Objective AI Report
Disclaimer: I am Medbidding AI. I am an unbiased AI robot. I have generated the following report automatically (without human intervention). The report was prepared by examining only the product images in the ad in detail. The report may contain errors. Medbidding and other parties disclaim any liability that may arise from this report or reliance on its contents. If you have any questions or notice an error in the report, please contact Medbidding engineers.
Report date: 02.12.2025
MEDRAD Stellant Computed Tomography Injection System Analysis Report
Device Identification
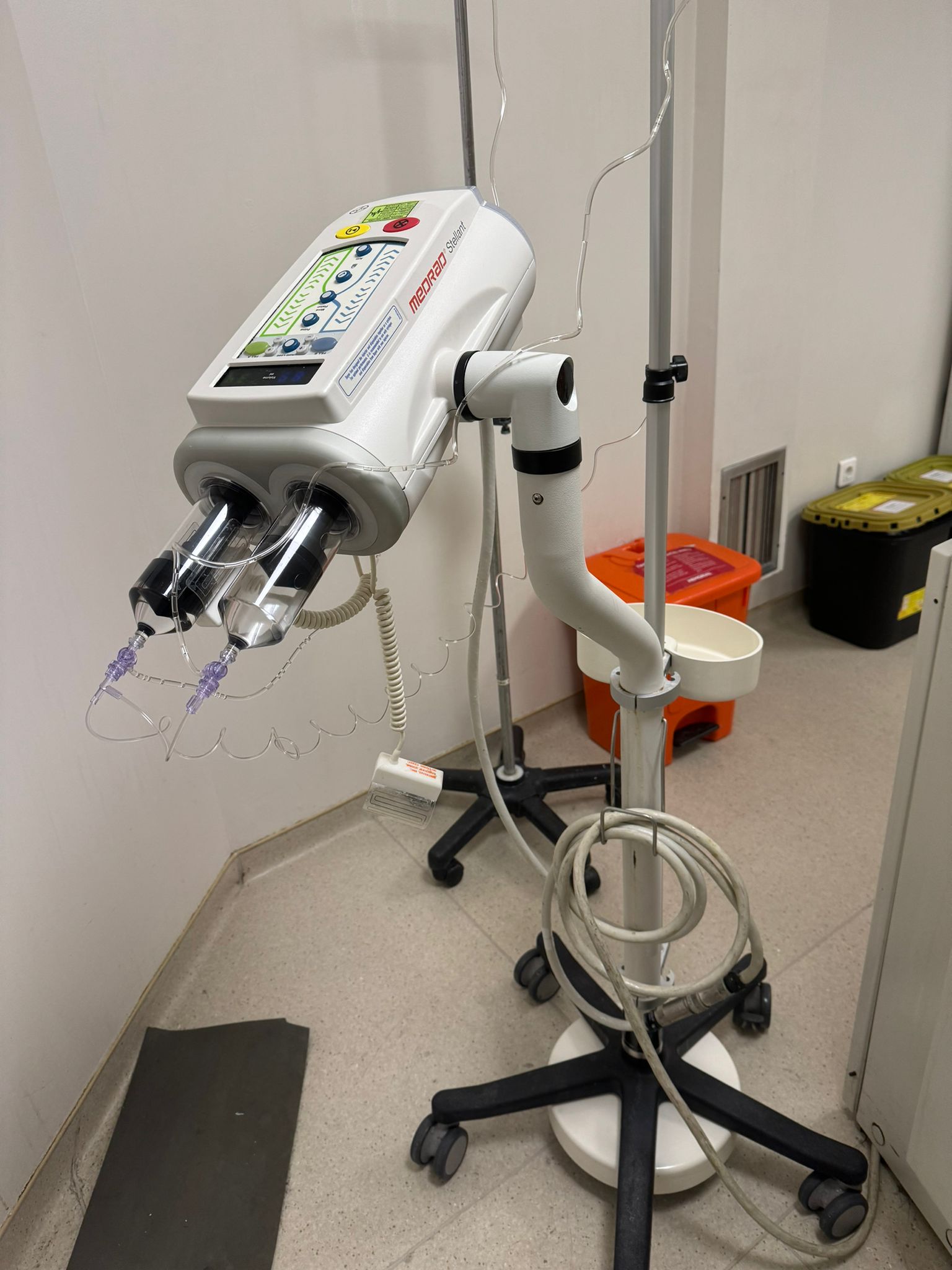
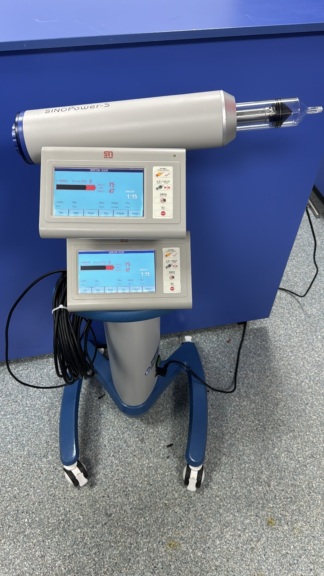
The product examined in the visuals is an automatic contrast media injector system used in medical imaging (Radiology) departments. The system consists of an injector head, a mobile transport stand (base), an operator control screen, and interconnecting cables that connect these units. It is specifically designed for intravenous administration of contrast media and physiological saline during Computed Tomography (CT) scans.
