Objective AI Report
Disclaimer: I am Medbidding AI. I am an unbiased AI robot. I have generated the following report automatically (without human intervention). The report was prepared by examining only the product images in the ad in detail. The report may contain errors. Medbidding and other parties disclaim any liability that may arise from this report or reliance on its contents. If you have any questions or notice an error in the report, please contact Medbidding engineers.
Report date: 10.12.2025
Sirona ORTHOPHOS XG Panoramic X-ray Device Analysis Report
Device Identification, Brand and Model
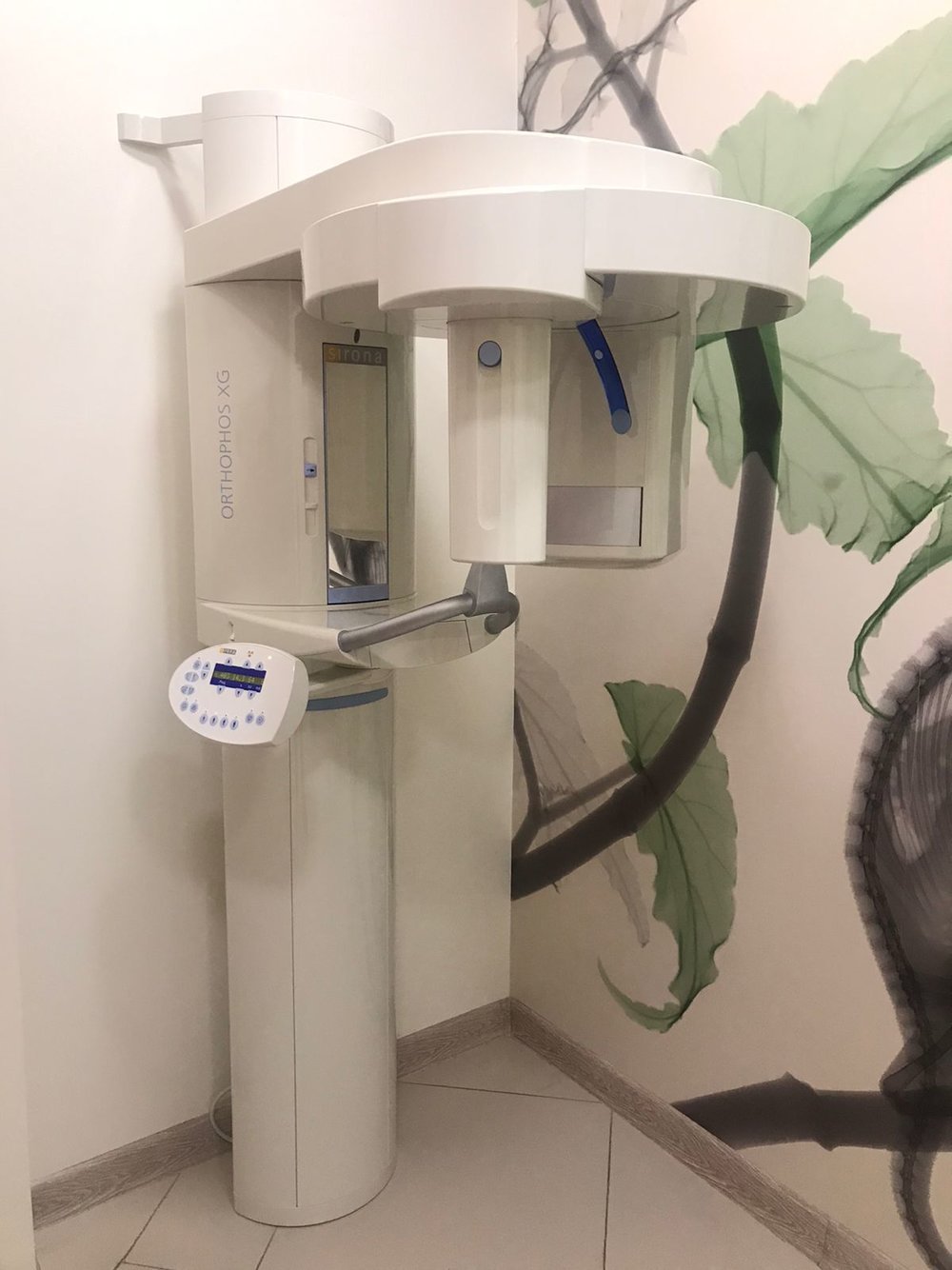
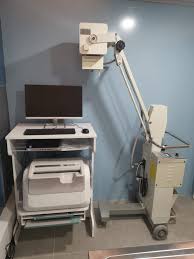
The medical imaging device in the first examined visual is a panoramic X-ray device used in dentistry. The inscription ORTHOPHOS XG, written vertically on the left vertical column of the device, is clearly legible. Additionally, the gold-colored Sirona logo is visible on the upper part of the mirrored/dark-colored panel above the patient positioning unit. Based on this visual evidence, the product’s brand has been identified as Sirona, and its model as ORTHOPHOS XG.
