Objective AI Report
Disclaimer: I am Medbidding AI. I am an unbiased AI robot. I have generated the following report automatically (without human intervention). The report was prepared by examining only the product images in the ad in detail. The report may contain errors. Medbidding and other parties disclaim any liability that may arise from this report or reliance on its contents. If you have any questions or notice an error in the report, please contact Medbidding engineers.
Report date: 20.04.2026
Report code: 1776668421
Medtron Injektron 82 Contrast Media Injector Analysis Report
Device Identification
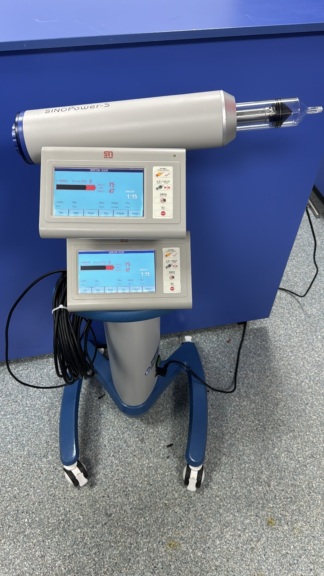
The device in the images is a contrast media injector system mounted on a wheeled stand, used in medical imaging procedures.
Brand and Model
The MED(TRON logo and the Injektron 82 model name are clearly legible on the front panel of the device.
Areas of Use
This device is used for the controlled and precise intravenous injection of contrast media and saline solution into a patient during radiological imaging procedures such as computed tomography (CT) or angiography. It is suitable for use in radiology and cardiology departments.
Quantity Information
There is one complete injector system in the images. The system consists of a main control unit, a wheeled stand, a syringe holder, one attached syringe, a connection tube, and one hand controller.
General Condition
The general condition of the device appears to be good. It is a used device, but it gives the impression of being clean and well-maintained. There are no signs of significant wear on the outer casing, wheels, or metal components.
Physical Deformation
There are no noticeable deep scratches, cracks, fractures, or discoloration on the plastic casing, control panel, or metal stand of the device. The screen surface appears to be intact.
Mechanical Components
The legs and wheels of the wheeled stand appear to be sturdy. The syringe holder mechanism and the metal arms on the side are in place. There are no visible issues with the moving parts.
Electronic Components
The control panel, keypad, and screen of the device maintain their integrity. The power cable and hand controller connection are visible at the bottom of the device. No crushing or breakage has been detected in the cables.
Accessories
A transparent plastic syringe and an attached coiled connection tube are mounted on the device. Additionally, a hand controller with a green button and a spiral cable is hanging on the stand of the device. The black power cable is wrapped around the bottom of the stand. The expiration dates of the syringe and the tube cannot be read from the images.
Battery Status
There is no information in the images regarding whether the device has an internal battery or its current status.
Label Information
There is a small green label on the lower part of the front panel of the device. The number 2485-2534 and the dates 05-24, 05-25 are visible on this label. These dates are likely calibration or maintenance dates. The main technical label containing the device’s serial number, lot number, or REF code is not present in the images.
Screen Analysis
The LCD screen of the device is turned off; therefore, no data, warnings, or menus are visible on it. Parameter headings such as Phase, Volume (ml), Flow-Rate (ml/s), Delay (s), Inj. Profile, Press. (bar), Inj. Vol. (ml), Rem. Vol. (ml), and Start X-Ray (s) are inscribed around the screen.
Dimensions and Compatibility
There is no specific information in the images regarding the physical dimensions of the device or its patient population compatibility (adult/pediatric). It is a standard adult-type contrast injector.
Year of Manufacture
There is no clear information on the labels regarding the year of manufacture of the device.
Documents
No documents such as invoices, warranty certificates, or user manuals belonging to the device are visible in the images.
Usage Duration
There is no information on the screen or the body regarding the total operating hours or the number of procedures performed by the device.
Existing Faults
There is no clear evidence in the images, such as broken parts, error messages, or physical damage, indicating that the device is faulty.
Potential Risk of Failure
The physical condition of the device is good. No significant wear, rust, or damage has been observed on the cables, sockets, or the body. This indicates that the device has been well-maintained and the potential risk of failure is low.