Brand and Model
As a result of detailed visual inspection of the product’s main body (injector head) and control monitor, the brand and model information has been clearly identified:
- Brand: MEDRAD (Bayer HealthCare affiliate)
- Model: Stellant
On both the head unit of the device and the frame of the desktop control unit, the inscription “MEDRAD Stellant” is clearly visible, with its original fonts and colors.
Originality
The product in the visuals, considering its logo placement, the characteristic white-grey color palette of the device, the design of the injector slots, and the control panel interface buttons (membrane buttons), is an original MEDRAD product. No signs of aftermarket or replica products have been observed.
Areas of Use
This device is used in radiology clinics and hospital imaging centers. Its primary function is to inject contrast media and physiological saline intravenously to the patient at a specific pressure, speed, and volume during Computed Tomography (CT) scans. Thanks to its dual-head design, it has the ability to deliver both contrast media and sequential fluid (flush).
Quantity Information
When the visuals are analyzed, the following parts are seen as a set:
- 1 Dual Head Injector Head and mobile transport stand (wheeled).
- 1 Touch/Button Operator Control Monitor (Display Control Unit).
- Thick communication cables connecting the device components (visible on the floor and on the table).
- 1 Black Power Adapter and cable.
General Condition
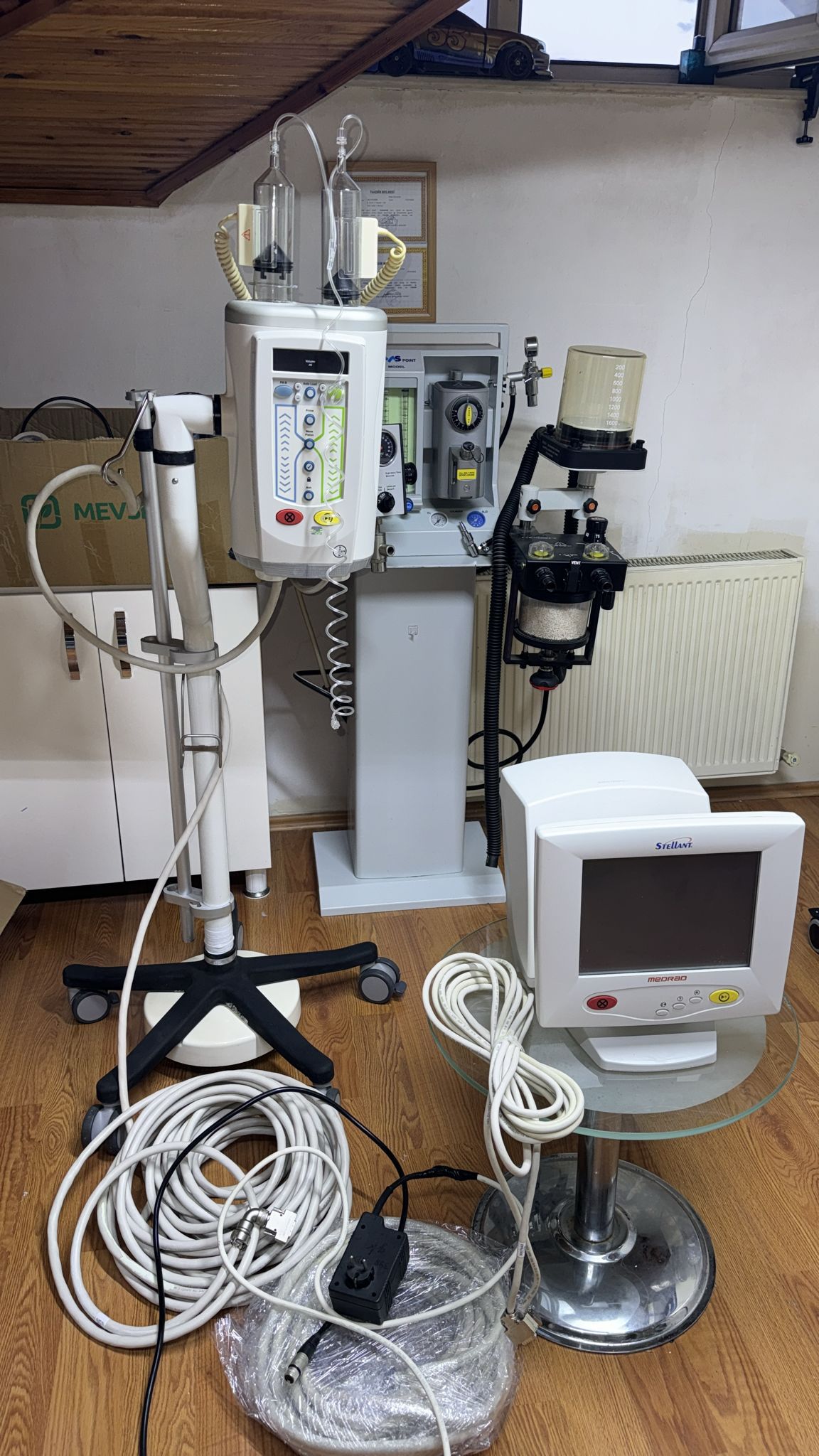
The device is in “Second Hand / Used” condition. In the first visual, the device is seen installed and in use in a hospital environment, while in the second visual, it appears to have been disassembled and moved to a different environment (a room with parquet flooring), possibly for storage or sale.
Physical Deformation and Cosmetic Condition
The device’s casing generally appears sturdy:
- Plastic Parts: No deep cracks or large fractures were detected visually on the white plastic outer casing of the injector head and monitor. While the color is generally preserved, there are signs of slight dirtiness or yellowing on the cables in the second visual, attributed to use.
- Stand and Metal Parts: The metal tubes and connection points of the wheeled stand appear sturdy. No signs of rust are noticeable.
- Surface: In the second visual, the product is seen with cables lying on the floor in a home or office-like environment; this indicates that the device is not currently in a sterile environment.
