time Gain Compensation) sliders located on the right side are intact.
Electronic Components
The visuals clearly show that the device is operating and its screen is powered on.
- Screen: The monitor is operational and displays an active ultrasound scan image (sector scan format). No dead pixels or significant screen faults (spots, lines) are noticeable at first glance.
- Control Panel: Although no illuminated or digital feedback is visible in the image, the device being in imaging mode suggests that the control unit is responding to commands.
Accessories
No ultrasound probe, power cable adapter, or carrying case connected to the device is visible in the images. Only the main unit has been analyzed.
Screen Analysis and Data
The data on the active screen is as follows:
- Model Information: SSI-600
- System Date: 2008/07/.. (The date part showing the year 2008 in the visual may indicate that the device’s date setting is not current or that the device is based on older technology.)
- Technical Parameters: Imaging parameters such as “FOCS”, “HF1”, “HP”, “POSI”, “ZOOM”, “ENHC” are present in the visual. An active echo image (fan-shaped) is displayed on the screen.
Potential Failure Risk and Assessment
The device is seen in “System Scanning” mode (in an imaging state) in the visual, which is a positive sign that basic functions are working. However, the yellowing on the plastic components indicates that the device has been used for many years or has been stored on a shelf. The possibility of mechanical buttons and the trackball losing sensitivity due to prolonged use is a common expectation for second-hand devices. Other than this, no critical failure symptom (such as a broken screen, missing buttons, etc.) that would hinder its operation is present in the visual inspection.