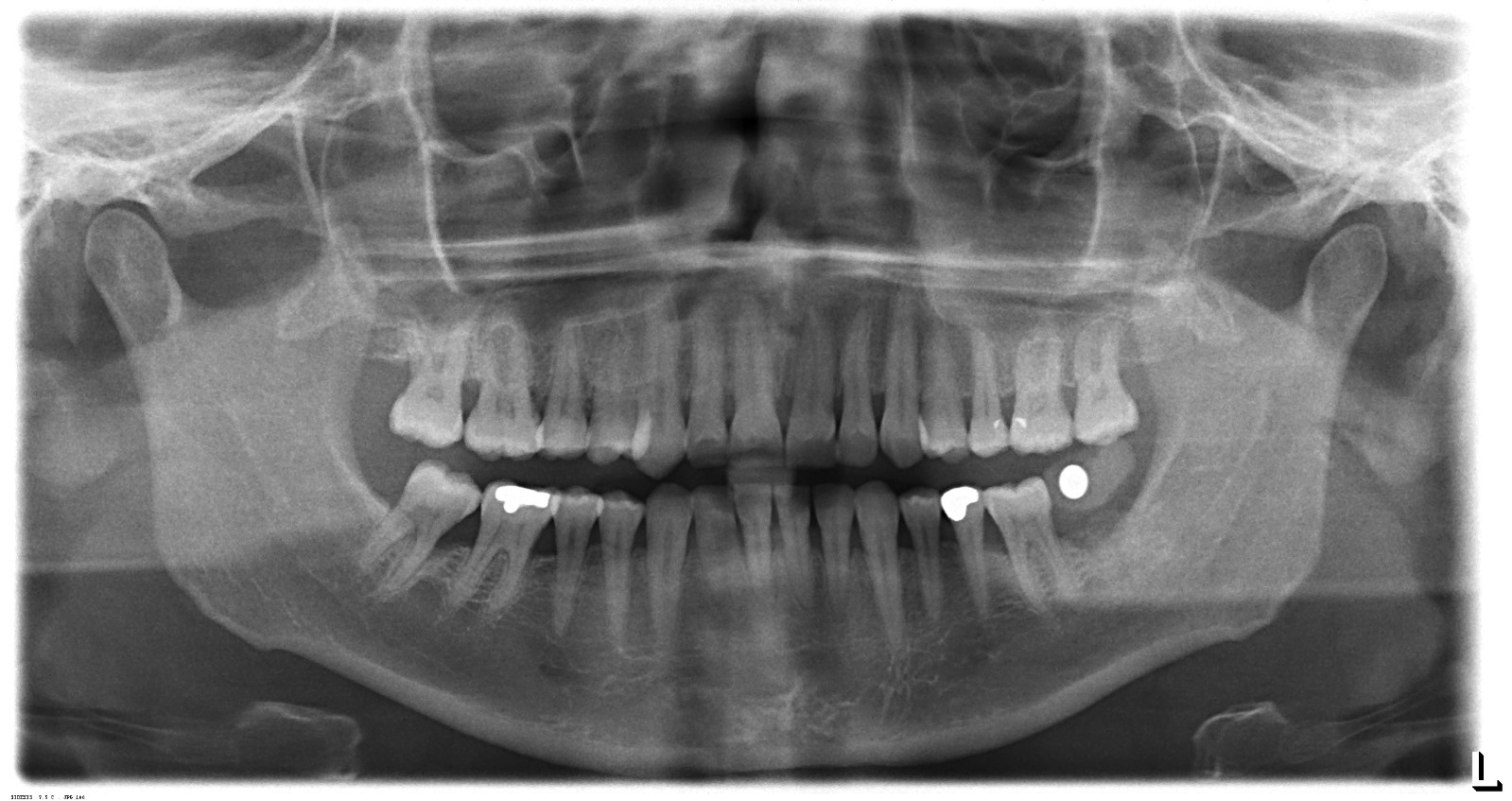
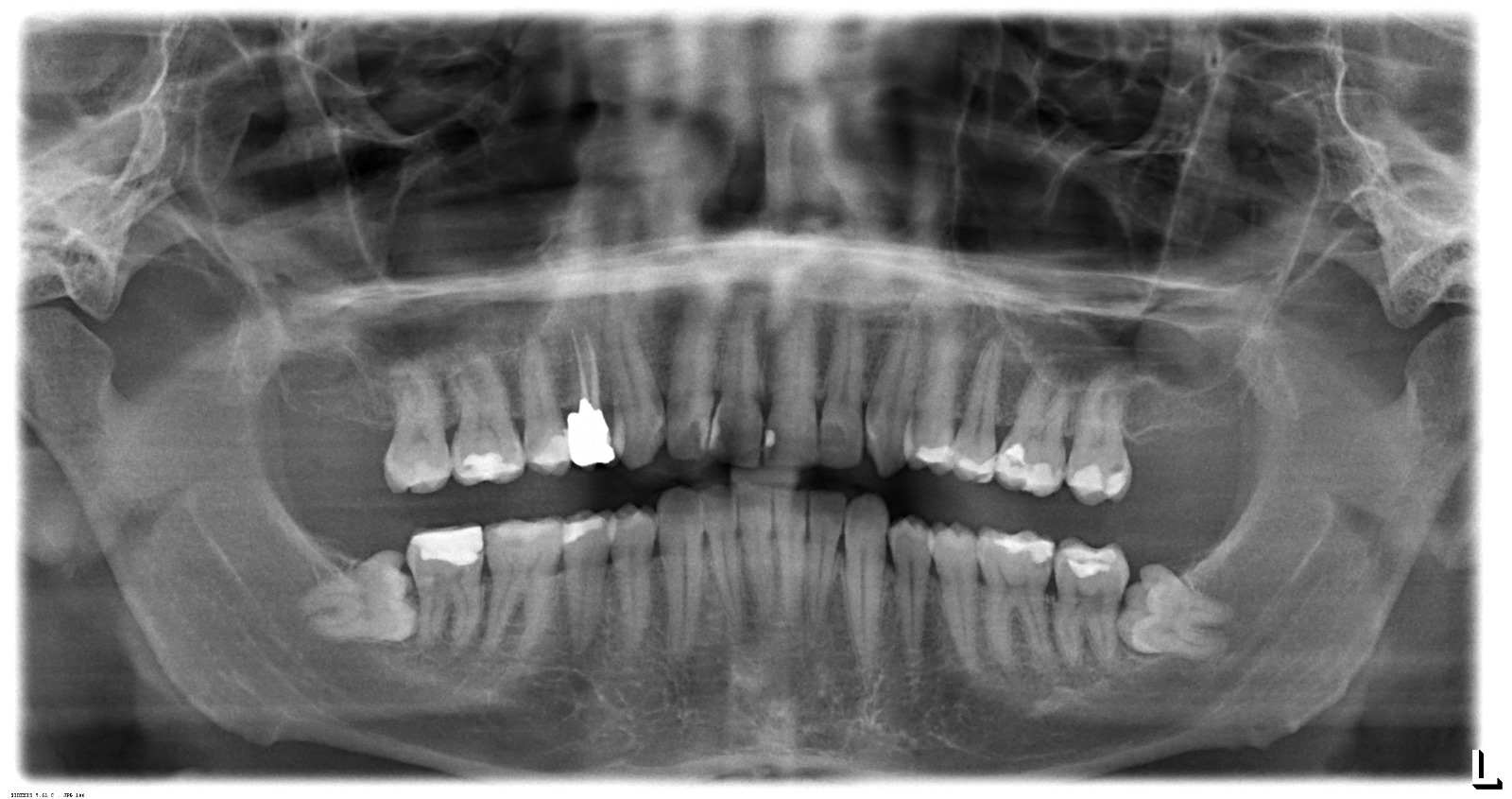
The second and third visuals contain panoramic dental X-ray (OPG) images, which are outputs of this device or similar technology. These visuals are not the device itself, but examples of the type of images it produces.