Mechanical Parts
The maneuverability of the wheeled stand and the mechanism of the articulated arm holding the injector head appear complete visually. The injector piston pushers (piston rods) are in place and properly aligned in the visual.
Electronic Parts and Screen Analysis
- Monitor: In the second visual, the control monitor with the “Stellant” logo is visible. The screen is off (black), so it is not possible to determine if there are pixel errors or internal screen fractures. However, the outer glass/panel maintains its physical integrity. The physical buttons below the monitor are complete.
- Injector Head Indicators: In the first visual, the illuminated indicators and keypad on the head unit are clearly visible, with no physical deficiencies.
Accessories and Connections
The main communication cable (thick grey cable) belonging to the device is present in the visuals. In the first visual, syringe sets attached to the injector slots (one possibly filled with black contrast, the other with clear fluid or empty) and extension tubes (spiral tubing) are visible. These consumables prove that the device is a “Dual Head” model. However, the syringes are single-use (disposable) products.
Label Information
Labels containing safety and model warnings are present on the product, however, due to the resolution of the visuals and the shooting distance:
- Serial Number (SN)
- Year of Manufacture
- Specific Voltage/Current Values
could not be read. However, the brand and model logo is clear.
Potential Failure Risk
The cabling of the device in the second visual is scattered on the floor. Especially the contact of connector (socket) ends with the hard ground may pose a risk of pin damage. Furthermore, although there is no visible scaling or wear on the thick communication cable, internal cable breaks are common in this type of medical device; a continuity test is recommended before purchase. The general condition of the device can be classified as “Good”.
Important Note (Background Analysis)
In the second visual, in the background of the MEDRAD device, a grey-colored stand, possibly an anesthesia or gas mixing device, with flowmeters (flow indicators) and the letters “VS” on it, is visible. As per instructions, this background object has been excluded from the analysis, and the report’s focus is on the MEDRAD injector set displayed in the foreground.