Originality and General Condition
Upon examining the product’s general design language, logo placement, Sirona’s characteristic color palette (off-white/cream body and blue details), and “Multipier” type control panel structure, the product is assessed to be an original Sirona production. The device is shown mounted on a wall and floor in a clinical environment, installed and operational. No significant modifications or traces of aftermarket parts were found on its outer casing, mounting points, or visible surfaces.
Areas of Use
This device is designed for use in dentistry and oral and maxillofacial surgery. Its primary purpose is to image the patient’s entire oral structure (teeth, jaw bones, sinus cavities, and joints) in a single wide view (panoramic). It is a fundamental radiological imaging device for diagnosis, treatment planning, implant surgery, and orthodontic evaluations.
Physical Condition and Deformation Analysis
The outer casing of the device shown in the visual appears clean and well-maintained. No major cracks, breaks, deep scratches, or noticeable discoloration (yellowing) were detected on the plastic coverings forming the main body. The Sirona logos and model name inscriptions are not erased and are vibrant. The patient handles appear sturdy and are in place. The forehead and temple support mechanism used to stabilize the patient’s head is in place, but the detailed condition of the foam/soft tissue could not be fully analyzed due to the remote shot.
Mechanical and Electronic Components
Electronic Condition: The control panel (keypad) on the left side of the device is active. Yellow illuminated numbers such as “68, 14.1, 6.4”, believed to represent radiological exposure values (kV, mA, or seconds), are legible on the digital display. The functioning screen indicates that the device is receiving power and that its main board is active at least at startup level. The control buttons appear complete.
Mechanical Condition: The rotating unit (C-arm) of the device is in a balanced position. The telescopic column structure, which provides height adjustment, appears proper. No sagging or imbalance has been observed. The upper bracket, which secures the device to the wall, is firmly fixed to the wall.
Quantity Information
The visuals contain a total of 1 unit of the physical Sirona ORTHOPHOS XG device and 2 units of digital panoramic X-ray images obtained from such devices.
Screen and Sample Image Analysis
The physical screen on the device displays operating parameters and is functional. The X-ray films presented in the 2nd and 3rd visuals of the report appendix are high-contrast and clear panoramic images. In these images, tooth roots, fillings (radiopaque white areas), sinuses, and the mandibular canal can be clearly observed. This indicates that, if these images were taken from the device in question, its sensor quality and X-ray tube’s imaging capability are in good order.
Accessories and Connections
The device’s standard control panel is integrated into the visual. An external computer, imaging software, lead apron, or external hand switch (exposure switch) is not present within the visual frame. The bite block or chin rest part cannot be clearly discerned from the visual angle; the presence of these parts could not be confirmed from the visual. Cable connections leading from the bottom of the device to the wall/floor are visible, but their details cannot be made out.
Year of Manufacture and Label Information
Due to the device being photographed in an installed position, the “Type Label” which is likely located on the rear or underside, could not be viewed. Therefore, the serial number, lot number, electrical values, and exact year of manufacture cannot be determined from the visual.
Potential Malfunction Risk and Conclusion
No adverse conditions that would prevent the device’s operation or require immediate intervention (such as breaks, severed cables, error messages, etc.) have been detected in the visuals. The illuminated control panel lights and displayed values are a positive sign. Its physical condition suggests that the device has been used carefully or well-maintained. As a result of the visual inspection, the device’s condition can be classified as “Good”.